Article 2 – Health Care Program Governance
1.2.1 Complete Care Oversight Team Committee
-
Procedure Overview
-
California Correctional Health Care Services (CCHCS) shall maintain a Complete Care Oversight Team (CCOT) committee to oversee statewide implementation of the Complete Care Model (CCM) for delivery of patient health care services. This committee is responsible for leading, coordinating, and communicating health care initiatives including the appropriate design and deployment of technical tools and systems (e.g., the Electronic Health Record System) to support safe, timely, and cost-effective patient care.
-
-
Procedure
-
The CCOT ensures full implementation of the CCM at all California Department of Corrections and Rehabilitation adult institutions by performing the following functions:
-
Committee Functions
-
Strategic Direction
-
Ensure the organization’s approach to health care services delivery applies the foundational principles and requirements set forth in the Health Care Department Operations Manual, Section 3.1.1, Complete Care Model.
-
Establish standards for the CCM and add new or modify standards based on new information or technology, best practices, or changes in evidence-based practices.
-
Plan and prioritize CCM implementation and improvement strategies in alignment with organizational goals and priorities.
-
Provide input to system-wide performance improvement objectives in the CCHCS Performance Improvement Plan.
-
-
Evaluation and Improvement
-
Review the Health Care Services Dashboard and other data and information to monitor delivery system performance.
-
Develop performance metrics and assessment tools.
-
Design and implement statewide initiatives, collaboratives, and other improvement activities to fully implement the CCM.
-
Use community literature, industry standards, and evidence-based strategies to standardize and integrate health care delivery processes.
-
Identify and redesign processes that pose safety risks to patients and staff or impede full implementation of the CCM.
-
Develop change packages promoting the use of improvement tools and techniques and decision support such as workflows, forms, user guides, implementation plans, checklists and more.
-
Identify and disseminate CCM best practices.
-
Evaluate resources supporting CCM implementation including, but not limited to:
-
Staffing
-
Equipment and supplies
-
Information technology
-
Physical plant
-
Policies, laws, and regulations
-
-
Recommend changes to relevant programs and stakeholders to resolve barriers to successful implementation.
-
-
Communication and Coordination
-
Ensure there is a mechanism for communicating the progress of CCM implementation and system performance to all levels of the organization.
-
Use existing forums at institutions, regional offices, and headquarters to share lessons learned including best practices.
-
Partner with committees, workgroups, and program areas at all levels of the organization to redesign health care processes and facilitate CCM implementation and improvements.
-
Refer CCM issues and concerns to other policy-making bodies and relevant committees as appropriate to support policy changes and inform resource management decisions.
-
Report progress of the CCM implementation and delivery system performance to the CCHCS Quality Management Committee at least quarterly.
-
-
Orientation and Training
-
Develop and implement training and staff development programs to help health care staff become oriented to and maintain up-to-date knowledge of the CCM principles and associated systems and processes.
-
Establish a structure for ongoing technical assistance by health care staff at headquarters and regional levels.
-
Provide regular feedback to institutions on their progress towards successful implementation of the CCM.
-
Promote a high performance culture of teamwork and continuous learning, improvement, and innovation.
-
-
-
Membership and Meetings
-
Chairperson(s): One or more CCOT members shall be selected to serve as chairperson(s) from the current CCOT membership for a period of at least 12 months. The chairperson(s) responsibilities are to ensure the following:
-
CCOT meetings occur regularly;
-
Meeting agendas reflect the responsibilities and actions described in this procedure;
-
Decisions are documented and communicated to relevant stakeholders as appropriate including agenda topics, discussion, conclusions/resolutions, and actions;
-
New CCOT members receive orientation to their new role and responsibilities; and
-
Each voting member chooses a designee to serve in their stead when necessary.
-
-
Voting Members: Regional Health Care Executives shall serve as voting members of the CCOT as well as Deputy Directors from the following CCHCS programs:
-
Business Services
-
Corrections Services
-
Dental Program
-
Information Technology Services
-
CCHCS Office of Legal Affairs
-
Medical Services
-
Mental Health Program
-
Nursing Services
-
Pharmacy Services
-
Policy and Risk Management Services
-
Quality Management Program
-
Resource Management
-
-
Non-voting Members, Designees, and Other Participants: Non-voting members, such as subject matter experts, presenters, and guests may attend as appropriate and as approved by the CCOT Chairperson.
-
Voting and Quorum: Each member has one vote, and a quorum shall exist when a simple majority of the voting membership is present.
-
Meeting Frequency: The CCOT shall meet as often as necessary to cover the responsibilities and actions described in this procedure, but no less frequently than monthly.
-
CCOT Subcommittees and Workgroups: The CCOT shall establish standing subcommittees and ad hoc workgroups to plan and develop or modify existing clinical program policies and processes; opine on resource requirements; and to coordinate and oversee changes or new designs in decision support and documentation within the Electronic Health Records System and reports used to manage patient populations, clinical operations, and organizational performance.
-
-
-
References
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.3, Quality Management Overview
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.4, Quality Management Program, Statewide Governance
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.5, Quality Management Program, Institution
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.1, Complete Care Model
-
California Correctional Health Care Services, Performance Improvement Plan, Quality Management – Performance-Improvement-Plan-2023-2025.pdf – All Documents (sharepoint.com)
-
Agency for Healthcare Research and Quality – Patient Centered Medical Home Resource Center, https://www.ahrq.gov/
-
The Joint Commission Primary Care Medical Home Certification, http://www.jointcommission.org/accreditation/pchi.aspx
-
National Committee for Quality Assurance – Patient-Centered Medical Home Recognition, http://www.ncqa.org/Programs/Recognition/Practices/PatientCenteredMedicalHomePCMH.aspx
-
Commonwealth Fund – Safety Net Medical Home Initiative, http://www.commonwealthfund.org/interactives-and-data/multimedia/videos/2011/safety-net-medical-home-initiative
-
-
Revision History
-
Effective: 07/2017
-
1.2.2 Clinical Documentation and Decision Support Committee
-
Policy
-
The California Correctional Health Care Services (CCHCS) shall maintain a Clinical Documentation and Decision Support (CDADS) Committee to ensure clinical documentation and decision-making support tools are evidence-based and promote best practices within CCHCS. The CDADS committee is responsible for the following:
-
Overseeing the research and development or the adaptation of existing evidence-based standards of care, including but not limited to clinical guidelines and pathways, procedures, and protocols in order to promote evidence-based practices for patients under care of the California Department of Corrections and Rehabilitation (CDCR).
-
Facilitating the dissemination of updated clinical guidance through the CCHCS Health Care Regulations and Policy Section (RPS).
-
Coordinating with other relevant committees, as indicated.
-
Consulting on documentation and clinical decision-making tools within the electronic health record system (EHRS) to promote best practices and ensure integration of standards of care into the EHRS.
-
Regularly monitoring and evaluating implementation of standards of care.
-
-
Responsibility
-
Statewide
-
The Deputy Director (DD), Medical Services, and the Deputy Medical Executive (DME), Policy and Provider Workforce, are responsible for the statewide planning, implementation, and evaluation of the CDADS policy and procedure.
-
-
Regional
-
Regional leadership is responsible for reviewing and providing feedback for documentation and decision support, as requested by the CDADS Committee, within the designated timeframes as well as facilitating dissemination of the guidelines to the appropriate staff.
-
-
Institution
-
Health care leadership is responsible for the following:
-
Reviewing and providing feedback for documentation and decision support, as requested by the CDADS Committee, within the designated timeframes.
-
Facilitating dissemination of guidelines to appropriate staff and implementation of evidence-based practice to the appropriate staff.
-
Ongoing monitoring to ensure sustainable incorporation into their practice utilizing designated patient care tools.
-
-
-
Procedure
-
Clinical Documentation and Decision Support Committee Membership and Meetings
-
The chairperson shall be a DME or Assistant DME appointed by the DD, Medical Services. The chairperson shall assist the DD, Medical Services in the selection of other members of the CDADS Committee when multiple candidates may be available.
-
The Continuing Health Care Education Planning Committee Chairperson shall serve as chairperson’s designee if the appointed chairperson of the CDADS Committee is unavailable to attend a committee meeting.
-
The chairperson and members serve on the committee for two-year, terms and may serve multiple terms with approval by the chairperson and DD, Medical Services. To ensure program continuity, terms shall be staggered so that no more than 50 percent of voting members will change in any given year.
-
Voting members (and alternate designees) shall be appointed by the DD, Medical Services from among CCHCS and CDCR staff nominated by clinical managers or supervisors for each discipline:
-
Two Medical Services physician managers or designees
-
Two headquarters Quality Management physician managers or designees
-
Statewide Chief Nurse Executive or designee
-
Two Chief or Senior Psychiatrists (headquarters or institution)
-
One Senior or Chief Psychologist (headquarters or institution)
-
One Chief Dentist (headquarters) or Supervising Dentist
-
One Physician & Surgeon (headquarters or Institution)
-
Statewide Chief, Pharmacy Services or designee
-
Two Statewide or Regional Pharmacy Services Managers
-
Four Institutional or Regional physician managers or designees
-
One Supervising Psychiatric Social Worker from the Statewide Mental Health Program (headquarters or institution)
-
One Advanced Practice Provider (headquarters or institution)
-
Chair, Continuing Health Care Education Planning Committee
-
-
CDADS Committee members may choose a designee to serve in their stead. Designees shall be approved by the chairperson.
-
Non-voting members of the CDADS Committee, non-members, and guests shall be approved by the chairperson.
-
Meetings
-
The CDADS Committee shall meet at least monthly unless there are no agenda items for discussion.
-
Each voting member shall have one vote. A quorum shall exist when at least nine voting members are present.
-
The chairperson may serve as a voting member if needed to meet quorum.
-
Committee actions shall be approved by a majority vote.
-
Meetings via teleconference shall be made available to members of the CDADS Committee. Meetings via teleconference shall be conducted in the same manner as an in-person meeting.
-
The CDADS Committee may use electronic voting to address issues when it is determined that waiting until the next scheduled meeting is suboptimal. Electronic voting may be used to resolve an existing agenda item or to address an urgent or emergent new agenda item. Issues addressed via electronic voting shall be approved by a majority vote. Electronic voting communications shall be maintained in digital format.
-
-
Conflict of Interest
-
A CDADS Committee voting member shall not participate in any decision if they have a personal conflict of interest.
-
A personal conflict is defined as a professional, financial, or other obligation or interest that is likely to limit the member’s ability to participate impartially in decision-making.
-
A member shall disclose to the chairperson any potential or actual conflicts of interest prior to the discussion of any agenda item involving the subject of the conflict or actual conflict.
-
The chairperson shall determine whether a conflict exists and is material, and if it is, bring it to the attention of the committee.
-
The committee shall then determine whether the conflict exists and is material, and in the presence of an existing material conflict, exclude the member with the conflict from participation in discussion or decision-making on the agenda item. Alternatively, the member may recuse themselves from the discussion of and decision-making on the agenda item.
-
-
If the committee has reasonable cause to believe a member has failed to disclose an actual or potential conflict of interest, they shall inform the member of the basis for such belief and afford the member an opportunity to explain the alleged failure to disclose.
-
If, after hearing the member’s response and after making any further inquiries as warranted by the circumstances, the committee shall determine whether the member may participate in any discussion or decision-making and whether any further action might be warranted, such as removal from the committee, or referral to the member hiring authority for further action.
-
-
-
-
CDADS Scope of Review
-
The CDADS Committee is authorized to take the following actions:
-
Oversee, adopt, review, or make clinical recommendations to guidelines, care guides, and patient education materials to meet the needs of patients, making appropriate adaptations in consideration of safety and security, and other issues which apply in the correctional environment.
-
Review literature, as needed, including published guidelines, to ensure documentation for review is consistent with community standards.
-
Review and make recommendations on additional health care guidance from other disciplines, when requested, including treatment protocols, procedures, related education or training programs, and accompanying patient education materials specific to patient care issues.
-
Use experience of medical experts to inform recommendations when medical literature or research is unavailable in certain areas of care.
-
-
Review Process
-
All clinical decision support material to be reviewed by the CDADS Committee shall be submitted to the committee support staff via email at CCHCS-CDADS@cdcr.ca.gov.
-
The committee shall review and approve clinical decision support materials detailed above in consultation with appropriate subject matter experts and CCHCS programs.
-
If not approved, the committee shall provide reasons for denial or deferral to the submitting program with recommended changes for all materials that are reviewed and not approved.
-
Upon resubmission of edited materials, the committee may either place the edited material on a subsequent committee agenda or provide approval via electronic voting.
-
Upon approval by the committee, any materials containing medication or pharmaceutical recommendations shall be forwarded to the Pharmacy and Therapeutics (P&T) Committee for review and other relevant committees, if indicated. The P&T Committee shall review the materials and recommend:
-
Approval;
-
Approval with revisions; or
-
Disapproval with revisions.
-
-
Any recommendations from other committees shall be communicated to the CDADS support staff who shall forward them to the submitting program. Edited material shall be returned to the CDADS email address above and will follow the process in Section (c)(3)(D).
-
Upon completion of review by the P&T Committee or other relevant committees and CDADS Committee approval, all materials will follow the established workflow developed by RPS for dissemination of updated clinical guidance.
-
-
-
References
-
California Code of Regulations, Title 15, Division 3, Chapter 1, Subchapter 5, Article 2, Sections 3409 and 3413
-
California Fair Political Practices Commission, www.fppc.ca.gov
-
Agency for Healthcare Research and Quality, www.ahrq.gov
-
National Commission on Correctional Health Care Standard P-G-01, Chronic Disease Services, https://www.ncchc.org/chronic-disease-services-spotlight/
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.11, CCHCS Systemwide Pharmacy and Therapeutics Committee
-
-
Revision History
-
Effective: 12/2003
Revised: 01/20/2026
-
1.2.3 Quality Management Program Overview
-
Background
-
California Correctional Health Care Services (CCHCS) delivers a continuum of health care services to patients by different professional disciplines across multiple levels of care in both outpatient and inpatient settings. In order to improve care and avoid unnecessary morbidity, mortality and costs, health care and correctional staff need to implement sustainable strategies that:
-
Improve processes and systems to ensure high reliability and sustainability.
-
Strengthen continuity and coordination of care.
-
Strengthen communication and collaboration among all professional disciplines and with the patient.
-
-
An overarching strategy that CCHCS has endorsed to improve patient care and system performance is the establishment of a primary care management model, which is a collaborative care model that is different than the traditional medical model that organizes services around acute and episodic events. Rather, the primary care model provides continuous services to patients with chronic and complex physical and behavioral health conditions who disproportionately drive risk and resources.
-
The Primary Care Model is based on a number of nationally recognized models and paradigms that emphasize:
-
Risk stratification of individual patients and patient populations using a classification system that determines each patient’s health care risk and places the patient at a facility best equipped to manage his or her health care needs in a cost effective manner.
-
A Comprehensive patient focus.
-
Interdisciplinary and consistent team-based care.
-
Evidence-based practices.
-
Active patient involvement and self-management.
-
Decision support and information systems to assist in managing individual patients and patient populations, and facilitating continuous improvements in patient outcomes, clinical practice and processes of care.
-
-
-
Policy
-
Consistent with private and public health care industry standards, CCHCS maintains a Quality Management (QM) Program to continuously evaluate and improve the performance of the health care system. The design of the QM Program draws from nationally recognized quality improvement organizations, principles, concepts, and models.
-
To support health care staff in consistently delivering timely, effective, efficient, and safe care, the QM Program:
-
Supports the identification of performance improvement priorities, and requires development of performance improvement plans and objectives at statewide and institution levels.
-
Provides structures, processes, tools and techniques to analyze and redesign health care processes and manage change.
-
Assists institutions in establishing, improving, and sustaining an integrated health care delivery system.
-
Maintains a performance evaluation program, including an organization Health Care Services Dashboard (Dashboard), and ongoing business intelligence to assess critical health care processes and outcomes, identifying potential areas for improvement, and evaluating performance at all levels of the organization.
-
Requires and provides Patient Registries, evidence-based guidelines, training, and deployment of improvement models and tools such as Focus-Plan-Do-Study-Act (Focus-PDSA) as elements of the performance improvement program.
-
Promotes a culture of teamwork and continuous learning and innovation.
-
-
The scope of the QM Program includes clinical operations, clinical practice, and program administration across all health care delivery settings. The specific priorities of the QM Program change but always strategically focus on key performance areas that most impact the access, quality, safety, utilization, cost, and value concerns.
-
-
Purpose
-
To support continuous health care system evaluation and positive change in order to:
-
Improve patient outcomes.
-
Improve health care quality, cost effectiveness, and patient and staff safety.
-
Improve cost-efficiencies and value-added work, and reduce waste.
-
Comply with legal and regulatory requirements.
-
-
Responsibility
-
Chief Executive Officers (CEOs) oversee implementation of the QM Program at the local level, and participate in statewide strategic planning and implementation and evaluation of statewide initiatives. Institutions may have a local Chief Quality Officer who provides subject matter expertise and supports implementation of the QM Program locally.
-
The Receiver, CCHCS, and Director, Division of Correctional Health Care Services delegate responsibility for statewide planning, implementation, and evaluation of the QM Program to the statewide Chief Quality Officer.
-
-
Quality Management Program
-
Identifying Improvement Priorities and Strategic Alignment
-
Overview
-
Improvement efforts are most effective when all levels of an organization are informed of improvement priorities and rally around a core set of improvement goals. To this end, the QM Program facilitates the setting of improvement priorities at the statewide level and provides support to institutions as they identify institution-specific priorities, communicate priorities to health care staff, and ensure that program planning and day-to-day operations align with improvement priorities.
-
-
Priority-Setting Process
-
Each year, CCHCS reviews health care areas considered to be high risk, high volume, high cost, and problem-prone and identifies organization-wide improvement priorities. CCHCS incorporates the highest-priority program areas or topics into a performance improvement plan, with one or more performance objectives established for all priority improvement areas. The development and pre-implementation vetting process involves health care leadership as well as external stakeholders. This same priority-setting process occurs at each institution:
-
Taking into consideration statewide strategic priorities and the Primary Care Model, institutions identify institution-specific improvement priorities and performance objectives, customizing an institution improvement plan for the facility’s health care mission, resources, and the needs of the patient population which is vetted through statewide leadership via the Quality Management Committee (QMC) and other forums.
-
The QM Program promotes the establishment of clear and measurable performance objectives for all improvement initiatives and sustainability planning to ensure that successful improvement strategies and positive results can be reproduced consistently in the future.
-
Upon developing an annual improvement plan, institution leadership is responsible for communicating improvement priorities to staff at all levels of the health care system and helping staff understand their role in achieving improvement objectives.
-
Institution leadership also guides the process of strategic alignment, by which managers and supervisors determine how program operations and day-to-day supervision will support performance objectives, and how care teams and other staff incorporate improvement activities for priority areas into day-to-day work.
-
-
-
Performance Evaluation
-
Overview
-
To assess the impact of change management efforts and progress toward performance objectives, CCHCS uses an objective and systematic process to measure and evaluate health care system performance. Taking into consideration the organizational mission and patient population risks and needs, CCHCS determines the clinical and administrative processes that are most important to measure, and chooses structural, process, and outcome measures critical for ongoing monitoring of the health care system and identification of quality improvement opportunities.
-
In determining performance measures, CCHCS considers the science or evidence supporting the selected measures, and comparable measures used broadly in the health care industry, including those employed by the Healthcare Effectiveness and Information Set and the Agency for Healthcare Research and Quality. Among other factors, the CCHCS takes into account the performance of other health care organizations and past organizational performance during the establishment of performance objectives.
-
-
Performance Evaluation
-
CCHCS consolidates key performance measures essential to fulfilling the organization’s mission in the Dashboard, a monthly organization-wide report widely distributed to health care staff. All performance improvement plan objectives are benchmarked and monitored through the Dashboard. The Dashboard provides performance data at statewide and institution levels, and trends performance over time.
-
In addition to the monthly Dashboard, CCHCS uses other methods to evaluate performance and inform decisions about improvement priorities, including, but not limited to:
-
Periodic performance reports and special studies, including morbidity and mortality analysis.
-
A health incident reporting system and a defined process for the tracking, analysis, and reporting of sentinel events.
-
Standardized audit tools to evaluate the quality of care and assess adherence to practice guidelines, policies and procedures, and federal and state laws.
-
Patient Registries or “exception reports” that identify individual patients who have not received services per treatment guidelines or who have abnormal findings.
-
-
CCHCS ensures that there are appropriate processes in place to validate performance data, particularly when:
-
A new measure is introduced or an existing measure is substantially changed.
-
Data will be made public on the internet or through other mechanisms.
-
There are unexpected changes in the data reported for an existing measure.
-
Data sources change (e.g., paper-based records become electronic).
-
-
-
Performance Data Validation
-
Validation processes may include, but are not limited to, use of statistically-valid samples, redundant data collection by independent parties, and corrective actions to address data discrepancies.
-
-
-
Performance Improvement Models, Tools, and Training
-
Overview
-
Consistent with health care industry standards, the QM Program uses the science of process improvement and change management, including techniques and tools, to assist health care staff in establishing and maintaining an integrated health care delivery system and achieving objectives outlined in improvement plans. The CCHCS QM Program promotes:
-
Establishing clear and measurable objectives for all improvement initiatives.
-
Using decision support such as care guides, chart review tools, forms, and checklists, and staff development programs, such as continuing education presentations, workshops, and on-the-job training to remind care teams of evidence-based guidelines and reinforce new health care processes.
-
Using information systems, such as the Dashboard, Institution Scorecard, Patient Registries, provider profiles, performance reports, and tracking systems to support proactive administrative and clinical management.
-
Re-evaluating critical health care processes on a regular and ad hoc basis through mechanisms such as Root Cause Analysis and Failure Mode and Effects Analysis, and redesigning processes and systems to improve outcomes.
-
Engaging patients as active and responsible participants in the health care process through patient education and self-management programs, and partnership with peer educators and incarcerated person groups such as Men’s/Women’s Advisory Council.
-
Formally documenting new processes, such as in a local operating procedure, ongoing use of decision support, orientation for new staff, and regular training updates for continuing staff to promote lasting and sustainable change.
-
-
Improvement Models
-
CCHCS uses the Cycle of Change Model as a framework for design and implementation of improvement initiatives at both the state and institution levels. The Cycle of Change Model is based on a number of Performance/QM models and the Chronic Care Model that support behavioral change and sustainable business processes that promote the Primary Care Model. Under the Cycle of Change, institutions:
-
Identify improvement priorities,
-
Set performance objectives,
-
Clarify performance expectations and create decision support to help staff meet performance expectations,
-
Provide training and staff development,
-
Provide targeted technical assistance from managers, supervisors, designated mentors or “champions” or other staff in areas where performance continues to lag, and
-
Regularly re-assess performance to determine progress toward performance objectives.
-
-
Within the health care and quality improvement industries, there are other established methods which include statistical analysis, Root Cause Analysis, the Model for Improvement, Lean Model, Six Sigma, Focus-PDSA, Failure Mode and Effects Analysis, Process Flow Diagramming, and Cause and Effect Diagramming for analysis of performance problems, development of solutions, and testing and evaluating interventions.
-
These improvement models provide a framework for redesigning health care processes and implementing improvement initiatives at the point of care, such as incorporating decision support and information systems into day-to-day practice.
-
-
Training on Quality Improvement Techniques
-
CCHCS, through the QM Program, is responsible for educating health care staff about improvement models, helping staff to develop the skills to use these models, and developing toolkits that support health care staff step-by-step through the application of an improvement model or concept.
-
-
-
Technical Assistance from Headquarters and Regional Staff
-
CCHCS staff based at headquarters and at regional levels support institutions as they redesign health care processes and establish well-functioning QM Programs.
