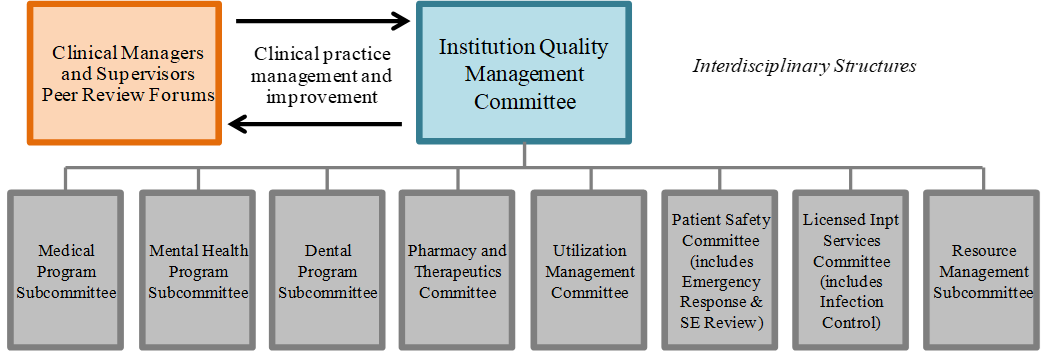
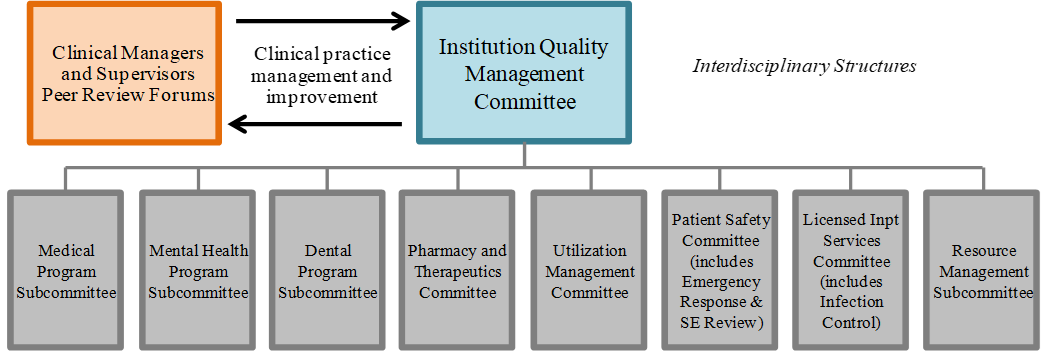
Article 1 – Statewide Health Care Governance
1.1.1 Governing Body
-
Policy
-
The California Department of Corrections and Rehabilitation (CDCR), Division of Health Care Services (DHCS), and California Correctional Health Care Services (CCHCS) [referred to jointly in this policy as “Health Care Services (HCS)”] shall promote organizational decision-making that is standardized and systematic, incorporates a broad range of perspectives, and creates alignment with the strategic vision, mission, and goals of the organization.
-
HCS shall maintain a standardized governance structure, driven by its Governing Body (GB) at the highest level, and consisting of multi-disciplinary leadership teams at headquarters and institution levels that will guide the statewide strategic vision and performance objectives for the organization. The governance structure shall also encompass standardized systems, processes, and procedures for key governance functions including, but not limited to:
-
Strategic planning at the enterprise, region, and institution levels, and within specific programs and departments to ensure that all levels of the organization are aligned in pursuing strategic goals.
-
Elevation of critical program concerns.
-
Making decisions, including mechanisms for gathering input from stakeholders and staff at various reporting levels throughout the organization, and conflict resolution.
-
Developing and vetting statewide policies and guidelines to ensure appropriate multi-disciplinary participation, high-quality products, and acceptance among staff implementing the policies or guidelines.
-
Communicating important organizational messages quickly and efficiently throughout the organization.
-
Routinely evaluating and improving program performance and holding leadership accountable for outcomes.
-
Sustaining processes that work well.
-
Providing orientation, ongoing development, and feedback for key leaders within the organization.
-
Maintaining the quality and safety of patient care through performance evaluation and improvement including peer review, quality management, and patient safety and risk management systems.