-
Among other activities, headquarters-based staff and regional teams from various disciplines and program areas:
-
Assist institutions in identifying improvement priorities and developing improvement plans.
-
Review program performance data, evaluate the effectiveness of quality improvement interventions, assess the performance of the integrated health care services delivery system, and determine progress toward performance objectives.
-
Perform quality of care reviews and assist institutions in planning and implementing activities to improve the quality of clinical practice.
-
Support the institution in taking action when quality or safety problems are identified including, but not limited to, sharing best practices and participating in improvement teams.
-
Assist institutions in preparations for licensing surveys, audits by external stakeholders, and inspections, and as they develop and manage implementation of improvement plans.
-
Promote adherence to policies, evidence-based guidelines, and standards of care through development of decision support tools and staff development programs, among other strategies.
-
Ensure dissemination of relevant, accurate, and timely performance information to institution management and staff.
-
Review QM governance activities and recommend program changes or further activity.
-
Identify, disseminate, and adapt best practices.
-
Support local staff development programs to orient all health care staff to the QM Program, promote the skills necessary to perform quality improvement work at institutions, and empower staff at all reporting levels to participate in quality improvement.
-
-
-
-
Quality Management Program Governance
-
Overview
-
The QM Program includes a management structure composed of inter-disciplinary committees and teams at statewide and local levels to support organizational communication, coordination, control and change. The organization’s leadership uses the QM Program management structure to:
-
Plan, implement, evaluate and improve the health care system’s performance.
-
Oversee quality improvement and patient safety initiatives.
-
Coordinate improvement activities across programs and disciplines.
-
Communicate strategic direction and measurement information.
-
Ensure orientation and training of staff as it relates to performance and quality improvement.
-
Promote a culture of continuous learning and innovation.
-
-
The committee network serves as a supplement to the traditional reporting structure at headquarters and at individual institutions, providing regular interdisciplinary forums for managers, supervisors, and line staff to manage improvement activities.
-
-
Quality Management Committees
-
The statewide QMC meets at least quarterly and the institution QMC meets at least monthly to determine improvement priorities and strategies, regularly review performance data and take action to improve performance, coordinate the activities of committees and teams that focus on process and performance improvement, and ensure that staff are trained in quality improvement concepts, tools, and techniques.
-
Quality Management Committees ensure that staff at the headquarters and local level receives the orientation and training necessary to participate in improvement activities and apply improvement skills, such as problem analysis and system redesign, in their day-to-day work.
-
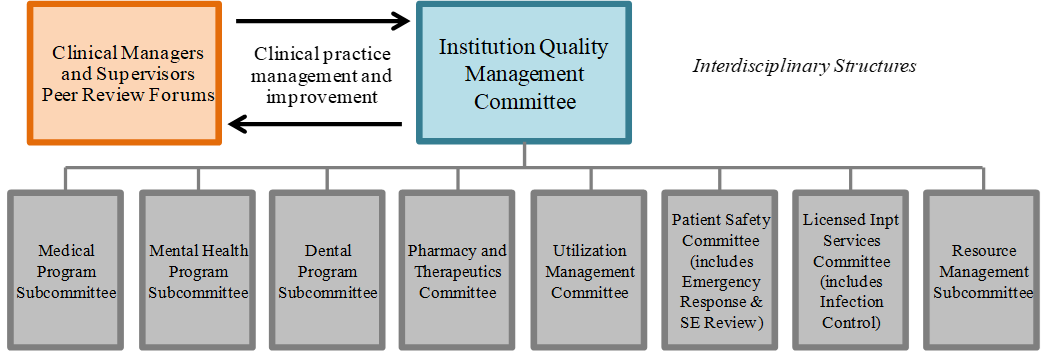
At the institution level, a defined network of program subcommittees, many of which are required by departmental policy or health care licensing regulations, perform the same types of activities as the institution QMC, but within a discreet program area, with reporting requirements to the institution QMC. Appendix 1, Program Specific Subcommittees, provides a matrix of program-specific subcommittees with references to requirements in department policy and state law. Figure 1 shows common subcommittees at the institution level.
-
All standing performance committees and subcommittees have the capacity to form Quality Improvement Teams (QITs), which are time-limited, multi-disciplinary teams convened to analyze and address a particular program or process performance problem. QITs will provide regular updates to the committee that chartered them including a final report at the close of the improvement initiative.
-
All local QMCs report to and receive direction from the statewide QMC. At some institutions, the QMC also reports to a Local Governing Body to meet regulatory requirements, which can serve as an opportunity to promote strategic management of the health care system and prison by the CEO, Warden, and their leadership.
-
Figure 1: Institution Quality Management Program Governance
-
-
-
-
Appendices
-
Appendix 1: Program Specific Subcommittees
-
-
References
-
The design of the QM Program draws from nationally-recognized quality improvement and patient safety organizations, principles, concepts, and models, including:
-
Joint Commission on Accreditation of Hospitals
-
The Care Model
-
Patient-Centered Medical Home
-
International Organization for Standardization (ISO) 9000:2000 standards
-
Institute of Medicine
-
Institute of Healthcare Improvement (Model for Improvement)
-
Baldrige Award Criteria for Process Management and Results
-
Six Sigma
-
Hospital Corporation of America (Focus-PDSA)
-
James Womack (Lean Thinking)
-
W. Edward Demings (Fourteen Points for Managing Quality)
-
Walter J. Shewhart (Cycle for Continuous Improvement)
-
Avedis Donabedian (Structure-Process-Outcome Paradigm)
-
Joseph M. Juran (Pareto Analysis and Juran Quality Trilogy)
-
American Society of Quality
-
-
-
Revision History
-
Effective: 01/2002
Revised: 12/2012
-
-
Appendix 1
-
Program Specific Subcommittees
-
Institution Governance Structure Detail
Committee Applicable Mandates Meeting Frequency Reporting Quality Management Committee HCDOM Section 1.2.3, Quality Management Program (Plata Order)
HCDOM Section 1.2.4, Quality Management Program, Statewide Governance (Plata Order)
HCDOM Section 1.2.5, Quality Management Program, Institution (Plata Order)
NCCHC Multidisciplinary QIC: NCCHC Standards, P-A-06; NCCHC Standards Appendix B; NCCHC Standards Appendix G.
NCCHC Administrative Meeting: NCCHC Standards, P-A-04.Monthly Health Care Services Quality Management Committee, also the Local Governing Body at some institutions with licensed beds Medical Program Subcommittee Quality Management Committee Mental Health Program Subcommittee Mental Health Services Delivery System Program Guide, Chapter10. Suicide Prevention and Response. (Coleman Order) Monthly Quality Management Committee Dental Program Subcommittee HCDOM Section 3.3.4.4, Dental Program Subcommittee (Perez Order) Monthly, or more frequently as necessary Quality Management Committee Pharmacy and Therapeutics Committee Title 22, Division 5, Chapter 12, Article 5, Section 79781 Required Committees.
HCDOM Section 1.2.11, Systemwide Pharmacy and Therapeutics Committee (Plata Order)Not Identified Quality Management Committee Utilization Management Specialty Services Committee Title 15, Division 3, Chapter 2, Subchapter 2, Article 1, Section 3999.200 Provisions of Care and Treatment Exclusions.
Title 15, Division 3, Chapter 1, Subchapter 4, Article 8, Section 3352 Institutional Utilization Management Committee
Title 15, Division 3, Chapter 1, Subchapter 4, Article 8, Section 3352.1 Headquarters Utilization Management Committee
HCDOM, Section 1.2.15, Utilization Management Program (Plata Order)Monthly Quality Management Committee Patient Safety Committee (includes Emergency Response and Sentinel Event Review) HCDOM, Chapter 3, Article 7, Emergency Medical Response (Plata Order) At least monthly Quality Management Committee Licensed Inpatient Services Committee (includes Infection Control) Title 22, Division 5, Chapter 12, Article 5, Section 79781, Required Documentation
HCDOM Sections 1.2.3-1.2.5, Quality Management ProgramAt least annually Quality Management Committee Resources Management
SubcommitteeNot identified Quality Management Committee
-
1.2.4 Quality Management Program, Statewide Governance
-
Purpose
-
To maintain a statewide Quality Management Committee (QMC) to provide oversight to the California Correctional Health Care Services (CCHCS) Quality Management (QM) Program. The structure and processes described in this procedure are designed to:
-
Support implementation of a sustainable, high-performing, integrated health care services delivery system.
-
Promote continuous health care system evaluation and improvement.
-
Promote a culture of teamwork and continuous learning and innovation.
-
-
Responsibility
-
The Statewide Chief Quality Officer (CQO) is responsible for statewide planning, implementation, evaluation and improvement of the QM Program.
-
-
Procedure
-
Overview
-
The statewide QMC:
-
Oversees organization-wide performance management activities, across programs and disciplines, and provides oversight to critical functions within the performance management system including statewide planning, prioritization, design and development of tools and training, evaluation, and improvement.
-
Ensures that the organization’s approach to performance improvement is strategic and, where appropriate, standardized.
-
Oversees efforts to build statewide quality improvement capacity including initiatives to promote skills development at individual institutions and organization-wide in areas such as program evaluation, problem analysis, prioritization of improvement initiatives, and use of evidence-based approaches to performance improvement.
-
Establishes standardized improvement tools, techniques and processes, and supports efforts to implement an organizational culture of continuous learning and improvement.
-
Serves as a supplement to the traditional reporting structure, providing regular interdisciplinary forums for managers, supervisors, and line staff to manage improvement activities.
-
-
The scope of the statewide QMC is separate and distinctly different from the statewide Medical Peer Review Committee (MPRC). The statewide MPRC evaluates and monitors individual clinician practice issues while the statewide QMC oversees organization-wide system performance and improvement activities.
-
-
Statewide Quality Management Committees Activities
-
The statewide QMC works with field leadership, headquarters program-specific subcommittees and workgroups to:
-
Support institutions in the successful implementation of an integrated health care delivery system through improvement initiatives, onsite technical assistance, assessments, and other activities.
-
Establish/update improvement plan with statewide improvement priorities and objectives.
-
Support institutions in identifying improvement priorities and achieving strategic alignment between improvement priorities and day-to-day program activities.
-
Manage statewide improvement initiatives designed to accomplish annual performance objectives.
-
Provide oversight to the statewide performance measurement system, including selection of measures, and ensure dissemination of relevant, accurate, and timely measurement information to headquarters and institution staff through the Dashboard and Institution Scorecards, including standardized processes for data reporting and validation.
-
Support the implementation of the statewide patient safety program, including a health incident reporting system and a defined process for the tracking and analysis of significant events including but not limited to sentinel events.
-
Coordinate with relevant committees and program areas at headquarters and regional level that oversee statewide program policy and operations to share performance information and implement improvement initiatives (e.g. Joint Clinical Executive Team and Clinical Operations Team).
-
Coordinate technical assistance activities by staff at headquarters and regional teams designed to support improvement initiatives.
-
Refer issues to other committees or programs, such as the statewide MPRC, when they do not fall under the purview of the QMC.
-
Implement statewide training and staff development programs to orient all health care staff to the QM Program, promote the development of quality improvement skills, and support staff at all reporting levels to participate in quality improvement.
-
Establish uniform processes and tools for the analysis of system performance problems, development of solutions, and testing and evaluation of interventions.
-
Establish forums for sharing lessons learned and disseminating best practices.
-
Actively foster an organizational culture of continuous learning, improvement, and innovation by clearly communicating performance improvement principles to staff; evaluating CCHCS governance structures and processes, policies, procedures, practices, training programs, and communications against those principles; and providing input to ensure alignment.
-
Establish a process to periodically review the statewide QM Program to evaluate the overall effectiveness of the QM Program.
-
-
QMC Membership and Meeting
-
QMC Chairperson
-
The statewide CQO and another executive leader chosen by the QMC members shall serve as Co-Chairpersons. The Co-Chairpersons are responsible for ensuring that the QMC meets at least quarterly, the committee agenda reflects the responsibilities and actions described in this procedure, and committee decisions are appropriately documented.
-
-
QMC Voting Members
-
Deputy Director, Statewide Mental Health Program
-
Deputy Director, Statewide Dental Program
-
Statewide, Chief Nurse Executive
-
Statewide, Chief Medical Executive
-
Director, Allied Health Services
-
Deputy Director, Medical Services
-
Deputy Director, Strategic Management
-
Director, Policy and Risk Management Services
-
Chief Information Officer, Information Technology Services
-
Deputy Director, Human Resources
-
Director, Administrative Support Services
-
Director, Corrections Services
-
Chief Executive Officer(s)
-
Statewide, Chief Quality Officer
-
Representative, Division of Adult Institutions
-
Other members nominated by one of the voting members and approved by the QMC. All voting members may choose a designee to serve in their stead.
-
-
QMC Meetings
-
The committee shall meet no less than quarterly. Each member has one vote and a quorum is designated as 50% of members.
-
The committee shall document each meeting through formal minutes and provide them to QMC members for review no later than one week prior to the next meeting.
-
-
Reporting Structure
-
The statewide QMC reports to the highest ranking health care official and executive staff at least annually on progress in meeting annual performance improvement objectives and patient safety goals.
-
Statewide program-specific subcommittees and institution QMCs shall report quality improvement and patient safety activities to the statewide QMC at least annually or more often as appropriate.
-
-
QMC and Subcommittees
-
The statewide QMC shall coordinate and communicate with subcommittees to establish and sustain a high-performing health care system consistent with the Primary Care Model and existing policies and procedures, state and federal law, and community standards of care. Examples of subcommittees may include:
-
Patient Safety Program
-
Mental Health Program
-
Dental Program
-
Medical Program
-
Pharmacy and Therapeutics
-
Diagnostic Services
-
Utilization Management
-
Health Information Management
-
Continuing Medical Education
-
Clinical Guidelines
-
Resource Management
-
-
Each Subcommittee Chairperson is responsible for reporting subcommittee program performance improvement activities, such as development and implementation of initiatives and improvement projects, to the statewide QMC on a routine basis through appropriate documentation (e.g., minutes) and verbal reporting.
-
All subcommittees shall meet at least as frequently as required in existing policy.
-
-
-
-
Revision History
-
Effective: 01/2002
Revised: 12/2012
-
1.2.5 Quality Management Program, Institution
-
Purpose
-
To describe strategies, processes, tools, and a governance structure that institutions use to plan, prioritize, develop, implement, and evaluate performance improvement initiatives and sustain improvements.
-
-
Responsibility
-
The Institution Chief Executive Officer (CEO) is responsible for implementation of this procedure.
-
-
Procedure
-
Performance Improvement Plan
-
Integrated Health Care Services Delivery System
-
Implementation of an integrated health care services delivery system with a strong primary care foundation has been identified as an overarching strategy for improving care and avoiding unnecessary morbidity, mortality, and costs. Health care staff at each institution are responsible for implementing care processes necessary for a sustainable integrated health care delivery system including, but not limited to, processes to:
-
Identify the health risk of each patient and ensure that the patient is placed at an institution and/or treatment setting with the capacity to address his or her health care needs, per an automated Clinical Risk Classification process.
-
Establish consistent interdisciplinary care teams that assume primary responsibility for the patients assigned to them.
-
Provide care coordination for patients as they move from one treatment setting to another, including transitions between care teams, and across levels of care.
-
Ensure that care teams work across disciplines to co-manage each patient as appropriate to his or her health needs.
-
Ensure care teams have necessary resources and information, such as updated health records, specialty care reports, discharge summaries, diagnostic study results, and patient registries, to provide planned care for patients.
-
Provide evidence-based care that is consistent with current guidelines and community standards.
-
Enhance access to services through open access appointments or other scheduling strategies, after-hours care, and effective communication techniques.
-
Ensure that patients have timely and consistent access to medications.
-
Use Patient Registries to manage subpopulations with specific health risk factors or chronic disease, including high risk patients and mental health patients that are high utilizers of services.
-
Promote patient self-management to improve outcomes.
-
-
Priority-Setting Process
-
Each year, institutions shall review health care areas considered to be high risk, high volume, high cost, and problem-prone, and identify improvement priorities taking into consideration statewide strategic priorities and customizing the plan for the facility’s health care mission, resources, and the needs of the patient population. The annual performance improvement plan shall include the following elements:
-
Priority areas for improvement to be targeted by institution staff in the coming year.
-
Performance objectives for each priority area (generally six to twelve) and associated timeframes.
-
Improvement strategies that will be used to achieve performance objectives.
-
-
Development of an Annual Performance Improvement Plan
-
As a first step in developing an improvement plan, each California Department of Corrections and Rehabilitation institution shall evaluate the health care delivery system and identify gaps in areas such as those listed above.
-
Each institution shall prioritize areas to be targeted in improvement initiatives, set measurable performance objectives for each area, and determine which strategies the institution will use to achieve objectives.
-
Upon developing an improvement plan for the year, institution leadership shall take steps to inform institution staff about the improvement priorities for the year and help managers, supervisors, and line staff to identify specific ways they might contribute to improvement efforts as part of their current duties. To this end, institution leadership is responsible for:
-
Communicating improvement priorities to staff at all levels of the health care system.
-
Helping staff understand their role in achieving improvement objectives.
-
Guiding the process of strategic alignment by which managers and supervisors determine how program operations and day-to-day supervision will support performance objectives and how care teams and other staff incorporate improvement activities for priority areas into their day-to-day work.
-
Updating health care staff about any changes to the improvement plan that occur throughout the year.
-
Keeping institution staff apprised of progress toward meeting objectives in the annual improvement plan.
-
-
-
-
Performance Evaluation
-
Overview
-
As part of the development of an annual improvement plan, each institution shall determine performance measures and objectives for each priority area identified in the improvement plan. Progress toward the achievement of improvement objectives should be monitored at least monthly and regularly conveyed to all health care staff. To make data as useful as possible as a catalyst for change, institutions may consider generating reports that break down performance by:
-
Clinic or care team.
-
Primary Care Provider, Primary Mental Health Clinician, Psychiatrist, or Dentist.
-
Patient subpopulation (e.g., high risk or mental health high utilizers) or other meaningful subgroup.
-
This may help health care staff determine which locations, staff, or patients might receive improvement interventions first, and is generally useful in identifying care teams or individual providers who might present best practices.
-
-
Health Care Services Dashboard
-
For the purposes of identifying opportunities for improvement and potential patient safety concerns, institutions are also required, through Quality Management Committee (QMC) meetings, Quality Improvement Team (QIT) meetings, and other forums, to monitor the performance of critical clinical and administrative processes monthly through the Health Care Services Dashboard (Dashboard).
-
The Dashboard includes institution-level performance data, trended over time, in the form of Institution Scorecards, trended composites, and other data displays. Some of the Dashboard data is derived from institution reports.
-
To ensure that Dashboard data is as useful as possible in informing management decisions, institutions shall take action to ensure accuracy in data reporting including:
-
Ensuring that staff applies standardized methods for data collection, in accordance with statewide policies, procedures, and detailed instructions.
-
Ensuring that staff collecting data are appropriately trained.
-
Periodic data validation through redundant data collection by an independent reviewer (e.g. for clinical practice reviews) and/or checking database information against another data source, such as patient charts to double-check accuracy of health care scheduling data.
-
Conducting inter-rater reliability analysis for reviewers of qualitative data.
-
Use of statistically-valid samples.
-
-
-
Performance Reports
-
In addition to the monthly Dashboard, California Correctional Health Care Services (CCHCS) issues periodic performance reports and special studies including morbidity and mortality analysis.
-
Institution CEOs are responsible for ensuring that these reports are broadly distributed to health care staff and discussed in various meeting forums such as staff meetings and quality improvement committee meetings.
-
-
Patient Registries
-
CCHCS provides lists of patients with targeted dental, mental health, and/or medical conditions to institution staff, updated at least monthly.
-
Patient Registries often include an alert function where patients with abnormal findings or who are missing services required by CCHCS guidelines are highlighted. Institution care teams are required to review registries at least monthly (and more often as appropriate) and take action to follow-up with patients as necessary to improve patient outcomes.
-
-
Data Integrity
-
When an institution determines that there may be a problem with data accuracy, the institution shall immediately notify the QM Section and shall institute corrective actions to ensure data reliability.
-
-
-
Quality Improvement Techniques, Tools and Training
-
Consistent with health care industry standards, institutions shall use improvement processes, techniques, and tools to assist health care staff in establishing and maintaining an integrated health care delivery system, achieving objectives outlined in annual improvement plans, and regularly evaluating and redesigning health care processes. Institutions shall:
-
Establish clear and measurable objectives for all improvement projects.
-
Re-evaluate critical health care processes on a regular basis and redesign operations to implement an integrated health care delivery system and improve outcomes and efficiency.
-
Apply improvement models used widely in the health care and quality improvement industries such as Root Cause Analysis, the Model for Improvement, Lean Model, Six Sigma, Focus-Plan-Do-Study-Act, Failure Mode and Effects Analysis, Process Flow Diagramming, and Cause and Effect Diagramming to analyze performance problems, develop solutions, and test and evaluate interventions.
-
Use Patient Registries to identify and manage high risk patients and patients with specific chronic conditions and standardized decision support tools disseminated by the QM Program such as care guides and quality of care review tools, in the application of improvement processes and techniques.
-
Take steps to sustain improvements when an initiative has proven successful such as memorializing new processes in Local Operating Procedures (LOP) or documenting new staff roles in duty statements.
-
Orient and train staff on QM Program structures, processes, and tools and ensure that staff attends statewide training programs designed to build improvement skills in the field.
-
Take actions to promote a culture of teamwork and continuous learning and innovation.
-
-
The Cycle of Change described in the Quality Management Program Overview presents a framework for the design and implementation of institution improvement initiatives, incorporating the principles described above.
-
-
-
Quality Management Program Governance
-
Overview
-
Each institution shall maintain an interdisciplinary QM Program structure to monitor and direct performance evaluation and improvement activities. The management structure includes standing committees at two levels:
-
An institution QMC that plays a central role in coordinating performance evaluation and improvement activities institution-wide, providing overall strategic direction for the institution quality QM system and communicating performance improvement activities to the statewide QMC.
-
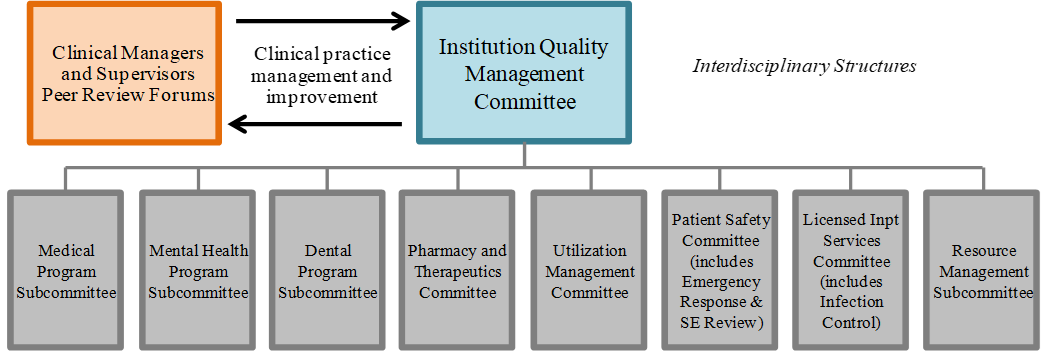
Subcommittees that evaluate performance at the program level, develop program-specific improvement plans and manage implementation of improvement initiatives. Image 1 provides a schematic of the management structure overseen by the institution QMC.
-
-
The institution QMC or any subcommittee may convene a QIT, a multi-disciplinary team charged with addressing a particular improvement opportunity. QITs typically exist for the duration of the improvement project and disband upon completion.
-
Other forums such as staff meetings, weekly provider meetings, Primary Care Team huddles, and monthly all Primary Care Team meetings serve as important conduits of performance improvement information and support implementation of improvement initiatives including, but not limited to:
-
Ensuring all staff understand the elements of the integrated health care services delivery system consistent with the Primary Care Model and the QM system.