-
-
-
Purpose
-
To:
-
Ensure delivery of appropriate, quality health care in a cost-effective manner with minimized risk to patients;
-
Manage and consider the needs and expectations of stakeholders;
-
Respond appropriately to changes in government and the health care industry, including changes in patient demographics and case mix, financial constraints, advances in research and technology, public expectations, and legal and regulatory mandates;
-
Preserve quality of care and economic viability over time; and
-
Adhere to federal and state laws and regulations.
-
-
Responsibility
-
The Undersecretary, Health Care Services, and Directors of CCHCS are responsible for statewide planning, implementation, and evaluation of the governance structure and processes.
-
HCS departmental leadership at all levels of the organization, within the scope of their authority, are responsible to support and participate fully in the governance structure and processes and to ensure their staff are aware of and adhere to governance processes and procedures.
-
-
Procedure Overview
-
The GB is the highest-level committee in the network of headquarters and institution committees that makes up the HCS organizational governance structure and has ultimate oversight but delegates decision authority to the local GBs and other committees as needed. Within the HCS Governance Structure, the GB is ultimately accountable for quality patient care and treatment and services provided by employees and contractors. Among other tasks, the GB defines the strategic direction of the organization, determines the organizational structure and delivery system design, ensures that the organization has sufficient resources to accomplish strategic goals including prepared and competent staff, and continuously evaluates and improves performance ensuring accountability, particularly among key leaders and licensed clinicians.
-
The membership of the GB is comprised of the highest-level executives within HCS as well as the leaders from essential stakeholder groups. The GB shall set the tone for the organizational communication, culture, leadership, and strategic direction. The GB shall also determine how the organization will achieve strategic goals and make critical decisions regarding delivery system design and department and program structure and resourcing. The GB shall evaluate organizational performance on an ongoing basis and set improvement priorities for the organization.
-
-
Procedure
-
Committee Scope and Responsibilities
-
The GB performs the following functions:
-
Strategic Direction
-
At least annually, conduct an organizational assessment to plan and manage change, analyzing the evolving needs of the patient population; organizational strengths and weaknesses; trends in government, the health care industry, and technology; legal and regulatory issues; and potential risks to the organization.
-
Define a clear vision for the organization, incorporating input from staff and stakeholders.
-
Publish the organization’s strategic plan and update at least annually to include the organization’s vision, mission, values, and strategic goals.
-
Ensure communication of the strategic plan to all levels of the organization.
-
Create a responsible, accountable environment of teamwork and continuous improvement that enables the organization to fulfill its mission and meet or exceed goals.
-
Determine the system for communicating important organizational messages through all reporting levels and periodically assess the effectiveness of communication methods.
-
Foster collaboration and coordination within the organization and with stakeholders in the public and private sector.
-
Create partnerships with other federal and state agencies, correctional organizations, non-profit groups, and other stakeholders to create synergies, leverage resources, and improve care.
-
Provide for and comply with the organization’s Code of Conduct.
-
-
Organizational Structure and Delivery System Design
-
Provide a framework to accomplish the goals of the strategic plan including resource allocation and organizational policies.
-
Determine the model of care that will be used to provide services to patients and the services that will be offered including the design of institution missions for cost-effective care.
-
Define and document the organization’s major programs and departments, the scope of services and goals for each major program and department, the organizational reporting structure and lines of authority within the organization, and span of control of key leadership positions.
-
Approve regulations and statewide policies and procedures.
-
Establish a system for conflict resolution between programs or departments.
-
-
Resources
-
Direct sufficient resources to attain the organization’s mission and vision, and meet strategic goals.
-
Plan and manage the organization’s budget including budget projections, approval of Budget Change Concepts and Proposals, Finance Letters and other revisions to the Governor’s Budget, and ongoing oversight and strategic management of spending deficits and surplus.
-
Approve:
-
Staffing formulas and matrices.
-
The statewide plan for staff recruitment, retention, development, and continuing education as well as the model for identifying and assessing civil service and contract staff competencies.
-
Master contracts for statewide services.
-
The statewide plan for facility modifications.
-
Statewide information systems and data infrastructure design.
-
-
Develop and coach leaders at every level of the organization to fulfill the organization’s mission and vision and meet strategic goals.
-
-
Performance Evaluation and Improvement – Health Care System
-
Maintain the quality and safety of patient care and promote performance evaluation and improvement.
-
Approve the design of the quality management and patient safety and risk management systems including the systems used to measure the performance of the organization’s governance, management, clinical, and support functions.