-
Communicating annual improvement priorities and objectives.
-
Reviewing performance data, particularly at the clinic or provider level.
-
Disseminating decision support tools and conveying expectations for use.
-
Conducting staff development activities consistent with priority improvement areas.
-
Developing and implementing improvement initiatives, including process redesign, that align with the institution’s improvement priorities and may be specific to a discipline (e.g., health records technicians) or work location (e.g., Yard B).
-
Sharing best practices.
-
Figure 1: Institution Quality Management Program Governance – Standing Committees
-
-
-
Institution Quality Management Committee
-
Overview
-
In general, the institution QMC provides oversight to local performance improvement activities and ensures that health care programs operate in adherence with applicable laws, regulations, policies, procedures, and standards of care; the institution QMC serves as a hub for change management, collaboration, and coordination across programs and disciplines as activities relate to performance management and improvement, and serves as a conduit of improvement and patient safety information from the institution to headquarters.
-
Among other responsibilities, the institution QMC:
-
Evaluates the performance of the integrated health care services delivery system at the institution, including CCHCS Primary Care Model, and directs efforts to improve care.
-
Sets improvement priorities, regularly evaluates program performance and takes action to make improvements, and coordinates the work of multiple standing committees focusing on particular health care programs or functions.
-
Ensures that institution staff is trained in quality improvement concepts, processes, and tools, promotes a culture of continuous learning and improvement at the institution.
-
-
The scope of the institution QMC is separate and distinctly different from the organized medical staff. The role of an organized medical staff at the institution, if it exists, is peer review and addressing individual clinician practice issues in conjunction with management while the institution QMC oversees organization-wide system performance and improvement activities, which requires involvement of all disciplines and line staff, supervisors and managers. (refer to Image 1)
-
-
Committee Actions
-
Specifically, the institution QMC takes on the following actions:
-
Strategic Direction
-
Identifies institution-specific improvement priorities and objectives through the development of an annual performance improvement plan.
-
Aligns institution performance improvement activities with statewide improvement priorities and initiatives.
-
Provides feedback to the statewide QMC on statewide improvement priorities, performance metrics, and initiatives.
-
-
Program Evaluation and Improvement
-
Reviews program performance data, including the organizational Dashboard and Institution Scorecards, statewide performance reports, QIT reports, and reports by outside entities, including the Office of the Inspector General, court monitors, and the Prison Law Office, at least monthly.
-
Uses performance data to identify improvement opportunities, evaluate the effectiveness of quality improvement interventions in the Institution Performance Work Plan, and assess the performance of the integrated health care services delivery system, and determine progress toward performance objectives.
-
Takes action when quality or safety problems are identified incorporating new initiatives in the Institution Performance Work Plan, including assigning interdisciplinary QITs to analyze quality problems, select solutions, and implement quality improvement initiatives.
-
Approves local improvement initiatives for implementation institution-wide and monitor them through the Institution Performance Work Plan including development or modification of health care programs, pilot programs, changes to LOPs, training programs, and development or modification of decision support.
-
Provides oversight during licensing surveys, audits by external stakeholders, and inspections, develops and manages implementation of improvement plans.
-
Oversees implementation of policies, evidence-based guidelines and standards of care.
-
-
Communication and Coordination
-
Ensures dissemination of relevant, accurate, and timely performance information to institution management and staff.
-
Ensures that QMC, subcommittee, and workgroup activities are appropriately documented.
-
Coordinates and reviews subcommittee activities and recommends program changes or further subcommittee activity.
-
Ensures effective communication between local improvement committees, especially in areas of interdisciplinary responsibility or system-wide issues.
-
Identifies and disseminates best practices.
-
Coordinates annual review and approval of institution LOPs.
-
Refer issues to other committees or programs such as the statewide Medical Peer Review Committee when they do not fall under the purview of the QMC.
-
Reports institution performance improvement plans and activities and recommendations for state-level improvements, to the statewide QMC at least annually.
-
-
Orientation and Training
-
Implements and oversees a local staff development program to orient all health care staff to the QM Program, promote the skills necessary to perform quality improvement work at institutions, and empower staff at all reporting levels to participate in quality improvement.
-
-
-
QMC Membership, Meetings, and Reporting
-
QMC Chairperson
-
The institution CEO shall serve as QMC Chairperson.
-
The QMC Chairperson is responsible for ensuring that the QMC meets at least monthly.
-
The committee agenda reflects the responsibilities and actions described in this procedure, program subcommittees report per a designated schedule, and committee decisions are appropriately documented.
-
-
QMC Members
-
The institution QMC shall include the following voting members:
-
Chief Executive Officer
-
Institution CQO
-
Institution Chief Medical Executive
-
Institution Chief Nurse Executive
-
Institution Chief Support Executive
-
Chief, Mental Health Program
-
Health Program Manager III, Dental Program
-
Pharmacist-in-Charge
-
Associate Warden of Health Care
-
Chairpersons of QMC Subcommittees
-
Other members as deemed appropriate such as the Director of Nursing, Supervising Registered Nurse II, Correctional Health Care Services Administrator, Chief Psychiatrist, Supervising Dentist, Nurse Instructor, Public Health Nurse, Health Program Specialists, Standards Compliance Coordinator.
-
-
QMC Meeting Frequency
-
It is recommended that the institution QMC meet weekly. However, at a minimum, the committee shall meet no less than monthly.
-
-
QMC Minutes
-
The Institution QMC shall document each meeting through formal minutes that shall be provided to QMC members for review no later than three business days prior to the next QMC meeting.
-
-
QMC Reporting
-
The Institution QMC reports to and receives direction from the statewide QMC.
-
At some institutions, the QMC also reports to a Local Governing Body to meet regulatory requirements, which can serve as an opportunity to promote strategic management of the health care system and prison by the CEO, Warden and their leadership.
-
-
-
-
QMC Subcommittees
-
Each institution shall maintain subcommittees to establish and sustain high-quality health care services within a defined program area consistent with existing policies and procedures, federal and state law, and community standards of care.
-
A number of standing subcommittees are required by departmental policy or state regulations. Other subcommittees may be established to review and improve performance of a specific program or resource area. Examples of common subcommittees include:
-
Medical Program
-
Mental Health Program, may include Suicide Prevention and Response (also see Patient Safety)
-
Dental Program
-
Pharmacy and Therapeutics
-
Utilization Management
-
Patient Safety
-
Licensed Inpatient Services focuses on compliance with Title 22 of the California Code of Regulations including Infection Control and Patient Care Policy Committees
-
Resource Management including Human Resources, Health Information Management and scheduling systems, Budgets and Physical Space.
-
-
Institutions with licensed facilities may also choose to incorporate the functions of a Patient Policy Committee into the QMC if appropriate.
-
Each Subcommittee Chairperson serves as a member of the QMC and is responsible for reporting subcommittee program performance improvement activities, such as development and implementation of initiatives and improvement projects, to the institution QMC on a routine basis through appropriate documentation (e.g., minutes) and verbal reporting.
-
All subcommittees shall meet as frequently as required in existing policy or state law and meeting minutes shall be completed and readily available for review.
-
-
Quality Improvement Teams (QITs)
-
All standing performance committees and subcommittees have the capacity to form QITs which are time-limited, multi-disciplinary teams convened to analyze and address a particular program or process performance problem.
-
QITs shall provide regular updates to the committee that chartered them and a final report at the close of the improvement initiative.
-
-
-
References
-
California Code of Regulations, Title 22, Division 5, Article 5, Section 79781, Required Committees
-
California Department of Corrections and Rehabilitation, Mental Health Services Delivery System Program Guide, Chapter 10, Suicide Prevention and Response
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.11, Systemwide Pharmacy and Therapeutics Committee
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.15, Utilization Management Program
-
Health Care Department Operations Manual, Chapter 3, Article 3, Section 3.3.4.4, Dental Program Subcommittee
-
-
Revision History
-
Effective: 01/2002
Revised: 12/2012
-
1.2.6 Statewide Patient Safety Program
-
Policy
-
California Correctional Health Care Services (CCHCS) shall maintain a Statewide Patient Safety Program to identify and improve problematic health care processes by emphasizing the prevention, reduction, reporting, and analysis of health care incidents that if left unaddressed may result in adverse drug reactions, sentinel events, or preventable patient harm.
-
The CCHCS Statewide Patient Safety Program encompasses:
-
Patient safety priorities that are reviewed and revised biennially to identify program objectives for statewide interventions and performance improvement activities.
-
A comprehensive multidisciplinary health care incident reporting and review process for identifying, reporting, and assessing health care incidents including sentinel events, in accordance with state law and health care industry best practices, to address potential systemic health care process issues and mitigate risk to patients, staff, and visitors.
-
A committee structure at headquarters to provide oversight to the Statewide Patient Safety Program, review patient safety reports and data, and take action to mitigate patient safety risks and prevent adverse patient outcomes.
-
A committee structure at each institution that oversees the local implementation of the Patient Safety Program by reviewing patient safety reports and data at the individual institution or care team level to identify and mitigate patient safety risks, and prevent adverse patient outcomes.
-
Regular communication in the form of patient safety alerts, aggregate reporting of findings and recommendations related to health care incidents or Root Cause Analyses (RCAs) that may be used to inform additional performance improvement efforts, patient safety initiatives, or recommendations to modify statewide policies and procedures.
-
Technical assistance, decision support tools (e.g., job aids, guides, forms, checklists, and flowcharts), and staff development and education programs to support problem analysis, RCA, and process redesign.
-
A patient safety culture that encourages staff to proactively identify and report health care incidents to mitigate risk to patients and emphasize continuous learning and improvement.
-
-
-
Purpose
-
To protect patients, staff, and visitors from poor health outcomes due to flawed health care processes; improve health care quality and cost effectiveness; increase health care process efficiencies and reduce waste; and comply with legal and regulatory requirements.
-
-
Responsibility
-
Statewide
-
California Department of Corrections and Rehabilitation (CDCR) and CCHCS departmental leadership at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial and clinical systems are in place and appropriate tools, training, technical assistance, and levels of resources are available to ensure appropriate, timely, safe, and cost-effective health care for patients.
-
The Statewide Patient Safety Committee, a subcommittee of the Statewide Quality Management Committee, is responsible for providing oversight of the Patient Safety Program at the statewide level, identifying and communicating program priorities, and managing implementation of patient safety initiatives.
-
The Statewide Health Care Incident Review Committee, a subcommittee of the Statewide Patient Safety Committee, is responsible for providing oversight of the health care incident reporting system and RCA process at the statewide level, and identifying and communicating related data and trends.
-
-
Regional
-
Regional Health Care Executives are responsible for implementation of this policy at the subset of institutions within an assigned region.
-
Regional Health Care Support Teams shall ensure health care staff utilize the centralized electronic Health Care Incident Reporting (eHCIR) system, and use incident data and trends to identify and take action to mitigate patient safety risks within an individual institution or across a region. Regional Health Care Support Teams shall provide oversight, support, and monitoring of RCAs assigned to institutions within their respective region.
-
-
Institutional
-
The Chief Executive Officer (CEO), or designee, is responsible for implementation of this policy at the institution level.
-
Institution leadership teams including the CEO, Chief Support Executive, Chief Medical Executive, Chief Nurse Executive, Chief of Mental Health, and the Supervising Dentist are jointly responsible for the planning, implementation, evaluation, and monitoring of the local Patient Safety Program and ensuring adherence to the Patient Safety Program policy and procedures. Institution leadership teams are responsible for:
-
Ensuring the institution utilizes the institution Quality Management Committee or other designated local committee to identify and address problematic patient safety trends identified through the eHCIR system.
-
Ensuring staff have access to resources including equipment, supplies, and health information or reporting systems.
-
Encouraging and supporting timely reporting of health care incidents including sentinel events.
-
Identifying Patient Safety Champions to provide subject matter expertise and technical support to staff.
-
-
-
-
Procedure
-
Patient Safety Program Committees and Plans
-
The Statewide Patient Safety Program shall maintain a Statewide Patient Safety Committee to provide oversight of the Patient Safety Program at the statewide level.
-
The Statewide Patient Safety Program shall identify program priorities on at least a biennial basis that describe the goals and objectives for the Patient Safety Program identified through system surveillance, health care incident reporting trends, root cause analysis findings and recommendations, and/or changes in community best practices.
-
-
Statewide Health Care Incident Report Review
-
The Patient Safety Program shall conduct an initial review of all health care incidents reported to the eHCIR system to determine an appropriate disposition and ensure potential sentinel events or other anomalous health care incidents with an assigned severity level of 4 through 6 are referred for review by the Statewide Health Care Incident Review Committee (HCIRC).
-
-
Statewide Patient Safety Committee
-
Responsibilities
-
The Statewide Patient Safety Committee (PSC) protects patient safety and improves the health care delivery system by:
-
Designing a surveillance system for the centralized collection and review of data pertinent to patient safety.
-
Maintaining an effective process for assessing, referring, and analyzing or making conclusions regarding reported health care incidents including, but not limited to:
-
A centralized eHCIR system for identifying and reporting patient safety issues, near misses, sentinel events, and other anomalous health care incidents.
-
A Health Care Incident Reporting Registry and Incident Summary accessible to select health care staff to ensure that institutions are informed of health care incident reports specific to their individual institution and patient care areas including licensed patient care areas which may require additional mandatory reporting to regulatory agencies.
-
A standardized process for the identification and examination of sentinel events using the RCA process and CCHCS RCA Tool Kit.
-
A process for referring health care incidents that involve blameworthy acts/reckless behaviors, including criminal activities, to the appropriate professional practice entities or hiring authority.
-
-
Reviewing program and surveillance data to identify problematic health care processes and establishing patient safety priorities and program objectives for statewide interventions and performance improvement activities.
-
Establishing a multidisciplinary Statewide HCIRC with headquarters level representation from all health care disciplines (i.e., Medical Services, Nursing Services, Pharmacy Services, Dental Services, and the Mental Health Program including Psychiatry) and custody.
-
Ensuring that Regional Health Care Executives designate Regional Health Care Support Teams to oversee the completion of RCAs assigned to institutions within their region and providing feedback and consultation as needed.
-
Collaborating with other program areas to redesign health care processes to improve patient safety.
-
Developing statewide training programs and decision support tools (e.g., tool kits, job aids, guides, forms, checklists, and flowcharts) to support patient safety surveillance, health care incident reporting and review, RCAs, and process redesign.
-
Coordinating patient safety activities with other committees and programs when appropriate including, but not limited to, the Statewide HCIRC, Mortality Review Committee, Systemwide Pharmacy and Therapeutics Committee, and Statewide Quality Management Committee (QMC).
-
Issuing a Patient Safety Dashboard at least quarterly that aggregates eHCIR data about health care incident reporting trends to inform continuous process improvement.
-
Issuing statewide patient safety alerts if a health care incident or trend reveal a problem that all institutions should immediately address or be aware of.
-
Supporting outreach and other activities that encourage reporting of all health care incidents including near misses, medication events, and sentinel events; and
-
Promoting an organizational culture of continuous learning and improvement.
-
-
Membership, Quorum Requirements, and Meeting Frequency
-
The Deputy Director, Quality Management, or designee, shall serve as chairperson of the Statewide PSC. The chairperson is responsible for ensuring that the statewide PSC meets regularly, the committee agenda reflects the responsibilities described in this procedure, and committee decisions are appropriately documented.
-
The statewide program lead in each respective health care discipline (i.e., Medical Services, Nursing Services, Pharmacy Services, Dental Services, and the Mental Health Program), as well as the program leads in California Correctional Health Care Services Office of Legal Affairs and Health Care Policy and Administration, shall select at least one headquarters designee to serve on the Statewide PSC. Other program representatives may be asked to serve as members at the discretion of the Statewide PSC.
-
All voting members may choose a designee to serve in their stead. Non-voting members, such as presenters and guests, may attend as appropriate and approved by the Statewide PSC.
-
Each member has one vote and a quorum exists when more than one-half of voting members are present.
-
The Statewide PSC shall meet at least quarterly or more often as necessary to carry out its responsibilities.
-
Electronic records of committee proceedings shall be kept in a secure location indefinitely.
-
-
Reporting Relationships
-
The Statewide PSC reports to the Statewide QMC, and provides oversight to the Statewide HCIRC.
-
-
-
Statewide Health Care Incident Review Committee
-
Responsibilities
-
The Statewide HCIRC protects patient safety and improves the health care delivery system and shall:
-
Conduct a review of all health care incidents reported to the eHCIR system with an assigned severity level of 4 through 6, potential sentinel events, and any other anomalous health care incidents to determine the appropriate disposition and ensure that any immediate danger to patients, staff, or visitors is addressed.
-
Communicate information about health care incidents to the appropriate regional and institution program leads that are reported by a person or entity not employed at an institution, such as court experts.
-
Refer health care incidents with identified clinical practice concerns or blameworthy acts/reckless behavior to the appropriate peer review committee or hiring authority.
-
The HCIRC shall coordinate with institution leaders of the appropriate discipline to determine whether clinical staff involved in the health care incident should be removed from providing patient care pending further analysis of the event.
-
Health care incidents assigned an RCA that result in a referral to peer review or the hiring authority or temporary redirection of health care staff from direct patient care shall have the RCA continue without delay or deferral and the staff person(s) referred shall be excluded from the RCA process.
-
-
Identify sentinel events and assign RCAs, including collaboration with Regional Health Care Support Teams and institution leadership, to further inform the case prior to assignment, and when appropriate, recommend grouping multiple incidents that are similar in nature into an aggregate RCA.
-
Work with Regional Health Care Support Teams to ensure RCAs are completed, approved, and submitted within 45 business days of assignment.
-
Upon receipt of an RCA Report, coordinate with the institution and Regional Health Care Support Team within 15 business days to provide feedback, including requesting revisions and clarification to the RCA Report if the report does not meet requirements for a thorough and credible RCA.
-
Upon approval of the RCA Report, monitor progress of the RCA Plan of Action for at least four months, and coordinate with the Regional Health Care Support Team to provide additional support or intervene as needed;
-
Advocate for changes to statewide policies and procedures in accordance with findings from patient safety surveillance, health care incident trends, or RCA recommendations.
-
Promote an organizational culture of continuous learning and improvement.
-
-
Committee Membership, Quorum Requirements, and Meeting Frequency
-
The statewide program lead in each respective health care discipline (i.e., Medical Services, Nursing Services, Pharmacy Services, Dental Services, and the Mental Health Program including Psychiatry) and custody shall select at least one headquarters designee to serve on the Statewide HCIRC.
-
The Statewide PSC shall elect one or two HCIRC members to serve as chairperson(s). The responsibility of the chairperson(s) is to ensure that the HCIRC meets regularly, the committee agenda reflects the responsibilities described in this procedure, and committee decisions are appropriately documented.
-
All members may choose a designee to serve in their stead. Non-voting members, such as presenters and guests, may attend as appropriate and approved by the HCIRC.
-
Each member has one vote, and a quorum exists when at least three voting members are present.
-
The HCIRC shall meet each business day to carry out its responsibilities.
-
Electronic records of committee proceedings shall be kept in a secure location indefinitely.
-
-
Reporting Relationships
-
The Statewide HCIRC reports to the Statewide PSC.
-
-
-
Regional Health Care Support Teams
-
Regional Health Care Executives shall designate Regional Health Care Support Teams that include regional representatives from Medical Services, Nursing Services, Pharmacy Services, Dental Services, and Mental Health Program, including Psychiatry, to provide support for institution RCAs as needed and on an individual case-by-case basis.
-
Regional Health Care Support Teams shall provide RCA consultation, guidance, facilitation, and follow-up to the subset of institutions within their assigned region including, but not limited to:
-
Reviewing health care incidents and collaborating with the Statewide HCIRC and institution leadership on identified sentinel events prior to the assignment of an RCA.
-
Ensuring institution RCA Teams are multidisciplinary and appropriate for the sentinel event under review.
-
Ensuring information is gathered including a chronology of the event before, during, and after the incident, as well as other relevant information, guidelines, best practices, and literature used to inform the brainstorming session and problem analysis.
-
Participating in brainstorming sessions, encouraging thorough discussion of factors contributing to the sentinel event, and ensuring that the fishbone diagram is complete and causal statements are sound.
-
Ensuring the RCA Team identifies root causes.
-
Ensuring that RCA Plans of Action adequately address the identified root causes and associated performance objectives are measurable, realistic, and attainable.
-
Reviewing draft RCA Reports and Plans of Action to ensure they are thorough and credible per CCHCS RCA Tool Kit guidelines.
-
Monitoring Plans of Action submitted by the institution to ensure progress is being made, determining whether additional support or intervention is necessary, and taking action as appropriate.
-
-
-
Confidentiality
-
Protected Proceedings and Records
-
It is critical that the proceedings and records of the health care incident review process be maintained as confidential and not be made available to unauthorized persons or organizations.
-
All staff participating in the health care incident review process discussed in this procedure shall adhere to these provisions regarding confidentiality.
-
The records of the committees and staff responsible for the evaluation and improvement of the quality of patient care shall be maintained as confidential and protected from discovery to the extent permitted by law.
-
-
-
References
-
California Health and Safety Code, Division 2, Chapter 2, Article 1, Section 1250
-
California Health and Safety Code, Division 2, Chapter 2, Article 3, Sections 1279, 1279.1, and 1279.2
-
California Code of Regulations, Title 22, Division 5, Chapter 12, Article 5, Section 79787, Reporting
-
California Department of Corrections and Rehabilitation, Department Operations Manual, Chapter 3, Article 2, Health and Safety Program
-
California Department of Corrections and Rehabilitation, Department Operations Manual, Chapter 5, Article 11, Section 51110.11, Written Reports
-
California Department of Corrections and Rehabilitation, Mental Health Services Delivery System Program Guide, Chapter 10, Suicide Prevention and Response
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.7, Institution Patient Safety Program
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.10, Mortality Review and Reporting
-
Health Care Department Operations Manual, Chapter 3, Article 5, Section 3.5.11, Pharmacy Quality Assurance Program
-
Food and Drug Administration, MedWatch: The FDA Safety Information and Adverse Event Reporting Program (http://www.fda.gov/safety/medwatch/default.htm)
-
The Joint Commission https://www.jointcommission.org/topics/patient_safety.aspx
-
National Commission on Correctional Health Care 2008 Standards for Health Services in Prisons
-
National Coordinating Council for Medication Error Reporting and Prevention
-
United States Department of Veterans Affairs – Veterans Affairs National Center for Patient Safety (http://www.patientsafety.va.gov/)
-
Veterans Health Administration Vision 2020
-
California Department of Public Health Center for Health Care Quality (https://www.cdph.ca.gov/Programs/CHCQ/Pages/CHCQHome.aspx)
-
-
Revision History
-
Effective: 11/2012
Revised: 11/2018
-
1.2.7 Institution Patient Safety Program
-
Procedure
-
Institution Patient Safety Oversight
-
The Institution Quality Management Committee or other designated local committee shall provide oversight of patient safety and improve the local health care delivery system by:
-
Reviewing local patient safety surveillance data, including the Patient Safety Dashboard and Health Care Incident Reporting Registry trends, to identify problematic health care processes and establish priority areas for intervention and patient safety risk mitigation activities, and when appropriate, recommend the addition of patient safety priorities to the Institution Performance Improvement Work Plan.
-
Coordinating and collaborating with other committees and program areas as appropriate to redesign local health care processes to improve patient safety.
-
Ensuring oversight and timely completion and monitoring of Root Cause Analysis (RCA), including coordination with Regional Health Care Support Teams and the Statewide Health Care Incident Reporting Committee.
-
Ensuring health care staff have access to patient safety training and decision support tools (e.g., tool kits, job aids, guides, forms, checklists, and flowcharts) to support patient safety surveillance, health care incident reporting and review, RCAs, and process redesign.
-
Supporting outreach and other activities that encourage reporting of all health care incidents including near misses, medication events, and sentinel events by institution health care staff.
-
Communicating patient safety alerts, issues, and RCA recommendations and actions to institution staff.
-
Identifying local Patient Safety Champions and promoting an organizational culture of continuous learning and improvement.
-
-
Identification of Health Care Incidents and Duty to Report
-
All California Department of Corrections and Rehabilitation/California Correctional Health Care Services (CCHCS) staff have a duty to report health care incidents using the centralized electronic Health Care Incident Reporting (eHCIR) system within 24 hours of occurrence or discovery. The eHCIR is available to all staff via Lifeline and allows health care incidents to be submitted anonymously.
-
While many health care incidents will be initially detected by institution health care staff, health care incidents may also be identified by other stakeholders.
-
Institutions with licensed beds, including Correctional Treatment Centers, Mental Health Crisis Beds, Psychiatric Inpatient Program/Intermediate Care Facilities, Skilled Nursing Facilities, Dialysis, or Hospice are required to report certain health care incidents to the California Department of Public Health. Reporting requirements to external agencies may differ for each institution and must be verified by the facility. All institutions shall comply with health care incident reporting requirements in departmental policy, and federal and state law.
-
Institution leadership, program leads, unit supervisors, and other required health care staff as identified shall monitor the Health Care Incident Reporting Registry and associated Incident Summaries daily for reported health care incidents at their institution and take appropriate action as required by federal and state law; for example, Pharmacy Quality Assurance reviews for medication errors, Professional Practice Reviews, and Psychiatric Inpatient Program incident review and referrals to regulatory agencies.
-
Program leads or any person with a leadership role or authority shall not prohibit or create any physical or process barriers that prevent or delay staff from reporting health care incidents to the eHCIR.
-
-
Immediate Action Following a Health Care Incident
-
Mitigate Risk
-
Upon realizing that a health care incident has occurred, institution health care staff shall immediately take steps to ensure patient safety including, but not limited to:
-
Stabilizing the patient by providing all necessary and appropriate care.
-
Removing all unsafe devices, equipment, and medications.
-
Determining whether the health care incident places other patients, staff, or visitors at immediate risk of harm and addressing those risks appropriately.
-
-
Document Care
-
Health care staff shall ensure that information related to the health care incident, such as treatment provided and communication with the patient and/or family, is documented appropriately in the health record.
-
For health care incidents involving specific patients, progress note documentation shall include preceding events, observations, examination findings, and assessment of the patient (e.g., vital signs, neurological checks, pain assessment).
-
-
Notification
-
During regular business hours, staff who have identified a health care incident shall immediately notify their direct supervisor.
-
After regular business hours, staff who have identified a health care incident shall immediately notify the unit supervisor or nursing program lead on duty.
-
For all deaths and sentinel events as defined in policy, including medication events with an assigned severity level of 4 through 6, the notified supervisor or nursing program lead shall immediately contact the Chief Executive Officer (CEO), who shall then determine which additional institution staff must be apprised of the situation and within what timeframes.
-
When appropriate, notification to the CEO shall include, at a minimum, the following information:
-
Name of patient, staff, and/or visitor involved.
-
Nature of the health care incident (what occurred).
-
Location of the health care incident.
-
Time of the health care incident.
-
Actions taken, treatment provided, and effects.
-
Current condition of patient, staff, and/or other visitor.
-
Any other pertinent information.
-
-
Institution staff shall document notification of patients as required by state law.
-
The supervisor shall notify the Pharmacy program lead of an adverse drug reaction. The Pharmacy program lead shall complete a United States Food and Drug Administration (FDA) MedWatch Form FDA 3500 if appropriate per FDA instructions.
-
-
Preserve Materials, Supplies, and Other Related Items
-
To ensure that physical materials are readily accessible during the health care incident review process and remain in the condition applicable at the time of the health care incident, staff shall collect and secure samples of physical items involved in the event, which may require examination by qualified personnel to safely handle and store any controlled substances or potentially hazardous materials. Examples of collectable physical items may include, but are not limited to:
-
Medical devices and equipment
-
Retained foreign objects
-
Medications, containers, package labels, or inserts
-
Intravenous bags and tubing
-
Syringes
-
Supply containers and packaging
-
Laboratory and pathology specimens
-
-
Tampering with, cleaning, or otherwise modifying any physical items could result in inaccurate review findings and is prohibited. Health care staff shall work with custody staff to obtain necessary camera equipment and take pictures when appropriate.
-
Health care staff shall work with information technology staff to preserve all electronic data affiliated with the health care incident including mechanisms to back up or otherwise store data. Health care staff shall obtain paper copies of electronic data if there is a risk that the information may be overwritten or lost.
-
-
Provide Relief and Support to Caregivers
-
The unit supervisor or nursing program lead managing the area where the health care incident occurred and the appropriate clinical program leads shall immediately evaluate the impact of the health care incident on all involved staff and shall provide support to health care staff as appropriate including addressing staffing and redistributing patient loads to allow caregivers time to cope with the situation.
-
Caregivers should be assured that any review of the health care incident shall be focused primarily on process and system breakdowns.
-
-
-
Deaths
-
Health care incidents that result in deaths shall receive a separate mortality review pursuant to the Health Care Department Operations Manual, Section 1.2.10, Mortality Review and Reporting and the Mental Health Services Delivery System Program Guide, Chapter 10, Suicide Prevention and Response, which covers a different scope than the RCA process.
-
Findings from the mortality review process may result in the assignment of an RCA.
-
-
Root Cause Analysis
-
Assigning an RCA and Convening the RCA Team
-
The CEO shall determine the scope and membership of the RCA Team.
-
As soon as possible, and no later than 24 business hours after an RCA is assigned by the Statewide Health Care Incident Review Committee (HCIRC), the CEO shall convene a multidisciplinary team to conduct the RCA. This team shall be responsible for identifying and analyzing the primary system or process lapses that contributed to the sentinel event and developing a detailed Plan of Action to prevent similar events from occurring in the future.
-
The respective Regional Health Care Support Team shall provide oversight of the local RCA process and conduct preliminary review of the RCA Report.
-
An institution may elect to initiate/self-assign an RCA if the leadership team deems it appropriate to inform continuous improvement of the local health care delivery system.
-
Self-assigned RCAs are not required to be reported to the HCIRC, but if requested, the Regional Health Care Support Team or HCIRC shall provide feedback and recommendations as appropriate.
-
In the event that a health care incident leading to a self-assigned RCA is identified as a sentinel event and formally assigned an RCA by the HCIRC, the institution shall meet all RCA requirements described in this procedure.
-
-
-
Understanding the Context of the RCA
-
Prior to beginning the RCA process, the RCA Team shall review the CCHCS Performance Improvement Culture Statement (Appendix 1) to ensure that all members understand the context of the RCA process and that the primary emphasis of the RCA is focused on system lapses, not the behavior of individual staff.
-
-
Completion of the RCA and Interim Reports
-
The RCA Team shall adhere to reporting and timeframe requirements in the CCHCS RCA Tool Kit, which is found on Lifeline.
-
During the RCA process and pending a final report, the institution shall implement any immediate and concurrent improvements as determined by the RCA Team to be appropriate.
-
The Regional Health Care Support Team may request concurrent documentation from all RCA Team meetings as interim reports of institution activities.
-
-
The Submission of the RCA Report and Implementation of the Plan of Action
-
The RCA Report, including review and approval by the CEO and Regional Health Care Executive, shall be completed and submitted to the HCIRC within 45 business days from the date that the RCA was assigned.
-
At a minimum, the RCA Report shall contain required elements per the CCHCS RCA Tool Kit.
-
Unless otherwise instructed by the HCIRC, the institution shall begin implementation of the Plan of Action as soon as practicable, but no later than upon submission of the RCA Report to headquarters.
-
-
Revisions to the RCA Report
-
If upon review of the RCA Report, the HCIRC requests clarification or revision of the report, the institution shall make necessary clarifications or revisions and submit the revised report to the HCIRC within 15 business days of the request.
-
-
Post-Submission Plan of Action Status Updates
-
The institution shall submit a monthly Plan of Action status update by the last day of the reporting month for a minimum of four consecutive months following submission of the RCA Report, or until the HCIRC closes the case.
-
-
-
Confidentiality
-
Protected Proceedings and Records
-
It is critical that the proceedings and records of the health care incident review process be maintained as confidential and not be made available to unauthorized persons or organizations.
-
All staff participating in the health care incident review process discussed in this procedure shall adhere to these provisions regarding confidentiality.
-
The records of the committees and staff responsible for the evaluation and improvement of the quality of patient care shall be maintained as confidential and protected from discovery to the extent permitted by law.
-
-
-
Appendices
-
Appendix 1: Performance Improvement Culture Statement
-
-
References
-
California Health and Safety Code, Division 2, Chapter 2, Article 1, Section 1250
-
California Health and Safety Code, Division 2, Chapter 2, Article 3, Sections 1279, 1279.1, and 1279.2
-
California Department of Corrections and Rehabilitation, Department Operations Manual, Chapter 3, Article 2, Health and Safety Program
-
California Department of Corrections and Rehabilitation, Department Operations Manual, Chapter 5, Article 11, Section 51110.11, Written Reports
-
California Department of Corrections and Rehabilitation, Mental Health Services Delivery System Program Guide, Chapter 10, Suicide Prevention and Response
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.6, Statewide Patient Safety Program
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.10, Mortality Review and Reporting
-
Health Care Department Operations Manual, Chapter 3, Article 5, Section 3.5.11, Pharmacy Quality Assurance Program
-
Food and Drug Administration, MedWatch: The FDA Safety Information and Adverse Event Reporting Program (http://www.fda.gov/safety/medwatch/default.htm)
-
The Joint Commission https://www.jointcommission.org/topics/patient_safety.aspx
-
National Commission on Correctional Health Care 2008 Standards for Health Services in Prisons
-
National Coordinating Council for Medication Error Reporting and Prevention
-
United States Department of Veterans Affairs – Veterans Affairs National Center for Patient Safety (http://www.patientsafety.va.gov/)
-
Veterans Health Administration Vision 2020
-
California Department of Public Health Center for Health Care Quality (https://www.cdph.ca.gov/Programs/CHCQ/Pages/CHCQHome.aspx)
-
-
Revision History
-
Effective: 11/2012
Revised: 11/2018
-
-
Appendix 1
-
Performance Improvement Culture Statement
-
Patient safety is the fundamental responsibility of every person in the correctional health care delivery system. California Correctional Health Care Services (CCHCS) actively cultivates a culture of continuous learning and improvement where all staff focus on making health care delivery processes and outcomes as safe and effective as possible; and develop and implement systems that support sustainable, high-quality performance. At CCHCS, leadership teams foster a culture of trust, which enables staff to report problems and encourages all staff to be actively involved in process improvement.
-
CCHCS recognizes that:
-
Human error is inevitable and we continually strive to improve systems to prevent errors.
-
Most health care incidents involve process or system breakdowns that must be addressed before performance can reliably improve.
-
A punitive environment does not fully take into account systems issues, nor does a blame-free environment hold individuals appropriately accountable.
-
A culture of continuous learning and improvement recognizes that people can make mistakes; acknowledges that even competent people can develop erroneous patterns of behavior, yet has zero tolerance for reckless behavior, blameworthy acts, and delayed reporting of health care incidents.
-
To identify opportunities for improvement, CCHCS staff at all reporting levels must be able to report health care incidents without being subject to unjust punitive investigation and penalties.
-
CCHCS staff will:
-
Report health care incidents within timeframes described in related policies and procedures.
-
Encourage and support reporting and review of all health care incidents.
-
Critically analyze processes and design improvements to ensure a safe health care environment.
-
Promote collaboration across ranks and disciplines to find sustainable solutions to patient safety issues.
-
Respond quickly and reasonably to actions, decisions, and behaviors that may result in unsafe acts, realizing that most actions, decisions, and behaviors do not warrant disciplinary action. The most severe penalties, such as demotion, reduction in pay, suspension with or without pay, and termination, are reserved for reckless behavior and blameworthy acts and, as warranted, delayed reporting.
-
Reckless Behavior and Blameworthy Acts:
-
CCHCS maintains a code of conduct for acceptable behavior, and behaviors that undermine patient safety. Although performance improvement processes will primarily target the identification and resolution of process breakdowns, reckless behavior and blameworthy acts discovered in this context will be appropriately addressed to ensure patient and staff safety. Reckless behavior includes situations in which an individual takes a substantial and unjustifiable risk that may result in patient harm. A blameworthy patient care act possesses one of the following three characteristics: it involves a criminal act, a purposefully unsafe act, or events involving patient abuse of any kind. Reckless behavior, a blameworthy act, intentionally withholding information, or providing misleading or false information may result in adverse action in accordance with the Disciplinary Matrix.
-
1.2.10 Mortality Review and Reporting
-
Policy
-
California Correctional Health Care Services (CCHCS) shall complete an independent review of every death of individuals in the custody of the California Department of Corrections and Rehabilitation (CDCR), in an effort to promote a safe, high quality health care system.
-
-
Applicability
-
This policy applies to all deaths occurring while in the custody of CDCR. In addition to the review conducted under this section, each death classified as a suicide or suspected suicide shall also receive a Suicide Case Review, pursuant to the current Mental Health Services Delivery System (MHSDS) Program Guide.
-
-
Purpose
-
To utilize mortality data to mitigate patient harm within the department, to identify opportunities for improvement (OFI) related to patient safety, quality of health care services, patient outcomes, and to fulfill mandated reporting requirements.
-
-
Responsibility
-
Statewide
-
CDCR and CCHCS leadership, at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial, and clinical systems are in place, and appropriate resources are available to ensure the mortality review and reporting process is maintained.
-
Headquarters (HQ) and regional executive representatives shall ensure progress is made on current mortality and morbidity initiatives and communicate new priorities to improve health care processes and patient outcomes and quality of services delivered.
-
Designated Medical Services Division (MSD) support staff shall comply with all federal and state reporting requirements. MSD support staff shall enter mortality review findings into the electronic Health Care Incident Reporting (eHCIR) system and produce daily and weekly mortality reports and any required reports.
-
-
Regional
-
Regional Health Care Executives have overall responsibility for ensuring implementation of this policy at the subset of institutions within an assigned region.
-
-
Institutional
-
The Chief Executive Officer (CEO) has overall responsibility for ensuring implementation of this policy at the assigned institution.
-
The institution is responsible for patient safety oversight as described in the Health Care Department Operations Manual (HCDOM), Section 1.2.7, Institution Patient Safety Program.
-
-
-
Procedure
-
Daily Mortality Reporting from Institutions to Headquarters
-
All in-custody deaths shall be entered in the health record on the Initial Inmate Death Record no later than 1200 hours on the next business day following the patient’s death. For a suicide, an Initial Inmate Suicide Report shall also be completed, pursuant to the current MHSDS Program Guide.
-
Deaths occurring in the following locations shall be reported:
-
An institution
-
A fire camp
-
A contracted facility
-
In a county facility while out-to court
-
An outside hospital or other medical facility setting
-
A skilled nursing facility or other long-term care facility
-
A re-entry facility, if not paroled
-
In transit
-
-
Deaths occurring in the following locations and populations are not required to be reported:
-
Sacramento Control Office
-
Western Interstate Corrections Compact
-
Parole
-
County incarcerated persons being housed with CDCR
-
-
-
Collecting Clinical Records for Review
-
Institution
-
Within five calendar days of the death, institution health records staff shall complete the scanning of any paper documents into the health record, including outside community documents related to emergency medical response and hospitalization.
-
-
Medical Services Division
-
Within five calendar days of the death, MSD support staff shall:
-
Contact the county coroner’s office to determine whether an autopsy will be conducted. If an autopsy is conducted, MSD support staff shall request the report from the county coroner and distribute the final report, when available to the institution. Final reports shall be distributed to the institution, HQ Health Information Management (HIM), and the Mental Health Program within seven calendar days of receipt. Reports shall be scanned into the health record by HQ HIM staff upon receipt.
-
Obtain the CDCR 837-A, B, and C, Crime/Incident Reports, from the Strategic Oversight Management System or request the reports from custody representatives at the institution where the death occurred.
-
Request CDCR 602 HC, Health Care Grievances, and responses for the six months prior to the death.
-
-
If the requested documents above are not received within five calendar days, MSD support staff shall notify the appropriate health care or custody staff to request assistance acquiring the documents.
-
-
-
Institution Mortality Review
-
Within 30 calendar days of the death, the institution CEO, or designee(s), shall complete a multidisciplinary review of the significant events leading to the patient’s death.
-
The review shall focus on identifying OFI at the institution and be submitted to MSD support staff at mortalityreview@cdcr.ca.gov for inclusion in the final mortality review at the discretion of the HQ mortality reviewers.
-
-
Headquarters Mortality Case Review
-
Each death shall be assigned to a nurse and provider reviewer.
-
The nurse reviewer shall be a Nurse Consultant Program Review Registered Nurse, assigned by the Deputy Director (DD), Nursing Services, or designee.
-
The provider reviewer shall be a board-certified physician or advanced practice provider assigned by the DD, Medical Services, or designee.
-
-
Initial training for all mortality case reviewers shall occur prior to starting mortality review responsibilities. Training shall be in accordance with the most current community standards for mortality review. Ongoing training shall be provided on an as-needed basis and periodically throughout the calendar year.
-
The nurse and provider reviewers shall complete the mortality review using the most current electronic form or data entry program.
-
The provider reviewer and the nurse reviewer shall coordinate in the reviewing of the deceased patient’s clinical records, which are relevant to the history of the patient’s cause of death, and use the information to complete the executive summary. This will include, at a minimum, records up to six months prior to the death; however, reviewers may include older records if necessary or relevant to determine the trajectory of the terminal event. Relevant records may include, but are not limited to:
-
Initial Inmate Death Record and Initial Inmate Suicide Report, if applicable.
-
Progress notes, diagnostic results, and other clinical information.
-
Records from an outside hospital or other medical facility.
-
CDCR 837-A, B, and C, Crime/Incident Reports.
-
Emergency response records.
-
Autopsy reports.
-
CDCR 602 HCs and subsequent responses.
-
Institution Multidisciplinary Mortality review.
-
-
The nurse reviewer shall document any findings related to the cause of death and any other possibly actionable findings that are discovered during the review and include relevant OFI received from the institution multidisciplinary review.
-
The provider reviewer shall add or modify the nurse review findings and OFI as needed in collaboration with the nurse reviewer.
-
If at any time during the review process, either reviewer identifies circumstances or processes that may represent an immediate patient safety issue, the nurse or the provider reviewer shall submit an eHCIR pursuant to the HCDOM, Section 1.2.6, Statewide Patient Safety Program, and the HCDOM, Section 1.2.7, Institution Patient Safety Program.
-
When a cause of death is unknown, including in potential drug overdose cases, reviewers shall work in collaboration with mental health case reviewers who are assigned to conduct reviews pursuant to the current MHSDS Program Guide to:
-
Collect and share information.
-
Determine if the cause of death could have been the result of suicide.
-
For suicides or suspected suicides, ensure timely sharing of information and findings.
-
Final mortality reports for suicides and final suicide reports shall be distributed to both disciplines.
-
-
For each death classified as a homicide or suspected homicide, if clarification is needed regarding custodial or security elements, a Division of Adult Institutions (DAI) staff shall be consulted.
-
The preliminary mortality reports shall be completed by reviewers within 60 calendar days of the death, absent a showing of good cause for an extension, in which case an extension may be granted by the DD, Medical Services, or designee.
-
-
Regional and Institution Review
-
The preliminary mortality reports shall be submitted electronically to health care leadership for review. Health care leadership staff are assigned headquarters Chief Nurse Executives and headquarter and regional physician managers who generally are supervisors for the provider reviewers.
-
Health care leadership shall review the report and accept or reject the mortality report within 14 calendar days of receipt.
-
If rejected, health care leadership shall facilitate any edits and necessary clarification with the nurse and provider reviewers.
-
-
The mortality review is considered final once health care leadership has accepted the report.
-
A final mortality report may be amended if new information that is material and relevant to the mortality review is obtained subsequent to the report being finalized. The amended report shall be submitted to health care leadership seven calendar days prior to redistributing to the stakeholders listed in Section (e)(6)(A). A response is not required from health care leadership unless the amended report is disputed.
-
-
-
Mortality Report Distribution
-
Finalized original and amended mortality reports shall be distributed to the following stakeholders:
-
The relevant institution and regional executives responsible for the patient’s health care prior to death. If multiple institutions were involved with the health care of the patient during the period of review and there are findings from that time period, a report shall be submitted to the leadership of each institution and region that were involved.
-
The Health Care Incident Reporting Committee and Statewide Patient Safety Program, uploaded via the eHCIR system. If appropriate, a recommendation for review by the Health Care Reporting Committee or Statewide Patient Safety Program shall be included.
-
DAI and institution Warden, if related to a drug overdose, suicide, homicide, or other deaths where a potential custody-related opportunity for improvement was identified.
-
-
-
Public Reporting
-
Upon the death of any person in departmental custody, MSD support staff shall provide the information identified in Section (e)(7)(B) below to the CCHCS Communications Office via the ServiceNow portal to facilitate posting on CCHCS’s public website.
-
Except as provided in Section (e)(7)(C) below, within 10 days of the death, the following information shall be posted on CCHCS’s public website:
-
The full name of the agency with custodial responsibility at the time of death.
-
The county in which the death occurred.
-
The facility in which the death occurred, and the location within that facility where the death occurred.
-
The race, gender, and age of the decedent.
-
The date on which the death occurred.
-
The custodial status of the decedent including, but not limited to, whether the person was awaiting arraignment, awaiting trial, or incarcerated.
-
The manner and means of death.
-
-
If CDCR seeks to notify the next of kin pursuant to the CDCR Department Operations Manual, Section 51070.10, Notification of Contact Listed and is unable to do so within 10 days of death, CDCR shall be given an additional 10 days to make good faith efforts to notify the next of kin before the information is publicly posted.
-
CCHCS shall update any publicly posted information within 30 days of a change of information.
-
-
Conflict of Interest
-
A provider, nurse, or health care leader shall:
-
Not participate in any decision under the breach of professional clinical peer review process if the individual has a personal conflict of interest (COI) or has provided direct patient care to the decedent.
-
Disclose any potential and actual COI prior to participating in decision-making.
-
-
A personal COI is a professional, financial, or other obligation or interest that is likely to limit the reviewer’s ability to participate impartially in decision-making.
-
-
Confidentiality
-
It is critical that the records of the mortality review process be maintained as confidential and not be made available to unauthorized persons or organizations.
-
All staff participating in the mortality review process discussed in this procedure shall adhere to these provisions regarding confidentiality.
-
The mortality review process is intended for improvements in the quality of patient care and shall be maintained as confidential and protected from discovery to the extent permitted by law.
-
-
-
References
-
Federal Death in Custody Reporting Act of 2000 (Public Law 106-297)
-
California Government Code, Title 2, Division 3, Part 2, Chapter 6, Article 2, Section 12525
-
California Penal Code, Part 3, Title 7, Chapter 1, Section 5021
-
California Penal Code, Part 3, Title 10, Section 10008
-
National Commission on Correctional Health Care, Standards for Health Services in Prisons (2016)
-
National Commission on Correctional Health Care, Standard P-A-10, Procedure in the Event of an Inmate Death
-
California Department of Corrections and Rehabilitation, Department Operations Manual, Article 7, Sections 51070.1 through 51070.20
-
California Department of Corrections and Rehabilitation, Mental Health Services Delivery System Program Guide, Chapter 10, Suicide Prevention and Response, Section E, Suicide Reporting, and Section F, Suicide Death Review
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.1, Complete Care Oversight Team Committee
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.3, Quality Management Program Overview
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.4, Quality Management Program, Statewide Governance
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.5, Quality Management Program, Institution
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.6, Statewide Patient Safety Program
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.7, Institution Patient Safety Program
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.15, Utilization Management Program
-
California Correctional Health Care Services Annual Inmate Mortality Reviews (2006-2022)
-
-
Revision History
-
Effective: 08/2008
Revised: 10/06/2025
-
1.2.11 CCHCS Systemwide Pharmacy and Therapeutics Committee
-
Procedure Overview
-
The California Correctional Health Care Services (CCHCS) Systemwide Pharmacy and Therapeutics (P&T) Committee shall:
-
Provide overall direction to Pharmacy Services at all California Department of Corrections and Rehabilitation (CDCR) institutions.