-
Ensure the organization’s participation in legally required public reporting and national quality measurement efforts.
-
Approve improvement priorities for the organization.
-
Assess the organization’s performance on an ongoing basis, intervening as necessary to ensure performance objectives and strategic goals are achieved.
-
Determine mechanisms and provide individual areas of the organization with regular feedback on their progress toward strategic goals.
-
Individual GB members: Personally participate in improvement initiatives.
-
Evaluate the effectiveness of quality management, patient safety, and risk management systems including the performance evaluation system, improvement projects, tools and techniques, and efforts to build improvement capacity through staff development and support modifications to these systems to make them more effective.
-
Review and approve an annual aggregate report of the Root Cause Analyses completed each year including the nature of the system failures they revealed and the actions taken to mitigate risk.
-
-
Evaluation and Improvement – Professional Practice
-
Approve policies governing statewide credentialing, privileging, and peer review processes (routine and non-routine) for licensed medical providers as well as professional ethics policies.
-
Evaluate the effectiveness of the professional practice system in establishing and maintaining a high quality clinical workforce.
-
Oversee and direct as needed the Health Care Executive Committee (HCEC) performance of the following:
-
Acting on all recommendations and Final Proposed Actions, which may include accepting findings and recommendations, accepting findings and determining a different Final Proposed Action, remanding matters for additional investigation or deliberation, and rescinding or terminating peer review and privileging actions.
-
Ensuring that licensed medical providers subject to HCEC review have been provided due process during that review and that HCEC findings and actions are supported by “substantial evidence.”
-
-
-
-
-
Committee Membership
-
The following staff shall serve as standing members of the Health Care Services’ GB:
-
Federal Court Receiver
-
Undersecretary, Health Care Services
-
Director, Health Care Services
-
Director, Health Care Policy and Administration
-
Director, Corrections Services
-
Director, Division of Adult Institutions
-
Director, Legislation, ISUDT, and Communications
-
Director, Information Technology Services Division
-
Chief Counsel, CCHCS Office of Legal Affairs (advisory role only)
-
-
Any changes or additions to the membership shall be approved by the GB.
-
Standing members shall not designate attendance at GB meetings or voting proxy to anyone without the prior approval of the Chairperson, and only where necessary to allow the GB to conduct business. If a quorum is present at a GB meeting, a request to designate an alternate individual to attend a meeting or to vote shall be disapproved. Any approved designee shall not be any lower than at the Director level organizationally.
-
The GB may invite guest participants to meetings at any time that additional insight, expertise, or testimony is needed to support discussion and decision-making.
-
Guests are only permitted with prior authorization from the GB Chairperson [refer to Section (e)(2)(G)].
-
Members of the GB are required to:
-
Understand the scope and purpose of the GB.
-
Review all materials in advance of GB meetings.
-
Present on agenda topics as requested by the Chairperson or other committee members.
-
Provide input in discussions.
-
Vote on motions.
-
Complete action items assigned to their by specific deadlines.
-
Serve as a performance improvement champion, demonstrating visible leadership by directly participating in high-priority improvement initiatives.
-
Notify the Chairperson of planned absences so that the Chairperson may determine whether a quorum of the GB will be present for the meeting at which a member or members may be absent and consider whether it would be appropriate or necessary for the absent member(s) to designate an alternate attendee.
-
-
The Director, Health Care Services, shall serve as the GB Chairperson. The Chairperson retains all the responsibilities of a GB member and also ensures that:
-
The GB meets at least quarterly and as often as necessary to manage the required Committee Scope and Responsibilities as outlined in Section (e)(1).
-
Agenda topics are consistent with the Committee Scope and Responsibilities.
-
Meetings do not proceed unless a quorum is present.
-
Meeting materials are distributed to GB members at least three calendar days in advance of meetings.
-
A formal record of all GB meetings is kept including committee topics, discussion, conclusions and resolutions, and actions.
-
-
-
Committee Voting
-
A quorum shall exist when a simple majority of the voting membership is present. A quorum is necessary to conduct the business of the GB. A member may attend, participate in, and be counted towards a quorum via telephone conference call. In the event of a tie, the Chairperson shall be allotted an additional vote in order to break the stalemate.
-
Each GB voting member shall have one vote on any matter that comes before the GB. Only duly appointed members or approved designees shall vote on GB matters. A motion carries when it receives a simple majority of the vote.