-
Report to the statewide Quality Management Committee (QMC).
-
Oversee subcommittees including, but not limited to, the Systemwide Medication Management Subcommittee and Drug Utilization Review Subcommittee.
-
-
Institutions shall have a Medication Management Subcommittee that reports to the local QMC to oversee medication management practices including local medication storage, distribution, administration and utilization locally; however, changes to approved, standardized pharmacy policies and procedures, the standard practice model, and the CCHCS Drug Formulary must be approved by the Systemwide P&T Committee. Recommended changes to policy, procedure, or the formulary shall be submitted to the Regional Deputy Medical Executive (DME) and Statewide Chief of Pharmacy Services for consideration by the Systemwide P&T Committee as appropriate.
-
-
Purpose
-
To ensure the safe, rational, evidenced-based, cost-effective, standardized use of therapeutic drugs and develop policies and procedures related to medication management within the CDCR.
-
-
Procedure
-
Membership
-
The chairperson is appointed by the Deputy Director, Medical Services, for a two-year term (eligible for reappointment) and shall be responsible for the Systemwide P&T Committee meeting.
-
The chairperson shall assist the Deputy Director, Medical Services, in selecting the other members of the Systemwide P&T Committee and determining terms of service.
-
Voting members shall include the following, or the members shall assign a designee with decision-making authority similar to the member that is unable to attend:
-
Deputy Director, Medical Services or designee at the executive level
-
Headquarters Deputy Medical Executive chairperson
-
Two Headquarters Physicians at the executive or management level
-
Four Institutional or Regional Physicians at the executive or management level
-
Deputy Director, Nursing Services or designee
-
One Headquarters Chief Nurse Executive
-
Deputy Director, Mental Health Services (if psychiatrist) or designated psychiatrist at the executive or management level
-
Statewide Chief Psychiatrist or designee
-
Statewide Chief Telepsychiatrist or designee
-
One Psychiatric Inpatient Program Chief Psychiatrist or designee
-
Deputy Director, Dental Services or designee
-
Statewide Chief, Pharmacy Services or designee
-
Two Pharmacy Services Managers
-
One Deputy Medical Executive, Quality Management or designee
-
-
The Systemwide P&T Committee shall maintain a list of permanent designees for voting members. Designees for voting members of the Systemwide P&T Committee are permitted. Prior notification to the chairperson at least three working days in advance is requested when a designee shall attend for a voting member. All designees must sign a confidentiality statement pursuant to Section (c)(3)(D).
-
Non-voting members shall include:
-
Deputy Director, Fiscal Services, or designee, at the Assistant Deputy Director level
-
An attorney from the CCHCS Office of Legal Affairs (COLA), to be designated by COLA’s Director and Chief Counsel
-
Pharmaceutical Program Manager and Pharmaceutical Consultant II, Department of General Services
-
Support staff
-
-
Guests for Systemwide P&T Committee meetings are permitted. Prior notification to the chairperson at least three working days in advance is requested. All guests must sign conflict of interest and confidentiality forms pursuant to Section (c)(3)(D) and understand that their attendance is as an observer and comments during the meeting are at the discretion of the chairperson.
-
-
Duties
-
The Systemwide P&T Committee is responsible for overseeing policies and procedures related to all aspects of medication use within CCHCS including, but not limited to:
-
Maintaining a formulary of medications, including standardizing the strengths and dosage forms for medications used across institutions.
-
Reviewing procurement and medication selection processes to promote cost-effective formulary management.
-
Conducting regular therapeutic category reviews for formulary selection.
-
Ensuring that pharmacy services address the health care and security needs of the institution.
-
Reviewing and monitoring medication usage and therapeutic use of medications within the CDCR.
-
Evaluating medication use and promoting safe medication practices.
-
Evaluating medication therapies and providing input to the development of disease management guidelines.
-
Reviewing and approving proposed changes to pharmacy policies. The Systemwide Medication Management Subcommittee shall be consulted for policymaking on any matters related to medication management processes.
-
-
Meetings
-
The Systemwide P&T Committee shall meet at least quarterly or as often as necessary to carry out its responsibilities.
-
Meetings via teleconference shall be made available to members of the Systemwide P&T Committee.
-
A record of the proceedings shall be kept which records committee activities, recommendations, and attendance.
-
Confidentiality/Non-Conflict
-
The proceedings and records of the Systemwide P&T Committee shall be kept confidential and protected from discovery to the extent permitted by law.
-
Any attendees shall provide a completed copy of a Conflict of Interest Form and a Confidentiality and Non-Disclosure Agreement. Completed copies of each document shall be maintained with the Systemwide P&T Committee records. Members may not have any financial or business relationships with entities doing business with the State of California.
-
If any attendee has an interest that may affect or be perceived to affect the member’s independence of judgment, the member shall recuse themself from the voting process for the drug class or other agenda items concerned. This recusal includes, but is not limited to, refraining from deliberation or debate, making recommendations, volunteering advice, and participating in the decision-making process in any way.
-
-
Voting
-
Each voting member shall have one vote. A quorum is designated as 50 percent of voting members, excluding vacancies and abstentions.
-
Action on a motion requires the total number of votes to be equal to or greater than a quorum.
-
The Systemwide P&T Committee may use electronic voting to address issues when it is determined that waiting until the next scheduled meeting is suboptimal. Electronic voting may be used to resolve an existing agenda item or to address an urgent or emergent new agenda item.
-
An abstention is considered a non-vote, and thus does not accrue to the total number of votes.
-
-
-
Subcommittees
-
The Systemwide P&T Committee may charter standing subcommittees and establish ad hoc workgroups to plan and develop new or modify existing programs. The charter for each subcommittee shall be reviewed and updated as often as needed but no less than every two years. Standing subcommittees include, but are not limited to:
-
Systemwide Medication Management Subcommittee
-
The Systemwide Medication Management Subcommittee is a multidisciplinary group that works to ensure medication practices support the safety of the individuals served and improve the quality of care by developing and modifying processes as they relate to planning, procurement, ordering, preparing and dispensing, distribution, storage, and administering medications with the goal to reduce any potential harm that could be caused by medications.
-
-
Drug Utilization Review Subcommittee
-
The Drug Utilization Review Subcommittee shall:
-
Review prescription drug utilization and costs and identify clinical criteria, decision support, and methods for managing prescription drug cost and optimizing prescribing practices.
-
Develop and provide evidence-based recommendations to the Systemwide P&T Committee with respect to formulary selection, electronic health record system (EHRS) medication catalog availability, and use criteria or prior authorization criteria, if needed.
-
-
340B Oversight Subcommittee
-
The 340B Oversight Subcommittee assists in monitoring, directing, and overseeing the CCHCS’ implementation of its 340B Program. The 340B Oversight Subcommittee’s reporting provides direction and ongoing surveillance of the organization’s 340B Program for compliance with policy and federal rules.
-
-
Nutrition Subcommittee
-
The Nutrition Subcommittee shall meet on an ad hoc basis when required to develop and provide evidence-based recommendations to the Systemwide P&T Committee with respect to enteral and parenteral nutrition and nutritional supplements that are deemed necessary to make available in the CCHCS Drug Formulary or make available in the EHRS medication catalog as a non-formulary item.
-
-
-
-
References
-
California Civil Code, Division 1, Part 2.6, Chapter 1, Section 56 et seq.
-
California Evidence Code, Division 9, Chapter 3, Section 1157
-
California Code of Regulations, Title 15, Division 3, Chapter 1, Subchapter 5, Article 2, Section 3413 (b)
-
California Code of Regulations, Title 22, Division 5, Chapter 3, Article 5, Section 72525, Required Committees
-
California Code of Regulations, Title 22, Division 5, Chapter 12, Article 5, Section 79781, Required Committees
-
California Government Code, Title 2, Division 5, Part 2.6, Chapter 2.5, Article 10, Section 19990
-
California Government Code, Title 9, Chapter 7, Article 1, Section 87100 et seq.
-
California Penal Code, Section 5024.2
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.4, Quality Management Program, Statewide Governance
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.12, Outpatient Dietary Intervention
-
Health Care Department Operations Manual, Chapter 3, Article 5, Section 3.5.5, CCHCS Drug Formulary
-
Health Care Department Operations Manual, Chapter 3, Article 5, Section 3.5.10, CCHCS Pharmacy Policy and Procedure Manual Review, Revisions, and Additions
-
-
Revision History
-
Effective: 03/2007
Revised: 10/22/2025
-
1.2.14 Medical Classification System
-
Policy
-
California Correctional Health Care Services (CCHCS) and California Department of Corrections and Rehabilitation (CDCR) shall utilize a Medical Classification System (MCS) to serve as the system for considering medical factors in making patient placement decisions. The MCS shall be used to match patients’ medical needs with the capabilities of facilities and programs. The MCS is intended to:
-
Ensure all patients are assigned Medical Classification Factors that allow matching of medical needs to institutions/facilities to support efficient bed management.
-
Eliminate redundant and unnecessary forms, screenings, and evaluations.
-
Reduce or prevent inefficiencies caused by disparity between patient medical needs and facility capabilities and resources.
-
Provide department-wide capacity to profile the medical needs of the patient population.
-
-
Purpose
-
To provide guidance to clinical and custody staff in considering medical factors in making patient placement decisions.
-
-
Responsibility
-
The Chief Executive Officer, or designee, is responsible for the implementation, monitoring, and evaluation of this policy and its corresponding procedures.
-
The Health Care Placement Oversight Program (HCPOP) at headquarters is responsible for various population management functions. HCPOP is responsible for maintaining a unit procedure, as well as for maintaining and issuing medical classification matrices and Decision Support Material.
-
-
Procedure
-
Medical Classification Chrono General Process
-
Medical Classification Chrono (MCC)
-
Physical Description: The MCC is generated via the electronic MCC database. It is viewable in the Strategic Offender Management System (SOMS).
-
Distribution: A copy of the MCC shall be printed for the patient’s records; the transmission must ensure confidentiality.
-
Decision Support Material: Decision Support Material shall be updated as necessary under the authority of the Chief Medical Executive (CME) and the Chief Nurse Executive. Issues, questions, and suggestions regarding the materials shall be routed to the HCPOP. CCHCS and CDCR shall issue Decision Support Material that includes reference material regarding:
-
Criteria for medical risk determination
-
Criteria for nursing acuity determination
-
Pregnancy Program
-
Transplant Program
-
Hemodialysis Program
-
Therapeutic Diet Policy
-
CDCR 7410, Comprehensive Accommodation Chrono
-
CDCR 1845, Disability Placement Program Verification
-
-
-
Completing the MCC in the Reception Center (RC)
-
An MCC is completed at the time of the RC physical examination. If the Medical Classification Factors (Refer to Appendix 1, The Medical Classification Factors) change during the RC stay, a new MCC must be issued.
-
There may be situations where further clinical data are needed in order to make a clear determination of the appropriate Medical Classification Factors. In this case, the MCC is completed based on medical judgment. For example, the patient has cirrhosis, but the degree of stability and severity has not been assessed because laboratory studies are pending.
-
-
Relationship to CDCR 1845 and CDCR 7410
-
The CDCR 1845 provides details regarding disabilities that impact placement and assistive devices such as wheelchairs, walkers, and hearing aids.
-
The CDCR 7410 provides details regarding other patient needs such as lower bunks and bottom tiers. Many of these needs do not impact placement.
-
Specified Medical Classification Factors, if present, require a matching CDCR 1845 or CDCR 7410 in order to provide supporting details. These factors are marked with a superscript “1.”
-
-
Issuing an MCC
-
The MCC is normally completed by a Primary Care Provider (PCP) (Physician, Nurse Practitioner, or Physician Assistant). Chief Physicians & Surgeons (CP&S) and CMEs may also complete the MCC.
-
The PCP may need to seek input from nursing staff in order to accurately determine the Nursing Care Acuity (Refer to Appendix 1). Nursing staff is responsible for reporting changes in patient status to the PCP that result in a change in Medical Classification Factors.
-
In the case of an outside hospital admission or discharge, the CP&S or designee is responsible for completing the new MCC.
-
Overrides may only be requested through HCPOP. This is normally initiated by the institution Classification and Parole Representative (C&PR) or RC Correctional Counselor III (CC-III). Only a Regional Deputy Medical Executive (RDME) or designee may issue an Override MCC. (Refer to Section (d)(3)(F)3).
-
-
Triggers for Change in Classification
-
A new MCC is issued whenever the patient’s medical condition changes in a way that changes Medical Classification Factors (Level of Care, Classification Factors, Intensity of Services, Specialized Services, and/or Institutional-Environmental). Whenever a patient’s need for a medical level of care changes, either to higher or lower level, a new MCC shall be issued. Examples include admission to or discharge from a Correctional Treatment Center (CTC), placement into or return from an outside hospital stay, or a new requirement for medium-intensity nursing.
-
The MCC must be completed promptly on admission and again on discharge from an outside hospital stay in order to show the placement into a new level of care. This placement into a new level of care may require a telephone order from the on-call provider.
-
-
Temporary and Permanent Chronos
-
Permanent Chronos: A Permanent Chrono indicates that the Medical Classification Factors are not expected to change in the next six months. Automatic or periodic reclassification is not needed if the MCC is a Permanent Chrono.
-
Temporary Chronos: A Temporary Chrono indicates that the Medical Classification Factors are expected to change within six months or that an automatic requirement for Medical Reclassification is needed within six months.
-
Defined Expiration Date: An MCC with a defined expiration date will cease to be valid on that date. On the defined expiration date, the patient will no longer have a valid MCC, which will require issuance of a new MCC. A temporary MCC is used, for example, when a patient is pregnant and qualifies for the pregnancy program. The expiration date would be set by the estimated date of confinement or estimated delivery date.
-
Undefined Expiration Date: An MCC with an undefined expiration date is used, for example, when a patient is admitted to an acute care hospital for an acute disease whose course is not certain. The “temporary” designation indicates that the need is expected to be time-limited; however, the duration of the need is unpredictable. The MCC must be reviewed and re-issued within six months.
-
-
-
-
Medical Classification Matrices
-
Medical Classification Planning Matrix
-
The Medical Classification Planning Matrix (MCPM) shows the Specialized Medical Bed (SMB) and medical program capacities and census at each CDCR institution. The MCPM allows CDCR to review and plan for an efficient match of Institution Attributes to the demand across the various custody levels and programs. Where custody program is important (e.g., Security Housing Unit, Special Needs Yard, Minimum Support Facility [MSF]), the MCPM includes information for that program.
-
The MCPM is maintained by HCPOP.
-
-
Medical Classification Matrix
-
The Medical Classification Matrix (MCM) is a tool that supports the task of matching a patient’s Medical Classification Factors with the available facility’s medical capabilities.
-
-
Maintaining and Issuing the Matrix
-
The MCM is maintained by HCPOP. As the availability of beds, programs, and other capacities change, HCPOP staff updates the MCM. The MCM is available via SOMS.
-
-
-
Placement Authority
-
Routine Placements
-
Routine placements for patients classified with Outpatient level of care are done using the CDCR endorsement process. The MCM and the MCC provide inputs to the endorsement process.
-
Specialized Outpatient (SOP) endorsements to California Health Care Facility, Stockton (CHCF) shall be made by HCPOP. SOPs who are not endorsed to CHCF shall be endorsed to an intermediate institution. All SOP transfer endorsements require transfer referral from a classification committee prior to endorsement. When necessary, HCPOP is authorized to endorse SOP patients to a basic institution to accommodate required case factors, such as Enhanced Outpatient Program, when there are no available intermediate institutions commensurate with the patient’s case factors.
-
All SMB endorsements shall be made by HCPOP. At HCPOP’s request, patients with an SMB level of care shall be taken to committee prior to endorsement.
-
-
Medical Classification Factor Priorities
-
Decision support for using the MCM includes a tool that shows which Medical Classification Factors represent absolute requirements and which represent preferences (Refer to Appendix 2, Medical Classification Factor Priorities). For example, “Requires Electrical Access” is an absolute factor: a patient on life-support equipment that requires electricity must be in housing with electricity available. “Proximity to Tertiary Consultations” is a preference factor: a patient who needs a retinal specialist is best managed where the specialist is available nearby, but the patient can be managed at any institution as long as transportation to a very distant specialist can be accomplished.
-
The Medical Classification Factors listed as preferences on Appendix 2 can be overridden by a Classification Staff Representative (CSR) or a CC-III with endorsement authority as part of matching overall patient medical needs with facility capabilities in a particular institution. If a CSR approves a placement using a Medical Classification Factor listed as a preference on Appendix 2, the CSR approval noted on the CDC 128-G, Chrono Classification (Regular), must include the reason for using the flexibility provided by the preference.
-
The Medical Classification Factors marked as absolute can only be overridden by a new MCC marked as “Override” and completed by the responsible RDME (Refer to Section (d)(3)(F)3) at the request of the institution CME.
-
-
Medical Placement Based on Level of Care Medical Classification Factors
-
Placement of a patient into and from the level of care factors (Refer to Appendix 1) is done by medical staff without classification action as part of the patient care routine. Placement based on level of care factors does not require endorsement action. CDCR will be notified of a change in level of care via the MCC.
-
If the institution does not have the needed level of care but has a higher level of care, the patient is normally placed into the higher level of care. Mismatches in actual level of care compared with needed level of care shall be referred to HCPOP for population management.
-
Returns from a higher level of care outside CDCR institutions and facilities that require a level of care not available at the institution shall be noticed to HCPOP for potential medical endorsement.
-
-
Medical Endorsement
-
All patients requiring placement into an SMB as indicated by their medical level of care on the MCC shall be reviewed and endorsed by HCPOP. The patient may be placed immediately into an appropriate health care setting by institution medical staff. HCPOP shall consider permanent endorsement into an appropriate institution SMB within 45 days of the date the MCC is completed.
-
Patient transfers to SMBs do not require classification committee action, as the placement decision is made to provide necessary health care treatment or access; however, at HCPOP’s request, patients with an SMB level of care shall be taken to committee prior to endorsement.
-
-
Decision Support Materia
-
HCPOP shall issue and maintain Decision Support Material that includes:
-
Detailed guidance regarding this policy
-
Medical Classification Factor Priorities (Refer to Appendix 2)
-
Sample chrono
-
-
Processes for Resolving Ambiguity or Difference of Opinion
-
Concerns from CSR
-
Concerns from the CSR shall be resolved by a case conference between the Classification Services Unit, the Population Management Unit, the Class Action Management Unit, and HCPOP. If the issue cannot be resolved, the RDME shall be consulted.
-
-
Resolution of Inappropriate Placement
-
Concerns from nursing staff, custody staff, or providers that a placement is medically inappropriate must be forwarded to the institutional CME. If the CME determines that a medical placement was inappropriate based on the Medical Classification System policy or patient safety considerations, the CME must notify the RDME. The CME must provide appropriate clinical detail.
-
The RDME shall take appropriate action, in cooperation with HCPOP if necessary, to resolve the issue. HCPOP shall be notified of the issue and resolution for tracking and quality improvement purposes.
-
-
Override Process
-
The institution C&PR or RC CC-III shall request guidance from HCPOP and the Classification Services Unit by a case conference if it appears that a placement that meets all classification and medical requirements is not possible. The RDME shall be consulted if the issue cannot be resolved during the case conference. If the RDME determines that an override of one or more Medical Classification Factors is appropriate, the RDME shall issue an MCC with an override.
-
If the RDME cannot resolve the issue, it shall be referred to the Departmental Review Board. All overrides shall be noticed to HCPOP for action and tracking.
-
-
-
Required Training
-
Orientation to the Medical Classification System shall be included in the orientation of new classification staff, PCPs, nursing staff, and health records staff.
-
-
Quality Improvement
-
The institution CME is accountable for the accuracy of MCCs issued by that institution. The institutional Local Operating Procedure must include processes for monitoring the consistency, reliability, accuracy, and variability of the MCCs. The results of monitoring are provided to the institution Quality Management Committee (QMC), which forwards them to the statewide QMC.
-
The institution Classification Committee Chairperson is accountable for the accuracy of placement recommendations by the Classification Committee. The accountability process is the same as used for custody placements.
-
-
Local Operating Procedure
-
Each Chief Executive Officer is responsible for ensuring that the institution has an approved and current Local Operating Procedure that includes, at a minimum:
-
Description of Institution Attributes including the Institution Medical Grouping for the institution (Refer to Appendix 3, Institutional Medical Groupings).
-
Contact information for the C&PR, RC CC-III (if applicable), RDME, the HCPOP contact, and Institution Medical Executive.
-
MCCs including a description of:
-
Who completes the MCC and when.
-
Who is responsible to complete an MCC when a patient changes level of care within a facility (sending provider or receiving provider).
-
When the MCC is completed in the RC and whether laboratory results are required before it can be completed.
-
Detailed process for obtaining a new MCC when a temporary has expired.
-
Interaction between nursing and providers to accurately determine Nursing Care Acuity.
-
Changes in patient condition that prompt nursing to notify the provider for completion of an updated MCC.
-
-
Distribution
-
How the patient copy is handled and how confidentiality is ensured.
-
-
Decision Support Material
-
How Decision Support Material will be distributed, where it will be available, and how obsolete versions will be removed.
-
Local additions to Decision Support Material (if any) and process for approval, distribution, and revision.
-
Process for recommending changes to Decision Support Material.
-
-
Quality Improvement
-
Periodic sampling for accuracy and completeness.
-
Statement that required detail is entered into the “Comments” section.
-
Statement that confidential information is entered into the “Confidential Comments” and not the “Comments” section.
-
Patient movements based on incorrect MCCs that required a re-endorsement of the patient.
-
Patients with more than three changes to their MCC in seven days; results of CME review.
-
-
Local Training Plan
-
Description of who provides local training, staff who receive training, and frequency of training.
-
-
Approval Process
-
Description of the Local Operating Procedure approval process including any local sign-off.
-
Statement that final approval is obtained from the Regional Health Care Executive, acting in concert with the Regional Leadership Team.
-
-
-
-
Unit Procedures
-
Classification Services Unit
-
The Classification Services Unit is responsible for maintaining a unit procedure that includes, at a minimum, a description of the following:
-
Training plan for the Classification Services Unit, C&PRs, RC CC-IIIs, and CSRs.
-
Process for elevating suggestions regarding revision to the Medical Classification System.
-
Process and responsibilities for the case conference between the Classification Services Unit and HCPOP.
-
-
Health Care Placement Oversight Program
-
HCPOP is responsible for maintaining a unit procedure that includes, at a minimum, a description of the following:
-
Detailed processes for maintaining the MCPM.
-
Detailed processes for maintaining the MCM.
-
Process for elevating suggestions regarding revision to the Medical Classification System.
-
Detailed process for endorsing a patient to an appropriate institution SMB.
-
Detailed process for approval, distribution, and revision of Decision Support Material.
-
Process and responsibilities for the case conference between the Classification Services Unit and HCPOP.
-
Detailed process for resolution of inappropriate placements in concert with the RDME, including tracking of cause, resolution, and impact on the patient and the system and interface with the QMC.
-
Process for HCPOP to receive notification of overrides from the RDME, including tracking and reporting and interface with the QMC.