-
All voting members may vote on any matters coming before the GB regardless of the subject matter, program area, or health care discipline at issue.
-
The GB may use electronic voting to address urgent issues that require action prior to the next scheduled meeting or to resolve an existing agenda item.
-
Parliamentary Law and Rules of Order for the 21st Century (Lochrie), most recent edition, shall govern parliamentary procedures.
-
-
Meeting Frequency
-
The GB shall meet at least quarterly and as often as necessary to manage the required Committee Scope and Responsibilities as outlined in Section (e)(1). The Chairperson may schedule special meetings of the GB at their discretion.
-
-
Documentation and Confidentiality Provisions
-
The proceedings and records of the GB shall be confidential and protected from discovery to the extent permitted by law.
-
-
Conflict of Interest
-
Regular voting members of the GB, as CDCR and CCHCS employees, shall comply with applicable laws and regulations regarding disclosure of outside employment, enterprises or activities, and prohibitions against engaging in conflicts of interest. These include the California Code of Regulations, Title 15, Sections 3409 and 3413 as well as pertinent provisions of the Government Code, Public Contracts Code, and the Fair Political Practices Act (FPPA). Among other things, these requirements prohibit CDCR and CCHCS employees from deriving any compensation from any entity doing or seeking to do business with the state.
-
Concurrently with their annual completion and submission of the Statement of Economic Interests (Form 700) pursuant to the FPPA, members of the GB shall provide a copy of their submitted Form 700 to the GB Chairperson to be kept on file for reference in the event that a member’s ability to participate in a GB decision may be impacted by an actual or potential conflict of interest.
-
If any matter of business before the GB represents an actual or potential conflict of interest for any member, they shall disclose the conflict or potential conflict to the GB and recuse themself from participating in any discussion or voting on the matter creating the conflict or potential conflict.
-
Final decisions regarding conflict of interest questions shall be decided by the GB Chairperson. In the event that the GB Chairperson has an actual or perceived conflict of interest, final decisions regarding the conflict of interest shall be decided by the Undersecretary, Health Care Services.
-
-
Closed Session
-
When considering matters from the HCEC, the GB may meet in closed session. The GB may exclude from its meeting any person the GB deems not authorized to participate in discussion regarding credentialing, privileging, or peer review matters. When considering matters from the HCEC, the confidentiality of those matters is protected by Evidence Code Section 1157.
-
-
-
References
-
Federal Health Care Quality Improvement Act of 1986 (42 USCA § 11101)
-
Plata v. Newsom, et al., U.S. District Court of the Northern District of California, Case No. C01-1351 JST
-
Evidence Code, Section 1157
-
California Constitution, Article VII Public Officers and Employees, Section 1-11
-
Business and Professions Code, Section 800, et seq.
-
Joint Commission Leadership Standards
-
Baldrige Award Criteria for Leadership, https://www.nist.gov/baldrige
-
Agency for Healthcare Research and Quality – Patient-Centered Medical Home Resource Center, https://www.ahrq.gov/
-
International Organization for Standardization (ISO) 9001:2015 and ISO/DIS 9004 standards
-
California Department of Corrections and Rehabilitation, Department Operations Manual, Section 33030.3.1, Code of Conduct
-
Health Care Department Operations Manual, Chapter 1, Article 3, Section 1.3.4, Health Care Executive Committee
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.1, Complete Care Model
-
-
Revision History
-
Effective: 12/2017
Revised: 06/16/2023
-
1.1.2 Local Governing Body
-
Policy
-
Where mandated by Title 22 of the California Code of Regulations (i.e., for those licensed facilities with a Correctional Treatment Center) or where required by an accrediting organization such as the Joint Commission, institutions shall have a Local Governing Body (LGB).
-
The LGB shall act at the institutional level for the California Department of Correction and Rehabilitation (CDCR) and California Correctional Health Care Services (CCHCS) Governing Body (GB), ensuring that:
-
Decision-making pertaining to the delivery of health care services at the institutional level are standardized and systematic, incorporates multi-disciplinary input, and is aligned with the CDCR and CCHCS’s strategic vision, mission, and goals for health care and rehabilitation.
-
Resources are allocated in accordance with priorities, goals, and direction of the CDCR and CCHCS and the needs of the incarcerated population.
-
There is a local system for elevating barriers to successful implementation of programs and initiatives that cannot be resolved locally and to share innovative work at the local level that could be beneficial to other institutions.
-
Institution communication methods provide staff the information necessary to provide high quality health care services.
-
The quality management and patients safety systems are evaluated annually and are both efficient and effective, and the institution culture promotes continuous improvement, which includes, but is not limited to, health equity, constant collaboration across program areas, open recognition of quality problems, and patient-centered services.
-
-
-
Purpose
-
Ensure delivery of appropriate, quality health care in a cost-effective manner with minimized risk to patients;
-
Manage and consider the needs and expectations of stakeholders;
-
Respond appropriately to changes in government and the health care industry, including changes in patient demographics and case mix, financial constraints, advances in research and technology, public expectations, and legal and regulatory mandates;
-
Preserve quality of care and economic viability over time; and
-
Adhere to federal and state laws and regulations.
-
-
Responsibility
-
The Chief Executive Officer (CEO) is responsible for planning, implementation, and evaluation of the health care governance structure and processes within the institution.
-
The CEO and Warden are ultimately responsible for ensuring other programs within the institution participate and support the governance structure to operate effectively and efficiently.
-
CDCR and CCHCS departmental leadership at all levels of the organization, within the scope of their authority, are responsible to support and participate fully in the governance structure and processes and to ensure their staff are aware of and adhere to governance processes and procedures.
-
-
Procedure Overview
-
The LGB is the highest-level committee in the network of institution committees that makes up the local health care governance structure.
-
The LGB is ultimately accountable for quality patient care, treatment, and services provided by employees and contractors at the institution. Among other tasks, the LGB defines the strategic direction of health care services within the institution, implements changes to the organizational structure and health care delivery system design, and ensures that the institution has sufficient resources to accomplish strategic goals, including prepared and competent staff. The LGB ensures that there are adequate systems in place to continuously evaluate and improve performance, ensuring accountability, particularly among key leaders and licensed clinicians. The LGB shall set the tone for the institution related to health care communication, culture, leadership, and strategic direction.
-
The membership of the LGB is comprised of the highest-level health care executives as well as the leaders from other major program areas within the institution.
-
-
Procedure
-
Committee Scope and Responsibilities
-
The LGB performs the following functions:
-
Strategic Direction
-
Oversees implementation of health care-related programs and initiatives, in alignment with the CDCR and CCHCS’s mission, vision, and goals for the delivery of health care services.
-
Establishes an effective system for developing and vetting local operating procedures to ensure appropriate multi-disciplinary participation, high-quality products, and acceptance among staff implementing the procedures.
-
Appointment of a health care administrator whose qualifications, authority and duties shall be defined in a written statement adopted by the LGB.
-
Appointment of a Medical Director, or at institutions with Mental Health Crisis Bed units or Psychiatric Inpatient Programs if the Chief Psychiatrist (CP) is not the Medical Director, delegation of clinical responsibilities of the Medical Director to the CP.
-
Approves local operating procedures consistent with statewide policies to enable access to and continuity of high quality patient care and as required by Title 22 regulations at least annually.
-
Establishes an effective system for elevating critical program concerns to higher levels of the organization.
-
Establishes a system for conflict resolution between programs and departments.
-
-
Resource Allocation
-
Directs sufficient resources to attain the organization’s mission and vision and meet strategic goals.
-
Plans and manages the organization’s budget including budget projections and provides ongoing oversight and strategic management of spending deficits and surplus.
-
Implements:
-
Staffing formulas and matrices.
-
Notification of appointment and reappointment of health care professionals to the health care staff.
-
Staff recruitment, retention, development, and continuing education as well as the model for identifying and assessing civil service and contract staff competencies, per the statewide plan.
-
Processes to ensure the confidentiality of written actions taken on health care staff.
-
Contracts for health care services, per statewide master contracts and, as appropriate, locally-generated contracts.
-
Facility modifications per the statewide plan and repairs as required through local resources.
-
Information systems and data infrastructure design per statewide direction.
-
-
Develops and coaches leaders at every level of the institution to fulfill the organization’s mission and vision and meet strategic goals.
-
-
Communication
-
Communicates the organization’s vision, strategic goals, and associated timeframes to staff, the patient population, and other key stakeholders.
-
Determines the system for communicating important organizational messages through all reporting levels quickly and efficiently and periodically assesses the effectiveness of communication methods.