-
Training plan.
-
-
-
-
-
Appendices
-
Appendix 1: The Medical Classification Factors
-
Appendix 2: Medical Classifications Factor Priorities
-
Appendix 3: Institutional Medical Groupings
-
-
References
-
Plata v. Newsom, Order Granting Plaintiffs’ Motion for Relief Re: Valley Fever at Pleasant Valley and Avenal State Prisons, June 24, 2013
-
California Code of Regulations, Title 15, Division 3, Chapter 1, Subchapter 4, Article 10, Section 3379, Inmate Transfers
-
California Code of Regulations, Title 22, Division 3, Subdivision 1, Chapter 8, Article 1, Sections 58018, Hospice Services
-
California Code of Regulations, Title 22, Division 5, Chapter 3, Article 1, Section 72103, Skilled Nursing Facility
-
California Code of Regulations, Title 22, Division 5, Chapter 12, Article 1, Section 79516, Correctional Treatment Center
-
California Code of Regulations, Title 22, Division 5, Chapter 12, Article 1, Section 79555, Outpatient Housing Unit
-
California Department of Corrections and Rehabilitation, Department Operations Manual, Chapter 6, Article 12, Sections 62080.1-62080.6
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.9, Health Care Transfer
-
Health Care Department Operations Manual, Chapter 3, Article 6, Section 3.6.2, Comprehensive Accommodation
-
National Fire Protection Association 1582, Standard on Comprehensive Occupational Medical Program for Fire Departments
-
-
Revision History
-
Effective: 11/2009
Revised: 09/2017
-
-
Appendix 1
-
The Medical Classification Factors
-
(a) Level of Care Based on Patient Need
-
This Factor rates the medical setting the patient currently needs; the patient may not actually be housed in that setting. For example, a patient may be currently housed in a Correctional Treatment Center but need only Outpatient Housing Unit level of care. By collecting this Factor, users of the Medical Classification System can take appropriate actions for bed management.
-
Outpatient (OP): No need for a medical setting that provides the patient with daily nursing care.
-
Specialized Outpatient (SOP): A high medical risk outpatient with the potential for clinical deterioration, decompensation, morbidity, or mortality who has long-term care needs. This patient population needs frequent supportive care management, care coordination, nursing education, nursing interventions, and may need specialized nursing care. Endorsements shall be made by the Health Care Placement Oversight Program (HCPOP). All SOP transfer endorsements require a classification committee referral to HCPOP.
-
Outpatient Housing Unit (OHU): A housing unit of a city, county, or city and county law enforcement facility established to retain patients who require special housing for security or protection. Typically, these are patients whose health condition would not normally warrant admission to a licensed heath care facility and for whom housing in the general population may place them at personal or security risk. Outpatient housing unit residents may receive outpatient health services and assistance with the activities of daily living. Outpatient housing unit beds are not licensed correctional treatment center beds.
-
Correctional Treatment Center (CTC): A health facility with a specified number of beds within a state prison, county jail, or California Division of Juvenile Justice facility designated to provide health care to that portion of the patient population who do not require general acute care level of services but are in need of professionally supervised health care beyond that normally provided in the community on an outpatient basis.
-
Acute Rehabilitation: An acute rehabilitation hospital provides intensive physical, occupational, and speech therapy and supportive nursing services to patients recovering from strokes, amputations, severe burns, etc. This is a community placement.
-
Hospice: Services that are designed to provide palliative care, alleviate the physical, emotional, social, and spiritual discomforts of an individual who is experiencing the last phases of life due to the existence of a terminal disease, and to provide supportive care to the primary care giver and the family. Care may be provided by a skilled or unskilled person under a plan of care developed by a physician or a multidisciplinary team under medical direction.
-
Skilled Nursing Facility: A health facility or a distinct part of a hospital which provides continuous skilled nursing care and supportive care to patients whose primary need is for availability of skilled nursing care on an extended basis. It provides 24-hour inpatient care and, at a minimum, includes physician, skilled nursing, dietary, and pharmaceutical services as well as an activity program.
-
-
(b) Classification Factors
-
These Medical Classification Factors guide the operation of procedures in the Medical Classification System rather than specify any placement eligibility.
-
Temporary Medical Hold: A Temporary Medical Hold is used when a patient requires medically necessary health care services, and it is medically prudent to provide these services at the institution where the patient is currently housed. The Medical Classification Chrono (MCC) will be “Temporary.” Examples of patients who should be reviewed for potential medical holds include, but are not limited to the following:
-
Medical:
-
Patients scheduled for major surgery or recovering from major surgery and requiring close post-operative review by the surgical team.
-
Patients having chemotherapy or radiation therapy treatment.
-
Patients undergoing a diagnostic workup.
-
Patients being fitted for a major prosthetic, requiring temporary prostheses adjustments and frequent visits.
-
Patients awaiting major Durable Medical Equipment.
-
Patients scheduled for a specialist visit, which cannot easily be duplicated elsewhere (e.g., surgical subspecialties such as retinal surgery, or specialized oncology surgery).
-
All urgent Requests for Services or specialty appointments.
-
Hemophiliac, Hepatitis C virus, post-transplant, or human immunodeficiency virus/acquired immunodeficiency syndrome patients requiring close management of medication access and continuity.
-
Patients in the middle of a speech therapy, occupational therapy, or physical therapy regimen which would be adversely impacted by transfer.
-
-
Dental:
-
Patients for whom an immediate denture was recently inserted.
-
Patients at a Program Facility awaiting completion of endodontic treatment.
-
Patients awaiting or in the middle of care for jaw fractures.
-
Untreated Dental Priority Classification 1A conditions.
-
-
Mental Health:
-
Patients receiving Clozapine.
-
Patients awaiting Court or other hearings.
-
-
Obstetrics and Gynecology:
-
Patients with high risk pregnancies, in late second trimester or third trimester.
-
-
Patients in the middle of a diagnostic workup for cancer or other high risk conditions.
-
Public Health:
-
Patients in quarantine or isolation for a variety of conditions including, but not limited to: TB, influenza-like illness, gastroenteritis, sexually transmitted diseases under treatment, source cases until clearance obtained, and contact cases until clearance obtained.
-
-
-
Temporary Medical Isolation: Temporary Medical Isolation means the patient may not be endorsed to another institution unless prompted by a Medical Reason for Endorsement. Patients requiring temporary medical isolation shall also have a temporary medical hold entered on the MCC. Medical Isolation may be confinement to quarters or isolation in a medical setting. For example, a patient with Methicillin-Resistant Staphylococcal infection may be placed on Medical Isolation. If that patient should develop a need for dialysis, the patient could be moved for medical reason to an institution with a dialysis program. The type of isolation must be listed in the Comments section. If the patient requires negative pressure respiratory isolation, the Respiratory Isolation Specialized Service factor must be checked as well. Medical Isolation is always temporary. Closure of institutions or housing units for public health issues does not require an MCC.
-
Long-Term Stay: This applies only to Levels of Care other than OP. This factor means that the patient is expected to continue to need at least the indicated Level of Care for the rest of his/her life.
-
Override: Override means that the MCC has been reviewed by a Regional Health Care Executive and permission has been granted to depart from the usual placement requirements for one or more Medical Classification Factors. The patient’s actual Medical Classification Factors are still completed according to usual procedure and the specific directions regarding permitted departures are listed in the Comments section.
-
-
(c) Intensity of Services
-
The Intensity of Services Medical Classification Factors are a set of scales that indicate the patient’s need for medical services and the institution’s ability to provide those services.
-
Proximity to Consultation indicates the frequency and intensity of the patient’s need for specialty medical services. These services are typically provided in the community by contracted providers. The availability and distance to the services varies by institution. A match between patient need and institution capability reduces risk and cost.
-
No Particular Need means there is no anticipated need for consultations at the present time.
-
Basic Consultations are consultative services typically available in a medium-sized community such as general surgery, orthopedics, obstetrics, radiology, ophthalmology, and internal medicine.
-
Infrequent – There is an anticipated need for fewer than four Basic Consultations per year.
-
Frequent – There is an anticipated need for more than four Basic Consultations per year.
-
-
Tertiary Consultation are consultative services typically available in the university or large medical center setting such as oncology, endocrinology, neurology, neurosurgery, radiation therapy, interventional cardiology, nephrology, and cardio-thoracic surgery.
-
Community Placement indicates the patient requires placement into a community hospital or other medical setting on a permanent basis. The patient should be assigned to an institution that can most efficiently provide the necessary custody services to that outside level of care.
-
-
Functional Capacity
-
Functional Capacity is a scale for the patient’s ability to be assigned to particular jobs. That ability affects placement into certain settings.
-
Vigorous Activity: Qualified for all assignments including food-handling and firefighting. Able to dig ditches, chop wood, haul water, and wear a respirator. Good mobility, endurance, and bilateral grip strength.
-
For California Correctional Health Care Services/California Department of Corrections and Rehabilitation (CDCR) purposes, Chronic Active Hepatitis is defined as patients who are antibody positive, viral load negative, and whose FIB is less than 1.45.
-
Patients must meet the National Fire Protection Association’s (NFPA) standards in order to work as firefighters at fire camps. Patients who do not meet the NFPA standards for firefighters may still be able to work at fire camps in non-firefighter positions (e.g., cooks, clerks, clerical support, porters, mechanics, and those who support other functions). Refer to Appendix 3, Institutional Medical Groupings, “Fire Camps Special Skills” section.
-
Below are the 2013 NFPA standards which disqualify patients from fire camps. (Note: See NFPA Annex A, Explanatory Material for all asterisked items within this section):
-
Head and Neck
-
Defect of skull preventing helmet use or leaving underlying brain unprotected from trauma.
-
Any skull or facial deformity that would not allow for a successful fit test for respirators used by that department.
-
Any head condition that results in the candidate not being able to safely perform one or more of the essential job tasks.
-
Any neck condition that results in the candidate not being able to safely perform one or more of the essential job tasks.
-
-
Eyes and Vision
-
*Far visual acuity less than 20/40 binocular, corrected with contact lenses or spectacles, or far visual acuity less than 20/100 binocular for wearers of hard contacts or spectacles, uncorrected.
-
*Color perception – monochromatic vision resulting in inability to use imaging devices such as thermal imaging cameras.
-
*Monocular vision.
-
Any eye condition that results in the candidate not being able to safely perform one or more of the essential job tasks.
-
-
Ears and Hearing
-
Chronic vertigo or impaired balance as demonstrated by the inability to tandem gait walk.
-
On audiometric testing, average hearing loss in the unaided better ear greater than 40 decibels (dB) at 500 Hz, 1000 Hz, 2000 Hz, and 3000 Hz when the audiometric device is calibrated to ANSI Z24.5, Audiometric Device Testing.
-
Any ear condition (or hearing impairment) that results in the candidate not being able to safely perform one or more of the essential job tasks.
-
*Hearing aid or cochlear implant.
-
-
Dental
-
Any dental condition that results in inability to safely perform one or more of the essential job tasks.
-
-
Nose, Oropharynx, Trachea, Esophagus, and Larynx
-
*Tracheostomy.
-
*Aphonia.
-
Any nasal, oropharyngeal, tracheal, esophageal, or laryngeal condition that results in inability to safely perform one or more of the essential job tasks including fit testing for respirators such as N-95 for medical response, P-100 for particulates and certain vapors, and SCBA for fire and hazmat operations.
-
-
Lungs and Chest Wall
-
Active hemoptysis.
-
Current empyema.
-
Pulmonary hypertension.
-
Active tuberculosis.
-
*A forced vital capacity (FVC) or forced expiratory volume in 1 second (FEV1) less than 70 percent predicted even independent of disease.
-
*Obstructive lung diseases (e.g., emphysema, chronic bronchitis, asthma) with an absolute FEV1/FVC less than 0.70 and with either the FEV1 below normal or both the FEV1 and the FVC below normal (less than 0.80).
-
*Hypoxemia – oxygen saturation less than 90 percent at rest or exercise desaturation by four percent or to less than 90 percent (exercise testing indicated when resting oxygen is less than 94 percent but greater than 90 percent).
-
*Asthma – reactive airways disease requiring bronchodilator or corticosteroid therapy for two or more consecutive months in the previous two years, unless the candidate can meet the requirement in 6.8.1.1.
-
Any pulmonary condition that results in the candidate not being able to safely perform one or more of the essential job tasks.
-
Lung transplant.
-
-
Aerobic Capacity
-
An aerobic capacity less than 12 metabolic equivalents (METs) (1 MET = 42 mL O2/kg/min).
-
-
Heart and Vascular System
-
*Coronary artery disease, including history of myocardial infarction, angina pectoris, coronary artery bypass surgery, coronary angioplasty, and similar procedures.
-
*Cardiomyopathy or congestive heart failure, including signs or symptoms of compromised left or right ventricular function or rhythm including dyspnea, S3 gallop, peripheral edema, enlarged ventricle, abnormal ejection fraction, and/or inability to increase cardiac output with exercise.
-
*Acute pericarditis, endocarditis, or myocarditis.
-
*Syncope, recurrent.
-
*A medical condition requiring an automatic implantable cardiac defibrillator or history of ventricular tachycardia or ventricular fibrillation due to ischemic or valvular heart disease, or cardiomyopathy.
-
Third-degree atrioventricular block.
-
*Cardiac pacemaker.
-
Hypertrophic cardiomyopathy including idiopathic hypertrophic subaortic stenosis.
-
Any cardiac condition that results in the candidate not being able to safely perform one or more of the essential job tasks.
-
Heart transplant.
-
Hypertension.
-
*Uncontrolled or poorly controlled hypertension.
-
*Hypertension with evidence of end organ damage.
-
*Thoracic or abdominal aortic aneurysm.
-
Carotid artery stenosis or obstruction resulting in greater than or equal to 50 percent reduction in blood flow.
-
*Peripheral vascular disease resulting in symptomatic claudication.
-
Any other vascular condition that results in the inability to safely perform one or more of the essential job tasks.
-
-
Abdominal Organs and Gastrointestinal System
-
Presence of uncorrected inguinal/femoral hernia regardless of symptoms.
-
Any gastrointestinal condition that results in the candidate not being able to safely perform one or more of the essential job tasks.
-
-
Metabolic Syndrome
-
Metabolic syndrome with aerobic capacity less than 12 METs.
-
-
Reproductive System
-
Any genital condition that results in inability to safely perform one or more of the essential job tasks.
-
-
Urinary System
-
Renal failure or insufficiency requiring continuous ambulatory peritoneal dialysis or hemodialysis.
-
Any urinary condition that results in the candidate not being able to safely perform one or more of the essential job tasks.
-
-
Spine and Axial Skeleton
-
Scoliosis of thoracic or lumbar spine with angle greater than or equal to 40 degrees.
-
History of spinal surgery with rods that are still in place.
-
Any spinal or skeletal condition producing sensory or motor deficit(s) or pain due to radiculopathy or nerve root compression.
-
Any spinal or skeletal condition causing pain that frequently or recurrently requires narcotic analgesic medication.
-
Cervical vertebral fractures with multiple vertebral body compression greater than 25 percent; evidence of posterior element involvement, nerve root damage, disc involvement, dislocation (partial, moderate, severe), abnormal exam, ligament instability, symptomatic, and/or less than six months post injury or less than one year since surgery.
-
Thoracic vertebral fractures with vertebral body compression greater than 50 percent; evidence of posterior element involvement, nerve root damage, disc involvement, dislocation (severe-with or without surgery), abnormal exam, ligament instability, symptomatic, and/or less than six months post injury or less than one year since surgery.
-
Lumbosacral vertebral fractures with vertebral body compression greater than 50 percent; evidence of posterior element involvement, nerve root damage, disc involvement, dislocation (partial, moderate, severe), fragmentation, abnormal exam, ligament instability, symptomatic, and/or less than six months post injury or less than one year since surgery.
-
Any spinal or skeletal condition that results in the candidate not being able to safely perform one or more of the essential job tasks.
-
-
Extremities
-
Joint replacement, unless all the following conditions are met:
-
Normal range of motion without history of dislocations post-replacement.
-
Repetitive and prolonged pulling, bending, rotations, kneeling, crawling, and climbing without pain or impairment.
-
No limiting pain.
-
Evaluation by an orthopedic specialist who concurs that the candidate can complete all essential job tasks listed in Chapter 5.
-
-
Amputation or congenital absence of upper-extremity limb (hand or higher).
-
Amputation of either thumb proximal to the mid-proximal phalanx.
-
Amputation or congenital absence of lower-extremity limb (foot or above) unless the candidate meets all of the following conditions:
-
Stable, unilateral below-the-knee amputation with at least the proximal third of the tibia present for a strong and stable attachment point with the prosthesis.
-
Fitted with a prosthesis that will tolerate the conditions present in structural firefighting when worn in conjunction with standard firefighting personal protective equipment.
-
At least six months of prosthetic use in a variety of activities with no functional difficulties.
-
Amputee limb healed with no significant inflammation, persistent pain, necrosis, or indications of instability at the amputee limb attachment point.
-
No significant psychosocial issues pertaining to the loss of limb or use of prosthesis.
-
Evaluated by a prosthetist or orthopedic specialist with expertise in the fitting and function of prosthetic limbs who concurs that the candidate can complete all essential job tasks listed in Chapter 5, including wearing personal protective ensembles and self-contained breathing apparatus while climbing ladders, operating from heights, and walking or crawling in the dark along narrow and uneven surfaces that may be wet or icy.
-
Has passed the department’s applicant physical ability test as a condition of appointment without accommodations or modification of the protocol.
-
-
Chronic non-healing or recent bone grafts.
-
History of more than one dislocation of shoulder without surgical repair or with history of recurrent shoulder disorders within the last five years with pain or loss of motion, and with or without radiographic deviations from normal.
-
Any extremity condition that results in the candidate not being able to safely perform one or more of the essential job tasks.
-
-
Neurological Disorders
-
Ataxias of heredo-degenerative type.
-
Cerebral arteriosclerosis as evidenced by a history of transient ischemic attack, reversible ischemic neurological deficit, or ischemic stroke.
-
Hemiparalysis or paralysis of a limb.
-
*Multiple sclerosis with activity or evidence of progression within previous three years.
-
*Myasthenia gravis with activity or evidence of progression within previous three years.
-
Progressive muscular dystrophy or atrophy.
-
Uncorrected cerebral aneurysm.
-
All single unprovoked seizures and epileptic conditions including simple partial, complex partial, generalized, and psychomotor seizure disorders other than as allowed in 6.17.1.1.
-
Dementia (Alzheimer’s and other neurodegenerative diseases) with symptomatic loss of function or cognitive impairment (e.g., less than or equal to 28 on Mini-Mental Status Exam).
-
Parkinson’s disease and other movement disorders resulting in uncontrolled movements, bradykinesia, or cognitive impairment (e.g., less than or equal to 28 on Mini-Mental Status Exam).
-
Any neurological condition that results in the candidate not being able to safely perform one or more of the essential job tasks.
-
-
Skin
-
Metastatic or locally extensive basal or squamous cell carcinoma or melanoma.
-
Any dermatologic condition that would not allow for a successful fit test for any respirator required by the fire department.
-
Any dermatologic condition that results in the candidate not being able to safely perform one or more of the essential job tasks.
-
-
Blood and Blood-Forming Organs
-
Hemorrhagic states requiring replacement therapy.
-
Sickle cell disease (homozygous).
-
Clotting disorders.
-
Any hematological condition that results in inability to safely perform one or more of the essential job tasks.
-
-
Endocrine and Metabolic Disorders
-
*Type 1 diabetes mellitus, unless a candidate meets all of the following criteria:
-
Is maintained by a physician knowledgeable in current management of diabetes mellitus on a basal/bolus (can include subcutaneous insulin infusion pump) regimen using insulin analogs.
-
Has demonstrated over a period of at least six months the motivation and understanding required to closely monitor and control capillary blood glucose levels through nutritional therapy and insulin administration. Assessment of this shall take into consideration the erratic meal schedules, sleep disruption, and high aerobic and anaerobic workloads intrinsic to firefighting.
-
Has a dilated retinal exam by a qualified ophthalmologist or optometrist that shows no higher grade of diabetic retinopathy than microaneurysms, as indicated on the International Clinical Diabetic Retinopathy Disease Severity Scale.
-
Has normal renal function based on a calculated creatinine clearance greater than 60 mL/min and absence of proteinuria. (Creatinine clearance can be calculated by use of the Cockroft-Gault or similar formula. Proteinuria is defined as 24-hour urine excretion of greater than or equal to 300 mg protein or greater than or equal to 300 mg of albumin per gram of creatinine in a random sample.)
-
Has no autonomic or peripheral neuropathy. (Peripheral neuropathy is determined by diminished ability to feel the vibration of a 128 cps tuning fork or the light touch of a 10-gram monofilament on the dorsum of the great toe proximal to the nail. Autonomic neuropathy might be determined by evidence of gastroparesis, postural hypotension, or abnormal tests of heart rate variability.)
-
Has normal cardiac function without evidence of myocardial ischemia on cardiac stress testing (to at least 12 MET) by electrocardiogram (ECG) and cardiac imaging.
-
Has a signed statement and medical records from an endocrinologist or a physician with demonstrated knowledge in the current management of diabetes mellitus as well as knowledge of the essential job tasks and hazards of firefighting as described in 5.1.1, allowing the fire department physician to determine whether the candidate meets the following criteria:
-
Is being successfully maintained on a regimen consistent with 6.20.1(1)(a) and 6.20.1(1)(b).
-
Has had hemoglobin A1C measured at least four times a year (intervals of two to three months) over the last 12 months prior to evaluation if the diagnosis of diabetes has been present over one year. A hemoglobin A1C reading of eight percent or greater shall trigger a medical evaluation to determine if a condition exists in addition to diabetes that is responsible for the hemoglobin A1C not accurately reflecting average glucose levels. This shall include evidence of a set schedule for blood glucose monitoring and a thorough review of data from such monitoring.
-
Does not have an increased risk of hypoglycemia due to alcohol use or other predisposing factors.
-
*Has had no episodes of severe hypoglycemia (defined as requiring assistance of another) in the preceding one year, with no more than two episodes of severe hypoglycemia in the preceding three years.
-
Is certified not to have a medical contraindication to firefighting training and operations.
-
-
-
Insulin-requiring Type 2 diabetes mellitus, unless a candidate meets all of the following criteria:
-
Is maintained by a physician knowledgeable in current management of diabetes mellitus.
-
Has demonstrated over a period of at least three months the motivation and understanding required to closely monitor and control capillary blood glucose levels through nutritional therapy and insulin administration. Assessment of this shall take into consideration the erratic meal schedules, sleep disruption, and high aerobic and anaerobic workloads intrinsic to firefighting.
-
Has a dilated retinal exam by a qualified ophthalmologist or optometrist that shows no higher grade of diabetic retinopathy than microaneurysms, as indicated on the International Clinical Diabetic Retinopathy Disease Severity Scale.
-
Has normal renal function based on a calculated creatinine clearance greater than 60 mL/min and absence of proteinuria. (Creatinine clearance can be calculated by use of the Cockroft-Gault or similar formula. Proteinuria is defined as 24-hour urine excretion of greater than or equal to 300 mg protein or greater than or equal to 300 mg of albumin per gram of creatinine in a random sample.)
-
Has no autonomic or peripheral neuropathy. (Peripheral neuropathy is determined by diminished ability to feel the vibration of a 128 cps tuning fork or the light touch of a10-gram monofilament on the dorsum of the great toe proximal to the nail. Autonomic neuropathy can be determined by evidence of gastroparesis, postural hypotension, or abnormal tests of heart rate variability.)
-
Has normal cardiac function without evidence of myocardial ischemia on cardiac stress testing (to at least 12 METS) by ECG and cardiac imaging.