-
Creates partnerships with other federal and state agencies, correctional organizations, non-profit groups, and other stakeholders to create synergies, leverage resources, and improve care.
-
-
Performance Evaluation and Improvement – Health Care System and Patient Safety Culture
-
Creates a responsible, accountable environment of collaboration and continuous improvement that enables the organization to fulfill its mission and meet or exceed its goals.
-
Fosters cooperation and coordination within the organization and with stakeholders in the public and private sector.
-
Provides for and complies with the organization’s Code of Conduct.
-
Maintains the quality and safety of patient care and promotes performance evaluation and improvement.
-
Oversees implementation of the quality management, patient safety and risk management systems including the systems used to measure the performance of the organization’s governance, management, clinical, and support functions.
-
Ensures the organization’s participation in legally required public reporting and the organization’s quality measurement efforts.
-
Approves improvement priorities for the organization.
-
Determines mechanisms for and provides individual areas of the organization with regular feedback on progress toward strategic goals.
-
Facilitates personal participation of individual LGB members in improvement initiatives.
-
Evaluates the effectiveness of quality management, patient safety, and risk management systems at least annually, including the performance evaluation system, improvement projects, tools and techniques, and efforts to build improvement capacity through staff development while supporting modifications to these systems to make them more effective.
-
-
Performance Evaluation and Improvement – Professional Practice
-
Implements local credentialing and privileging procedures in accordance with statewide credentialing and privileging policies, peer review processes (routine and non-routine) for licensed medical providers as well as professional ethics policies.
-
Assigns to the medical staff, or when appropriate, to the CEO or designee reasonable authority for implementing mechanisms to improve the quality and appropriateness of patient care, and may also assign to the medical staff or chiefs of the relevant clinical disciplines responsibility for the clinical performance of individuals with delineated privileges, and supports these activities and mechanisms.
-
Evaluates the effectiveness of the professional practice system in establishing and maintaining a high quality clinical workforce.
-
-
Accountability
-
Assures that health care staff rules and regulations are subject to LGB approval, which approval shall not be unreasonably withheld; and that health care staff are permitted to appeal decisions they feel are unfair or wrong.
-
-
-
Committee Membership
-
The following staff shall serve as voting members of the LGB:
-
CEO (Chairperson)
-
Warden or designee (Associate Warden for Health Care or Chief Deputy Warden)
-
Associate Warden, Business Services
-
Associate Warden, Central Services
-
Associate Warden, Health Care
-
Chief Support Executive
-
Chief of Mental Health
-
CP, or a designated Psychiatrist (in absence of CP)
-
Health Program Manager III, Dental Services
-
Supervising Dentist
-
Chief Medical Executive (CME)
-
Chief Nurse Executive
-
Health Program Manager III, Quality Management
-
-
The term of the appointment for LGB members shall be the duration of their appointment to their respective positions.
-
Standing members shall not designate attendance at LGB meetings or voting proxy to anyone without the prior approval of the Chairperson, and only when necessary to allow the GB to conduct business.
-
The LGB may invite guest participants to meetings at any time that additional insight, expertise, or testimony is needed to support discussion and decision-making.
-
Guests are only permitted with prior authorization from the LGB Chairperson.
-
Members of the LGB are required to:
-
Understand the scope and purpose of the LGB.
-
Review all meeting materials in advance of LGB meetings.
-
Present on agenda topics as requested by the Chairperson or other committee members.
-
Provide input in discussions.
-
Vote on motions.
-
Complete action items assigned by specific deadlines.
-
Serve as a performance improvement champion, demonstrating visible leadership by directly participating in high-priority improvement initiatives.
-
Notify the Chairperson of planned absences so that the Chairperson may determine whether a quorum of the LGB shall be present for the meeting at which a member or members may be absent and consider whether it would be appropriate or necessary for the absent member(s) to designate an alternate attendee.
-
-
The CEO shall serve as the LGB Chairperson. The Chairperson retains all the responsibilities of a LGB member and also ensures that:
-
The LGB meets at least quarterly and as often as necessary to manage the required Committee Scope and Responsibilities as outlined in Section (e)(1).
-
Agenda topics are consistent Section (e)(1).
-
Meetings do not proceed unless a quorum is present.
-
Meeting materials are distributed to LGB members at least three calendar days in advance of meetings.
-
A formal record of all LGB meetings is kept including committee topics, discussion, conclusions and resolutions, and actions.
-
-
-
Committee Voting
-
A quorum shall exist when a simple majority of the voting membership is present. A quorum is necessary to conduct the business of the LGB. A member may attend, participate in, and be counted towards a quorum via telephone conference call. In the event of a tie, the Chairperson may vote in order to break the stalemate.
-
Each voting member shall have one vote on any matter that comes before the LGB. Only duly appointed members or approved designees shall vote on LGB matters. A motion carries when it receives a simple majority of the vote.
-
All voting members may vote on any matters coming before the LGB regardless of the subject matter, program area, or health care discipline at issue.
-
The LGB may use electronic voting to address urgent issues that require action prior to the next scheduled meeting or to resolve an existing agenda item.
-
Parliamentary Law and Rules of Order for the 21st Century (Lochrie), most recent edition, will govern parliamentary procedures.
-
-
Meeting Frequency
-
The LGB shall meet at least quarterly and as often as necessary to manage the required Committee Scope and Responsibilities. The Chairperson may schedule special meetings of the LGB at their discretion.
-
-
Documentation and Confidentiality Provisions
-
The proceedings and records of the LGB shall be confidential and protected from discovery to the extent permitted by law.
-
-
Conflict of Interest
-
Regular voting members of the LGB, as CDCR and CCHCS employees, shall comply with applicable laws, regulations, and departmental policies regarding disclosure of outside employment, enterprises or activities, and prohibitions against engaging in conflicts of interest. These include the California Code of Regulations, Title 15, Sections 3409 and 3413 as well as pertinent provisions of the Government Code, Public Contracts Code, and the Fair Political Practices Act (FPPA). These requirements prohibit CDCR and CCHCS employees from deriving any compensation and gifts from any entity doing or seeking to do business with the state.
-
Concurrently with their annual completion and submission of the Statement of Economic Interests (Form 700) pursuant to the FPPA, members of the LGB shall provide a copy of their submitted Form 700 to the LGB Chairperson to be kept on file for reference in the event that a member’s ability to participate in a LGB decision may be impacted by an actual or potential conflict of interest.
-
If any matter of business before the LGB represents an actual or potential conflict of interest for any member, they shall disclose the conflict or potential conflict to the LGB and recuse themselves from participating in any discussion or voting on the matter creating the conflict or potential conflict.
-
Final decisions regarding conflict of interest questions shall be decided by the LGB Chairperson. In the event that the LGB Chairperson has an actual or perceived conflict of interest, final decisions regarding the conflict of interest shall be decided by the Regional Health Care Executive overseeing the institution.
-
-
Closed Session
-
When considering professional practice matters, the LGB may meet in closed session. The LGB may exclude from its meeting any person the LGB deems not authorized to participate in discussion regarding credentialing, privileging, or peer review matters. When considering professional practice matters, the confidentiality of those matters is protected by Evidence Code Section 1157.
-
-
Delegated Authority
-
The LGB shall implement requirements set forth by the statewide GB at a local level.
-
During the absence of the CME or CP, the Medical Officer of the Day, the Physician on Call, or Psychiatrist On-Call shall act with authority in clinical matters, and, when indicated, the administrator shall act with authority in administrative matters.
-
-
-
References
-
Federal Health Care Quality Improvement Act of 1986 (42 USCA § 11101)
-
Plata v. Newsom, et al., U.S. District Court of the Northern District of California, Case No. C01-1351 JST
-
Evidence Code, Section 1157
-
California Constitution, Article VII Public Officers and Employees, Section 1-11
-
Business and Professions Code, Section 800, et seq.
-
Joint Commission Leadership Standards
-
Baldrige Award Criteria for Leadership, https://www.nist.gov/baldrige
-
Agency for Healthcare Research and Quality – Patient-Centered Medical Home Resource Center, https://pcmh.ahrq.gov/
-
International Organization for Standardization (ISO) 9001:2015 and ISO/DIS 9004 standards
-
California Department of Corrections and Rehabilitation, Department Operations Manual, Section 33030.3.1, Code of Conduct
-
Health Care Department Operations Manual, Chapter 1, Article 3, Section 1.3.4, Health Care Executive Committee
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.1, Complete Care Model
-
-
Revision History
-
Effective: 01/2002
Revised: 06/16/2023
-