-
Has a signed statement and medical records from an endocrinologist or a physician with demonstrated knowledge in the current management of diabetes mellitus as well as knowledge of the essential job tasks and hazards of firefighting as described in 5.1.1, allowing the fire department physician to determine whether the candidate meets the following criteria:
-
Is maintained on a stable insulin regimen and has demonstrated over a period of at least three months the motivation and understanding required to closely monitor and control capillary blood glucose levels despite varied activity schedules through nutritional therapy and insulin administration.
-
Has had hemoglobin A1C measured at least four times a year (intervals of two to three months) over the last 12 months prior to evaluation if the diagnosis of diabetes has been present over one year. A hemoglobin A1C reading of eight percent or greater shall trigger a medical evaluation to determine if a condition exists in addition to diabetes that is responsible for the hemoglobin A1C not accurately reflecting average glucose levels. This shall include evidence of a set schedule for blood glucose monitoring and a thorough review of data from such monitoring.
-
Does not have an increased risk of hypoglycemia due to alcohol use or other predisposing factors.
-
*Has had no episodes of severe hypoglycemia (defined as requiring assistance of another) in the preceding one year, with no more than two episodes of severe hypoglycemia in the preceding three years.
-
Is certified not to have a medical contraindication to firefighting training and operations.
-
-
-
Any endocrine or metabolic condition that results in the candidate not being able to safely perform one or more of the essential job tasks.
-
-
Systemic Diseases and Miscellaneous Conditions
-
Any systemic condition that results in the candidate not being able to safely perform one or more of the essential job tasks.
-
-
Tumors and Malignant Diseases
-
Malignant disease that is newly diagnosed, untreated, or currently being treated, or under active surveillance due to the increased risk for reoccurrence.
-
Any tumor or similar condition that results in the candidate not being able to safely perform one or more of the essential job tasks.
-
-
Psychiatric Conditions
-
Any psychiatric condition that results in the candidate not being able to safely perform one or more of the essential job tasks.
-
-
Chemicals, Drugs, and Medications
-
Those that require chronic or frequent treatment with any of the following medications or classes of medications:
-
Narcotics, including methadone.
-
Sedative-hypnotics.
-
Full-dose or low-dose anticoagulation medications or any drugs that prolong prothrombin time, partial thromboplastin time, or international normalized ratio.
-
Beta-adrenergic blocking agents at doses that prevent a normal cardiac rate response to exercise, high-dose diuretics, or central acting antihypertensive agents (e.g., clonidine).
-
*Respiratory medications: inhaled bronchodilators, inhaled corticosteroids, systemic corticosteroids, theophylline, and leukotriene receptor antagonists (e.g., Montelukast).
-
High-dose corticosteroids for chronic disease.
-
Anabolic steroids.
-
Any chemical, drug, or medication that results in the candidate not being able to safely perform one or more of the essential job tasks.
-
-
Tobacco use shall be a Category A medical condition (where allowed by law).
-
Evidence of illegal drug use detected through testing, conducted in accordance with Substance Abuse and Mental Health Service Administration, shall be a Category A medical condition.
-
Evidence of clinical intoxication or a measured blood alcohol level that exceeds the legal definition of intoxication according to the authority having jurisdiction at the time of medical evaluation shall be a Category A medical condition.
-
-
-
-
Full Duty: Qualified for all institutional assignments (including food-handling) without restrictions.
-
Limited Duty: Restrictions on duty assignment, which are listed in the Comments section. For example, no assignment to work where standing for longer than two hours is required. Qualified for food-handling unless specifically noted.
-
Totally Disabled: Incapable of any duty assignment.
-
-
Medical Risk
-
Medical Risk provides a scale of the risk of adverse outcome caused by the patient’s medical conditions.
-
Low Risk: Routine medical conditions, focused on preventative care. Chronic care of common conditions in good control throughout the last year.
-
Medium Risk: Chronic care of well or moderately-controlled common conditions. Requires time-sensitive laboratory studies.
-
High Risk: Chronic care of complicated, unstable, or poorly-controlled common conditions (e.g., asthma with history of intubation for exacerbations, uncompensated end-stage liver disease, hypertension with end-organ damage, diabetes with amputation). Chronic care of complex, unusual, or high risk conditions (e.g., cancer under treatment or metastatic, coronary artery disease with prior infarction). Implanted defibrillator or pacemaker. High risk medications (e.g., chemotherapy, immune suppressants, Factor 8 or 7, anticoagulants other than aspirin). Transportation over a several day period would pose a health risk, such as hypercoagulable state. Case management is required.
-
-
Nursing Care Acuity
-
Nursing Care Acuity is a scale for the extent, frequency, and complexity of nursing interventions and activities needed.
-
Basic, Uncomplicated Nursing (Population Risk Stratification Level I: Primary Prevention): Care of largely well population; prevention and wellness; stable, uncomplicated chronic disease; episodic care of acute injury and illness; routine nursing care in primary care clinic; annual or semi-annual patient service plans; Keep on Person Medications available seven days per week or Nurse administered (NA) medications no more frequent than twice daily.
-
Low-Intensity Nursing (Population Risk Stratification Level II: Secondary Prevention): Care of chronic stable disease; functional limitations compensated by adaptive equipment; patients able to participate in Activities of Daily Living; maintenance of status; prevention of exacerbation; symptom control and management of pain; uncomplicated wound care (time-limited); uncomplicated chemo/radiation therapy; quarterly patient service plans; Unit medication line: Direct Observed Therapy, Nurse Administered Medications, Intramuscular or subcutaneous injections, and Keep on Person Medications.
-
Medium-Intensity Nursing (Population Risk Stratification Level III: Tertiary): Care of complex, stable or at-risk patients; uncomplicated post-surgical care; dementia, quadriplegia, hemiplegia who are able to participate in self-care; uncomplicated wound care; high risk for skin breakdown; Outpatient Housing Unit (OHU) placement; monthly or every two month patient service plans. Case management/care coordination is required.
-
High-Intensity or Specialized Nursing (Population Risk Stratification Level IV: Catastrophic/Complex): Direct, total and/or specialized nursing care of complex, complicated, unstable, or high risk patients; daily nursing plan update; significant dementia, paraplegia, hemiplegia or quadriplegia who are unstable and unable to participate in self-care; complex medication protocols. Care management/care coordination is required. Inpatient level of care.
-
-
-
(d) Minimum Support Facility Criteria
-
To be medically eligible for Minimum Support Facility (MSF) Placement, patients must have a MCC Risk Category designation of Low Risk or meet the Medium Risk criteria for MSF eligibility (see Clinical Risk Definitions on the Lifeline Quality Management Portal). Additionally, patients must also meet all custodial criteria required for MSF placement (i.e., Time in Custody, no S or R Suffix, etc.).
-
-
(e) Specialized Services
-
Specialized Services are special programs or patient needs that are provided by certain specified institutions.
-
Pregnancy Program: Medical program for pregnant and post-partum patients.
-
Transplant Center: Medical program at institutions with agreements with a local transplant center. Currently these patients are managed as part of the continuum of care; this factor then flags these patients for purposes of population management and census.
-
Hemodialysis: Medical program for patients requiring hemodialysis. The program may provide dialysis within the institution or by transportation outside the institution.
-
Dementia: Medical program for patients with dementia. Currently these patients are managed as part of the continuum of care; this factor then flags these patients for purposes of population management and census.
-
Therapeutic Diet: Specified therapeutic diets are available to outpatients and in medical settings. Authorized therapeutic diets include:
-
Gluten-free diet
-
Hepatic diet
-
Renal diet
-
Pre-renal diet
-
-
Respiratory Isolation: Low-pressure respiratory isolation room is required. These rooms are included in CTCs and used primarily in the care of patients with active tuberculosis.
-
Speech/Occupational Therapy: Speech and occupational therapy services. These are most commonly provided for patients being rehabilitated from strokes who are being cared for in a medical setting.
-
Physical Therapy: Physical therapy services, which can be provided both in medical settings and as outpatients.
-
Durable Medical Equipment: Provisioning and repair of durable medical equipment including wheelchairs, prostheses, portable oxygen concentrators, and continuous positive airway pressure devices which are available both in medical settings and as outpatients.
-
Transgender: Medical program for transgender patients.
-
-
(f) INSTITUTIONAL-ENVIRONMENTAL
-
These Classification Factors are related to institutional capabilities or characteristics that are due to the physical location and architectural design.
-
Restricted-Altitude: The patient has a condition that is placed at risk by high altitude (above 3,500 feet) including patients who require supplemental oxygen and patients with sickle-cell disease (sickle-cell trait does not require restriction).
-
Restricted-Cocci Areas
-
Institutions in Restricted-Cocci Area 1 include: Avenal State Prison, California City Correctional Center, California Correctional Institution, California Men’s Colony, California State Prison, Corcoran, California Substance Abuse Treatment Facility and State Prison at Corcoran, Kern Valley State Prison, North Kern State Prison, Pleasant Valley State Prison, Wasco State Prison, and any Community Correctional Facility/Modified Community Correctional Facility that has these institutions as their hub.
-
Patients who are designated as Restricted-Cocci Area 1 are precluded from endorsement to the institutions listed above.
-
Patients with Clinical Category 1 or 2, pregnancy, a history of lymphoma, status post solid organ transplant, chronic immunosuppressive therapy, moderate to severe Chronic Obstructive Pulmonary Disease (on intermittent or continuous O2,) or cancer patients on chemotherapy and/or radiation therapy are restricted from placement in Cocci Area 1, unless they have a history of cocci disease.
-
-
Institutions in Restricted-Cocci Area 2: Avenal State Prison, Pleasant Valley State Prison, and any Community Correctional Facility/Modified Community Correctional Facility that has these institutions as their hub.
-
Patients who are designated as Restricted-Cocci Area 2 are precluded from endorsement to the institutions listed above.
-
High Medical Risk patients and those who test negative with the cocci skin test, have not been offered the cocci skin test, or have an incomplete skin test (e.g., consented to testing but the test has not yet been completed) are absolutely restricted from Cocci Area 2, unless they have a history of cocci disease; these patients cannot waive the restriction.
-
Patients with diabetes or who are Filipino or African American are restricted from Cocci Area 2, unless they have a history of cocci disease or test positive with a cocci skin test; these patients may waive the restriction.
-
-
-
Restricted-No Stairs: Patients who require an environment without stairs for their activities of daily living. This may be due to mobility impairment or to other functional impairments such as heart failure.
-
Requires Electrical Access: Patients with electrically-operated supportive equipment such as portable oxygen concentrators or continuous positive airway pressure devices that require an electrical outlet within six feet of the head of the bed.
-
Requires Adaptive Equipment: Patients who require adaptive equipment in their living area such as grab bars in the toilet or shower or trapeze bars over the bed.
-
Requires Medical Transport: The patient cannot safely be transported using custody staff and a state car, state bus, or state transport van. For example, a quadriplegic with autonomic instability.
-
See 1845 and 7410: The patient has medical needs that are specified on a CDCR 1845, Disability Placement Program Verification, and/or a CDCR 7410, Comprehensive Accommodation Chrono.
-
-
(g) COMMENTS
-
Specified Medical Classification Factors, if present, require supporting details to be written into the “Comments” section of the MCC. These factors are marked with a superscript “*”.
-
Comments that contain protected health information should be entered into the “Confidential Comments” section.
-
-
Appendix 2
-
Medical Classification Factor Priorities
Medical Classification Factor Absolute (A) or Preference (P) Comment Level of Care P
A
APreference ONLY in that a higher level of care may be used. For example, an Outpatient Housing Unit or Hospice patient may be housed in a Correctional Treatment Center.
Specialized Outpatient transfers require classification committee referral and endorsement to California Health Care Facility, Stockton or an intermediate institution.
Op-OutpatientProximity to Consult A Can always be obtained at high extra cost if needed. Functional Capacity A Important particularly with regard to Camp placement and Armstrong. Medical Risk A
PIn California Department of Corrections and Rehabilitation (CDCR) institutions, Medium Intensity Nursing can be provided at extra cost.
If alternative is Basic InstitutionNursing Care Acuity P
AIn California Department of Corrections and Rehabilitation (CDCR) institutions, Medium Intensity Nursing can be provided at extra cost.
In all other cases.Clinical Category 1 A
PIn anything but CDCR institutions.
Can be done in a Clinical Category 2 institution at extra cost.Clinical Category 2 A Pregnancy Program A Transplant Center P Can always be obtained at extra cost if needed. Hemodialysis P Can always be obtained at high extra cost if needed. Dementia P No special program yet exists; tracked for reporting purposes only. Therapeutic Diet A Respiratory Isolation A Speech/Occupational Therapy P Can always be obtained at extra cost if needed. Physical Therapy P Can always be obtained at extra cost if needed. Durable Med Equipment A Transgender A Restricted – Altitude A Restricted – Cocci Area 1 A Restricted – Cocci Area 2 A Restricted – No Stairs A Requires Electrical Access A Requires Adaptive Equipment A Requires Medical Transport A -
Appendix 3
-
Institutional Medical Groupings (Medical Classification System Criteria)
Description of Institutional Setting Functional Capacity Level of Care Proximity to Consultation Medical Risk Nursing Care Acuity Minimum Support Facilities: These settings require that patients be located in a facility attached to but separate from an institution. Nursing and primary care provider care is available, but patients must be taken into the secure perimeter in order to access urgent care. Limited Duty (Or Better) Outpatient (OP) Frequent Basic Consultation (Or Less) Medium Risk (Or Less) Basic, Uncomplicated Nursing or Low Intensity Nursing (Or Less) Community Correctional Facilities: These settings require that the patient be able to be located in a small to medium sized contracted facility that may be many miles from a hub institution. These facilities provide limited nursing and primary care provider access. Patients must be taken to local emergency rooms or transported to the hub for urgent care. Limited Duty (Or Better) OP Infrequent Basic Consultation (Or Less) Low Risk Basic, Uncomplicated Nursing Fire Camps Firefighters: These settings require that the patients be able to be located in remote areas, capable of vigorous physical activity if in firefighter assignments, and require no daily nursing care. Vigorous Activity OP No Particular Need Or Infrequent Basic Consultation Medium Risk
(Or Less)Basic, Uncomplicated Nursing Fire Camps Special Skills: Non-firefighters assigned to fire camps such as cooks, clerks, clerical support, porters, mechanics, and those who support other functions. These patients shall not be assigned to firefighting duties. Vigorous Activity or Full Duty OP No Particular Need Or Infrequent Basic Consultation Medium Risk (Or Less) Basic, Uncomplicated Nursing Out-of-State Facilities: These settings require that patients must be able to be located in a medium-sized contracted facility in another state. These facilities provide nursing and primary care provider services on a continuous basis and can provide urgent care onsite. Short and long term placements into Outpatient Housing Unit (OHU) or Correctional Treatment Center (CTC) are available onsite. Patients must be able to be transported to and from California
using routine custody transportation.Limited Duty (Or Better) OP Frequent Basic Consultation (Or Less) Medium Risk (Or Less) Low Intensity Nursing
(Or Less)Basic Institutions: These facilities provide nursing and primary care provider services on a continuous basis and can provide urgent care onsite. Short and long term placements into OHU or CTC are available onsite. Basic consultations (general surgery, orthopedics, obstetrics, radiology, ophthalmology, internal medicine) are available. OP, OHU, or CTC Frequent Basic Consultation (Or Less) Medium Risk (Or Less) Medium Intensity Nursing
(Or Less). Example: Calipatria State PrisonIntermediate Institutions: These facilities provide nursing and primary care provider services on a continuous basis and can provide urgent care onsite. Short and long term placements into OHU and CTC are available onsite. Basic Consultations (general surgery, orthopedics, obstetrics, radiology, ophthalmology, internal medicine) and Tertiary Care Consultations (oncology, endocrinology, neurology, neurosurgery, interventional cardiology, nephrology, cardio-thoracic surgery) are close and readily available. Specialized Services, such as HIV Clinical Category 1, pregnancy services, therapeutic diets, and hemodialysis may be provided. OP, OHU, or CTC Tertiary Consultation (Or Less) High Risk (Or Less) High Intensity or Specialized Nursing (Or Less). Example: Mule
Creek
State PrisonCenter Institutions: These facilities are restricted to patients with significant medical needs. They provide nursing and primary care provider services on a continuous basis and can provide urgent care onsite. Short and long term placements into OHU and CTC are available onsite. Basic Consultations (general surgery, orthopedics, obstetrics, radiology, ophthalmology, internal medicine) and Tertiary Care Consultations (oncology, endocrinology, neurology, neurosurgery, interventional cardiology, nephrology, cardio-thoracic surgery) are close and available. Specialized Services, including HIV Clinical Category 1, pregnancy services, therapeutic diets, speech therapy, occupational therapy, dementia support program, transplant center, respiratory isolation, complex durable medical equipment, and hemodialysis are all provided. Specialized Outpatient Tertiary Consultation (Or Less) High Risk (Or Less) High Intensity or Specialized Nursing
(Or Less). Example: California Medical Facility.
1.2.15 Utilization Management Program
-
Policy
-
The California Correctional Health Care Services’ (CCHCS) Utilization Management (UM) Program shall ensure the appropriate use of limited health care resources including, but not limited to, medical procedures, consultations with specialists, diagnostic studies, inpatient beds, and outpatient beds allocated for health programs to promote the best possible patient outcomes, eliminate unnecessary cost, and maintain consistency in the delivery of health care services. The UM Program shall:
-
Implement evidence-based medical necessity criteria statewide.
-
Manage requests for specialty services to reduce backlogs, wait times, custody and transport demands, and to improve timely access to care.
-
Manage care transitions for patients in community hospitals and specialized health care housing beds (e.g., Outpatient Housing Unit, Correctional Treatment Center, Skilled Nursing Facility) through care coordination and complex case management to optimize patient health outcomes and the use of resources.
-
Provide a centralized process for reviewing and analyzing clinical, financial, and operational data to identify trends and patterns in the use of contract medical services and health care beds within California Department of Corrections and Rehabilitation (CDCR).
-
Develop the statewide UM Improvement Work Plan at least biennially to include improvement priorities, performance objectives, and associated strategies and activities.
-
Maintain a committee structure at headquarters and in the field to provide oversight of the UM work plans and UM Program requirements.
-
-
-
Purpose
-
The purpose of the UM Program is to optimize the value of contract medical services and the use of specialized health care housing by ensuring appropriate, timely, safe, and cost-effective care for patients who require specialty, hospital, emergency, skilled nursing, and diagnostic services and who are admitted to specialized health care housing within CDCR.
-
-
Responsibility
-
Statewide
-
The Deputy Director, Medical Services, and Deputy Medical Executive, UM, are jointly responsible for the planning, implementation, evaluation, and monitoring of the UM Program at the statewide level.
-
CDCR and CCHCS departmental leadership at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial, and clinical systems are in place and resources are available to ensure appropriate, timely, safe, and cost-effective care for patients.
-
The Headquarters UM Committee (HUMC) is a subcommittee of the statewide Quality Management Committee (QMC) and is responsible for providing oversight of the UM Program at the statewide level, identifying and communicating program goals, developing program-specific improvement plans, and overseeing and supporting implementation of improvement initiatives.
-
-
Regional
-
Regional Health Care Executives are responsible for adherence to the UM Program policy and procedure at a subset of institutions within an assigned region.
-
The UM Nurse Consultant Program Reviewers are responsible for:
-
Regular monitoring of dashboards and patient registries to identify and address potential issues in accessing contract medical services.
-
Ensuring UM Registered Nurses are adequately trained on their role and responsibilities including the use of dashboards, patient registries, and other clinical decision support tools to support timely, safe, efficient, and cost-effective use of contract medical services and specialized health care housing beds at a subset of institutions within an assigned region.
-
-
-
Institutional
-
The Chief Executive Officer (CEO), Chief Support Executive (CSE), Chief Medical Executive (CME), and Chief Nurse Executive (CNE) are responsible for the planning, implementation, evaluation, and monitoring of the UM Program and ensuring adherence to the UM Program policy and procedure at the institutional level.
-
The CSE has overall responsibility for the administrative and support functions of the health care system ensuring local policies and procedures align with and support the goals and objectives of the UM Program.
-
The CEO has overall responsibility for implementation and ongoing oversight of the health care system at the institution. The CEO may delegate decision-making authority to the CME and CNE for daily operations including, but not limited to, the following:
-
Ensuring that resources are effectively deployed to support timely, safe, efficient, and cost-effective use of contract medical services and specialized health care housing beds.
-
Providing access to equipment, supplies, health information systems, patient registries, patient summaries, and evidence-based guidelines.
-
Reviewing and comparing institution Care Team performance including:
-
The overall quality of services.
-
Health outcome data.
-
Assignment of consistent and adequate resources.
-
Utilization of dashboards, patient registries, patient summaries, and decision support tools.
-
Addressing issues as necessary.
-
-
Updating procedures, roles and responsibilities, and training as new tools and technology become available.
-
-
The CME is responsible for the overall medical management of patients and ensures resources are available to meet the needs of the population.
-
The CNE is responsible for managing and overseeing the daily operations of the scheduling system and coordinating the delivery of health care services by monitoring, identifying, and addressing barriers in accessing contract medical services.
-
All members of the institution leadership team are responsible for establishing an organizational culture that promotes interdisciplinary teamwork and continuous process improvement.
-
The Institution UM Committee (IUMC) is a subcommittee of the institution QMC and is responsible for oversight of the UM Program at the local level, developing program-specific improvement plans, and managing implementation of improvement initiatives.
-
The institution QMC reports to the statewide QMC and is responsible for coordinating institution-wide performance evaluation and improvement activities and communicating UM Program performance improvement activities to the statewide QMC.
-
-
-
Procedure Overview
-
This procedure outlines major structures, processes, resources, and requirements of the UM Program.
-
-
Procedure
-
Utilization Management Case Review Process
-
The UM Program shall require review of select patient cases that are high cost, high risk, exceptional, or complex. This process includes up to three levels of review and shall cover prospective, concurrent, and retrospective reviews. The UM case review process shall follow the procedure outlined in the Health Care Department Operations Manual (HCDOM), Section 3.1.11, Outpatient Specialty Services.
-
-
Utilization Management Program Committees and Plans
-
Headquarters and institution committee structures shall be maintained to provide oversight of the UM Program. At least biennially, the statewide UM Program shall prepare an improvement plan that describes statewide priorities and performance objectives for the UM Program. The institution shall establish and maintain a UM Subcommittee Improvement Priorities List and Project Pipeline that tracks underperforming areas per the UM Measurement Plan, identifies other quality or patient safety issues related to UM processes, and assesses each issue to determine a risk score for prioritization and initiation of improvement activities.
-
-
Institutional Utilization Management Committee
-
Responsibilities
-
The IUMC duties shall include, but are not limited to, the following:
-
Ensure compliance with the HCDOM for the Complete Care Model policies and procedures including systems and processes that support timely access, population health management, care coordination, and complex care management.
-
Ensure compliance with the UM Program policy and procedure and other related policies and procedures.
-
At least quarterly, review and analyze data including, but not limited to, dashboards, Health Care Incident and Potential Quality Issue reports, and UM operational reports to ensure timely, safe, efficient, and cost-effective access to specialized health care housing beds and to specialty, hospital, emergency, diagnostic, and other contract medical services.
-
Develop and monitor UM work plans that improve timely, safe, efficient, and cost-effective access to and utilization of specialty, hospital, emergency department, and other contract medical services.
-
Establish interdisciplinary workgroups to conduct Root Cause Analyses, and to assess and improve timely access to health care services including, but not limited to, specialty services, care coordination, and care management to ensure that these systems and processes are highly reliable over time.
-
Ensure that institutions’ health care staff receive training on the UM Program policy and procedure and that providers receive feedback regarding adherence to the policy and procedure.
-
Identify gaps in specialty provider network resources and provide this information to the HUMC through the institution QMC.
-
-
-
Membership
-
The IUMC shall consist of, but not be limited to, the following members:
-
CME (Chairperson)
-
CEO (health care)
-
CNE or Supervising Registered Nurse (SRN) III
-
CSE
-
Supervising Dentist
-
Chief Deputy Warden or Associate Warden for Health Care
-
Chief, Mental Health Services
-
Chief Psychiatrist
-
Chief Physician and Surgeon
-
SRN II staff involved with UM, Triage and Treatment Area, specialty services, and hospital services
-
-
-
Reporting Structure
-
The IUMC reports to the institution QMC. The IUMC shall submit timely and accurate reports at least quarterly to the institution QMC that include its major activities, accomplishments, requests for assistance and training, and recommendations that may include changes to contracts, policy, clinical criteria, or decision support.
-
-
Meetings
-
The IUMC shall meet as often as necessary to carry out its responsibilities, but not less frequently than monthly.
-
Meetings shall be conducted informally using a consensus approach. If a consensus cannot be reached on an agenda item, the Chairperson may call for a vote.
-
A quorum of members must be present at all meetings to ensure diversity of view point and well-rounded discussion. A quorum is met when a minimum of five members are in attendance, either in person or telephonically.
-
Records of committee proceedings shall be kept at a secure, accessible medical program site for a period of three years. At a minimum, the record shall describe all committee actions and recommendations.
-
The proceedings and records of the IUMC shall be confidential and protected from discovery to the extent permitted by law.
-
-
-
Headquarters Utilization Management Committee
-
Responsibilities
-
The HUMC’s duties shall include, but are not limited to, the following:
-
Develop and ensure adherence to the UM Program policy and procedure and clinical criteria that define medical necessity.
-
Implement and oversee the Statewide UM Program Improvement Plan.
-
Develop and report UM Program performance indicators based on the Statewide UM Program Improvement Plan.
-
Analyze and report trends and patterns related to utilization and cost associated with contract medical services.
-
Establish interdisciplinary workgroups to assess and improve timely access to health care services including, but not limited to, specialty services, care coordination, and care management to ensure that these systems and processes are highly reliable over time.
-
Assess and recommend interventions to improve timeliness, safety, efficiency, and cost effectiveness of contract medical services and specialized health care housing.
-
Provide direction for specialty care referrals and bed usage through defining medical necessity, selecting appropriate referral, admission and discharge criteria, and setting statewide standards for UM.
-
Recommend strategies to modify provider network capacity and medical contracts.
-
Refer institution-specific concerns to the respective regional leadership and IUMC for appropriate action.
-
Refer to other headquarters committees for action as appropriate.
-
-
-
Membership
-
The HUMC shall consist of the following voting members:
-
Deputy Medical Executive, UM Program (Co-Chairperson)
-
Deputy Director, Field Operations, Corrections Services (Co-Chairperson)
-
Deputy Director, Business Services
-
Deputy Director, Dental
-
Deputy Director, Fiscal Services
-
Deputy Director, Mental Health Program
-
Deputy Director, Medical Services
-
Deputy Director, Nursing Services
-
Deputy Director, Quality Management
-
Regional Health Care Executives, Region I, II, III and IV
-
-
Members may designate another manager from their program area, and a Co-Chairperson may add additional members and invite other stakeholders as necessary.
-
-
Reporting Structure
-
The HUMC reports to the statewide QMC.
-
-
Meetings
-
The HUMC shall meet as often as necessary to carry out its responsibilities but not less frequently than bimonthly.
-
Meetings shall be conducted informally using a consensus approach. If a consensus cannot be reached on an agenda item, a Co-Chairperson may call for a vote.
-
A quorum of members must be present at all meetings to ensure diversity of view point and well-rounded discussion. A quorum is met when a minimum of 50 percent of the members are in attendance, either in person or telephonically.
-
Records of committee proceedings shall be kept at a secure, accessible medical program site for a period of three years. At a minimum, the record shall describe all committee actions and recommendations.
-
The proceedings and records of the HUMC shall be confidential and protected from discovery to the extent permitted by law.
-
-
-
-
References
-
California Civil Code, Division 1, Part 2.6, Section 56 et seq.
-
California Evidence Code, Division 9, Chapter 3, Section 1157
-
California Code of Regulations, Title 15, Division 3, Chapter 2, Article 1, Section 3999.98
-
California Code of Regulations, Title 15, Division 3, Chapter 2, Subchapter 2, Article 1, Section 3999.200
-
Health Care Department Operations Manual, Chapter 1, Article 2, Sections 1.2.2 through 1.2.9
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.11, Outpatient Specialty Services
-
-
Revision History
-
Effective: 12/2003
Revised: 07/30/2025
-
1.2.16 Gender Affirming Surgery Review Committee
-
Policy
-
California Correctional Health Care Services (CCHCS) patients may request gender affirming surgery (GAS) or revisions to GAS, pursuant to the California Code of Regulations Title 15, Section 3999.200. Patient requests for GAS, including revisions to GAS shall be referred by the institution to the Gender Affirming Surgery Review Committee (GASRC).
-
-
Procedure
-
Institution Referral Process
-
Patient Request for GAS or Revisions to GAS
-
A patient may request GAS or revisions to GAS (excluding complications that need timely follow up by a surgeon) by completing a CDCR 7362, Health Care Services Request Form or by verbally informing their primary care provider (PCP).
-
When a patient submits a CDCR 7362, a PCP appointment shall be scheduled pursuant to the Health Care Department Operations Manual (HCDOM), Section 3.1.5, Scheduling and Access to Care, to review the form.
-
-
Primary Care Provider
-
The PCP shall submit an electronic order for “Request for GAS Review Committee” at the time of the initial visit, and the Electronic Health Record System (EHRS) will generate a “MH GAS Evaluation Initial” order upon placement.
-
If, during the initial PCP visit, a Gender Dysphoria (GD) diagnosis is not currently established by a mental health (MH) clinician, the PCP shall order a “Mental Health Gender Dysphoria Diagnostic Evaluation.”
-
-
Mental Health Staff
-
When a “Mental Health Gender Dysphoria Diagnostic Evaluation” is ordered, the MH Evaluator shall complete and submit the “Mental Health Gender Dysphoria Diagnostic Criteria” PowerForm within 14 calendar days.
-
MH staff who author MH GAS Evaluations are required to complete the associated statewide training.
-
When a “MH GAS Evaluation Initial” order is generated, the MH Evaluator shall:
-
See the patient within 45 calendar days from the date of submission of the electronic order for “Request for GAS Review Committee.” If needed, subsequent “MH GAS Evaluation Follow Up” visits may be ordered and completed within seven calendar days of order entry.
-
Complete and submit the “MH Gender Affirming Surgery (GAS) Evaluation” PowerForm within 90 calendar days from the date of submission of the electronic order for “Request for GAS Review Committee.”
-
-
-
Utilization Management Registered Nurse
-
The Utilization Management Registered Nurse (UMRN) shall complete and submit the “CDCR 7466 Gender Affirming Surgery Request Checklist” PowerForm within 14 calendar days from the completion date of the “MH Gender Affirming Surgery (GAS) Evaluation” PowerForm.
-
-
-
GASRC Membership
-
Co-Chairpersons
-
The Co-Chairpersons shall:
-
Be appointed by the Deputy Director, (DD), Medical Services, and the DD, Statewide Mental Health Program.
-
Consist of a minimum of one Deputy Medical Executive (DME), Assistant DME, Chief Medical Executive (CME), or Chief Physician and Surgeon (CP&S); and a minimum of one Mental Health Assistant DD, Chief Psychologist, or Chief Psychiatrist.
-
Be non-voting members, unless needed to reach a quorum as noted below.
-
Designate a voting member to chair the GASRC in their absence.
-
-
There must be at least one Co-Chairperson, or designee, present at each GASRC meeting.
-
-
Voting members
-
Voting members (and alternate designees) shall be appointed by the DD, Medical Services, and the DD, Statewide Mental Health Program, and shall consist of:
-
A minimum of three physicians or advanced practice providers (APPs) from Medical Services, at least one of which must be a physician;
-
A minimum of three psychiatrists from the Statewide Mental Health Program; and
-
A minimum of three psychologists, licensed clinical social workers (LCSWs), licensed marriage and family therapists (LMFTs), or licensed professional clinical counselors (LPCCs) from the Statewide Mental Health Program.
-
-
Training
-
Mandatory standardized training requirements approved by the GASRC shall be completed prior to serving as a committee Co-Chairperson or voting member for the GASRC.
-
-
-
GASRC Meetings
-
A quorum for purposes of voting shall include at least one voting psychologist, LCSW, LMFT, or LPCC, one voting psychiatrist, and one voting physician or APP from Medical Services. In reaching the quorum requirement, the Co-Chairpersons may serve as a member for their clinical discipline, if necessary, to reach the GASRC quorum.
-
Non-voting participants may include the author of the MH evaluation (or designated MH clinician), the PCP or a provider familiar with the patient’s physical health, and, if deemed appropriate for the discussion by the Co-Chairpersons:
-
Institution medical, MH, or nursing leadership.
-
Warden or designee.
-
The patient’s care team, including medical, MH, or nursing.
-
Nursing representation from headquarters including, but not limited to, the Statewide Chief Nurse Executive or designee.
-
Other pertinent CCHCS or CDCR staff.
-
-
The GASRC shall be scheduled to meet weekly unless there are no requests for the committee to consider.
-
-
GASRC Conflict of Interest
-
A committee member shall not participate in the committee deliberation or vote on a case being reviewed if the member:
-
Was a designated provider in the current GASRC evaluation or review, or
-
Was the MH Evaluator, or
-
Has provided longitudinal care for the patient in the past 12 months.
-
-
When a member has a conflict, the member shall:
-
Not deliberate or vote on the case, but they may still present information to the committee and be available for questions.
-
Self-identify a potential conflict and voluntarily notify the committee Co-Chairpersons and recuse themselves for that case(s).
-
-
-
GASRC Scope of Review
-
Submissions for GAS shall be considered by the GASRC in the order in which they are received, absent extenuating circumstances approved by the Co-Chairpersons.
-
For GASRC submissions that include more than one requested surgical procedure, each surgical procedure request shall be considered separately.
-
The GASRC shall review cases within 90 calendar days from date of submission of the “CDCR 7466 Gender Affirming Surgery Request Checklist” PowerForm, absent extenuating circumstances.
-
The GASRC shall review, evaluate, and discuss the information provided by the institution (and the patient, if applicable) and obtain additional information as deemed necessary.
-
If the GASRC deems additional information is required prior to making a final decision, the GASRC review shall be deferred until the additional information is collected.
-
If the GASRC requires additional information from a surgeon who specializes in GAS, the patient shall be scheduled for a consultation with the patient’s care team and the GASRC review deferred until such time that the consultation notes are available in the EHRS.
-
The following shall be considered by the GASRC when reviewing a GAS request, if applicable based on the current version of the World Professional Association for Transgender Health Standards of Care and the type of surgery requested:
-
If a diagnosis of GD is supported with appropriate documentation and clinical justification.
-
Any known medical or MH contraindications to surgery or conditions that may impact surgical recovery have been fully assessed and well-controlled for an appropriate amount of time specific to the patient.
-
The level of distress related to GD demonstrated by the patient. The GASRC shall:
-
Review all relevant documents and determine whether the patient’s GD symptoms are primarily due to environmental factors, mental illness, or any other factor.
-
Consider whether available treatments other than GAS could improve or alleviate the patient’s GD symptoms.
-
-
If there is evidence suggestive of any external coercion or predation.
-
If evidence exists that suggests the patient does not have the ability to successfully and safely transfer and adjust medically and psychologically to their new environment postoperatively including the ability to follow medical or MH advice.
-
If applicable to the GAS requested, whether the:
-
Patient received 6 to 12 continuous months of medically supervised gender affirming hormone therapy (GAHT) appropriate to their gender embodiment goals (unless they have a medical contraindication, are unable or do not desire to take hormones, or their gender embodiment goals do not include the use of GAHT).
-
Patient’s hormone levels meet the minimum requirements for the requested procedure (unless they have a medical contraindication, are unable or do not desire to take hormones, or their gender embodiment goals do not include hormone levels at a specific minimum threshold/range).
-
-
If applicable to gonadectomy, whether there is documentation in the EHRS to reflect discussions and understanding by the patient that the procedure will result in complete loss of fertility.
-
Any other information available that may be relevant to the discussion or determination.
-
-
-
GASRC Decisions
-
The decisions of the committee shall be based on the following:
-
When a quorum of less than six voting members results in a unanimous vote, the decision will stand.
-
When a quorum of less than six voting members results in a split vote, the case shall be brought back to a larger committee consisting of at least two representatives from each aforementioned discipline (excluding the Chairperson), whereby a decision will be based on a majority vote.
-
In the event of a tie, the Chairperson has the option to request additional information and review the case at future GASRC meetings or serve as the tiebreaker if no additional information is needed or available.
-
-
The Chairpersons may postpone any GASRC meeting to request additional information and review the case at future GASRC meetings.
-
If additional information is obtained after the GASRC has issued an approval but before the completion of surgery, the GASRC may reconsider the approval if deemed necessary. The committee may determine that a rescission of a former approval is warranted.
-
Once the GASRC has made a decision (approval, non-approval, or rescission of former approval), a memorandum shall be completed conveying the decision, the factors considered in the decision based on the criteria noted in Section (b)(5)(G) above, and any specific information that the GASRC determines would be helpful to the patient and their care team to understand the decision. The decision memorandum shall be addressed to the CME, and copies shall be provided to the following:
-
Patient
-
CP&S
-
Chief of Mental Health
-
Chief of Psychiatry
-
Chief Nursing Executive
-
PCP
-
Mental Health Primary Clinician, if applicable
-
Mental Health Primary Psychiatrist, if applicable
-
UMRN
-
Designees from Health Care Invoicing and Direct Care Contracts applicable only to approvals or rescissions
-
-
GASRC support staff shall distribute the decision memorandum to the CME via email within seven calendar days from the GASRC decision, absent extenuating circumstances.
-
The GASRC decision shall be entered into the EHRS by a committee Co-Chairperson or designee.
-
Patients who are approved for GAS shall be scheduled:
-
With an MH clinician to discuss the findings of the GASRC within 14 calendar days of the decision entry in the EHRS. A copy of the GASRC decision memorandum shall be provided to the patient at that time.
-
For an initial visit with the surgeon, when an approval is entered into the EHRS.
-
-
Patients who are not approved for GAS, or who have had their former approval rescinded:
-
Shall be scheduled separately with an MH clinician and PCP to discuss the findings of the GASRC within 14 calendar days of decision entry in the EHRS. A copy of the GASRC decision memorandum shall be provided to the patient at the MH appointment.
-
May submit a new request for the same GAS no sooner than one year after the date of the last issued GASRC decision memorandum, unless new information is provided that was not previously considered at the time the GASRC reviewed the case and rendered a decision.
-
May submit information regarding a particular GAS request, that was not previously available at the time of the most recent GASRC review, only through the process under Section (b)(1).
-
May grieve any GASRC decision without submitting information that was not previously available at the time of the GASRC review, only through the health care grievance process set forth in the HCDOM, Section 5.1.7, Health Care Grievance.
-
-
The GASRC support staff shall scan the GASRC decision memorandum and documents considered in making the decision into the EHRS within seven calendar days of receipt of the signed GASRC decision memorandum.
-
-
Record and Information Requests related to Gender Affirming Care or Services
-
Any court order, administrative order, or subpoena requesting medical records related to gender affirming health care shall be processed by Health Information Management in accordance with the HCDOM, Section 2.3.4, Release of Protected Health Information.
-
Refer to the HCDOM, Section 4.1.7, Gender Dysphoria Management, for information regarding the disclosure of gender affirming related medical information to an individual, agency, or department from another state, or to a federal law enforcement agency.
-
-
-
References
-
California Civil Code, Division 1, Part 2.6, Chapter 2, Section 56.109
-
California Code of Regulations Title 15, Division 3, Chapter 2, Subchapter 2, Article 1, Section 3999.200
-
California Department of Corrections and Rehabilitation, Department Operations Manual, Chapter 6, Article 12, Section 62080.14, Transgender or Intersex Inmates
-
Health Care Department Operations Manual, Chapter 2, Article 3, Section 2.3.4, Release of Protected Health Information
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.5, Scheduling and Access to Care
-
Health Care Department Operations Manual, Chapter 4, Article 1, Section 4.1.7, Gender Dysphoria Management
-
Health Care Department Operations Manual, Chapter 5, Article 1, Section 5.1.7, Health Care Grievance
-
American Psychiatric Association, Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition Text Revision, 2022
-
World Professional Association for Transgender Health (WPATH) Standards of Care for the Health of Transgender and Gender Diverse People, Version Eight (8), 2022
-
-
Policy Control
Executive Sponsors: Deputy Directors, Medical Services and the Statewide Mental Health Program
Effective: 07/21/2021
Revised: 6/30/2023, 10/09/2024, 6/10/2026
1.2.17 Continuing Health Care Education Planning Committee
-
Policy
-
The Continuing Health Care Education (CHCE) Planning Committee provides oversight for the development, implementation, and dissemination of continuing medical education activities that serve to maintain, develop, or increase the knowledge, skills, and professional performance of health care providers. Continuing Medical Education for health care providers improves the quality of care provided to patients. California Correctional Health Care Services (CCHCS) shall provide continuing education courses and activities which are eligible for Continuing Medical Education credits pursuant to requirements set forth in Business and Professions Code, Section 2190, et seq. and California Code of Regulations, Title 16, Section 1336, et seq.
-
-
Responsibility
-
Statewide
-
California Correctional Health Care Services (CCHCS) Deputy Director (DD), Medical Services, and the Deputy Medical Executive (DME), Policy and Provider Workforce, are responsible for the statewide planning, implementation, and evaluation of the CHCE policy.
-
-
Regional
-
The Regional leadership are responsible for ensuring that primary care providers and other applicable clinical staff attend Continuing Medical Education trainings and apply evidence-based recommendations to their practice as appropriate.
-
-
Institution
-
Health care leadership is responsible for the following:
-
Overall health care staff adherence to this procedure at the institution within the appropriate discipline.
-
Health care staff participation in appropriate discipline-specific Continuing Medical Education trainings.
-
Implementation of evidence-based health care practices consistent with the content presented within Chief Medical Executive (CME) activities.
-
-
Medical Services Division
-
Education and Training Unit (ETU) staff are responsible for responding to audits of CME activities and reviewing to ensure the activities meet all criteria if audited by the accrediting organization.
-
-
-
CHCE Planning Committee Membership and Meetings
-
The Chair shall be a DME or Assistant DME appointed by the DD, Medical Services. The Chair shall assist the DD, Medical Services, in selecting members of the committee from among CCHCS/California Department of Corrections and Rehabilitation (CDCR) staff.
-
Voting members (and alternate designees) shall be appointed by the DD, Medical Services, based on demonstrated expertise and active interest in CHCE from among CCHCS/CDCR staff nominated by relevant clinical leaders for each discipline:
-
Chair, Clinical Documentation and Decision Support Committee
-
Two Headquarters (HQ) Physician Managers
-
One Regional Physician Manager
-
Three Institution Physician Managers
-
One CNE or Nurse Consultant Program Review
-
One Chief or Senior Psychologist
-
One Chief or Senior Psychiatrist
-
One Chief or Supervising Dentist
-
One Quality Management Representative
-
One Pharmacy Services Representative
-
-
Committee members may choose a designee to serve in their stead. Designees must be approved by the Chair or the Chair’s designee.
-
Non-voting participants shall include:
-
The author of the activity application.
-
Presenter(s).
-
Subject matter expert(s).
-
-
A quorum for purposes of voting shall include at least six committee members. In the event that six voting committee members are not present or if a tie occurs, agenda items and applicable materials shall be sent to committee members to vote via electronic vote (e-vote).
-
All voting members (including when there are more than required for a quorum) present at the committee meeting shall be eligible to vote to approve or reject an activity application.
-
Committee meetings shall be scheduled monthly but may be cancelled if there are no items for review.
-
-
Continuing Medical Education Requests
-
Continuing Medical Education Category 1 Credit Application
-
A CME Category 1 Credit Application is required by the committee and shall be submitted for approval. All sections of the form must be filled out prior to submission. A copy of the CME Category 1 Credit Application can be found on the Education and Training Unit SharePoint site on Lifeline under Medical Services: CME Application form.doc (sharepoint.com).
-
The CME Category 1 Credit Application includes extensive planning to outline the Continuing Medical Education description, support, and implementation to ensure education provided is thorough, complete, and appropriate to receive Continuing Medical Education credit.
-
If the application is approved, the committee shall contact the author and determine a mutually acceptable timeframe to present the activity.
-
In preparation for the committee meeting, the author shall create a presentation or visual to share. The presentation shall be sent to the CHCE Planning Committee support staff prior to the presentation date to ensure appropriate visibility and functionality.
-
-
CHCE Planning Committee Scope of Review
-
Submissions for Continuing Medical Education credits shall be considered by the committee in the order in which they are received, absent extenuating circumstances approved by the Chair.
-
The committee shall review, evaluate, and discuss the information provided by the author of the CME Category 1 Credit Application and obtain additional information as deemed necessary.
-
The committee shall consider the following criteria when evaluating a CME activity:
-
Activity description that includes location, anticipated dates, and type of activity (e.g., internet or webinar, live course, enduring material, other).
-
Professional practice gaps to be addressed and learning objectives to be achieved through activity (e.g., data, sources, physician attributes or core competencies, alignment with CHCE and CCHCS missions).
-
Educational design/methodology and educational strategies to achieve objectives.
-
Cultural and linguistic competency implemented to address competency as required by California Business and Professions Code, Section 2190.1.
-
Addressing biases in health care.
-
Evaluation and outcome measurements based on knowledge, performance, and patient outcomes.
-
-
-
Continuing Health Care Education Committee Decisions
-
After reviewing each Category 1 Credit Application, the committee shall take one of the following actions:
-
Approve the application. If the committee approves the activity, the committee support staff shall promptly notify the author in writing of the approval following the meeting.
-
Return the application to the author for revisions. Once the author has made the recommended revisions, the author shall submit the revised application for further evaluation by the committee. The committee shall determine whether an additional meeting is required to further discuss the application, or the committee may determine that no further discussion is required and may vote on the application electronically. The committee support staff shall promptly notify the author in writing of the approval following the meeting.
-
Deny the application. The committee may determine in its discretion that a proposed activity should not be implemented. The committee support staff shall promptly notify the author in writing of the approval following the meeting.
-
-
When an application is approved, the author shall then proceed to work with the ETU to implement their activity.
-
-
Live Continuing Medical Education Activities
-
To enroll for an upcoming CME course, clinical staff shall review the corresponding Activity Announcement emails sent from the ETU mailbox, and save the corresponding appointment invite to their Outlook calendar.
-
Attendance for live activities delivered via webinar is captured electronically via the designated platform when attendees sign in from their CDCR account. Attendance for live in-person activities may be captured via a sign-in sheet.
-
CME credit for in-person activities shall be obtained by submitting sign-in sheets to ETU@cdcr.ca.gov.
-
-
Enduring Material Continuing Medical Education Activities
-
An enduring material is an on-demand activity that does not have a specific time or location designated for participation; rather, the participant determines whether and when to complete the activity. Enduring material can be found on the ETU SharePoint site under ‘Continuing Medical Education Activities – Enduring Material.’
-
CME credit for enduring materials shall be obtained by completing the post-activity assessment associated with the activity. Credit for completing CME enduring material is given commensurate with the approved AMA PRA Category 1 Credit™ hours.
-
-
Reporting AMA PRA Category 1 Credit™ hours
-
For staff other than Medical Doctors (MDs) and Doctors of Osteopathic Medicine (DOs), attendance for each activity is tracked by CHCE Planning Committee support staff, and annual transcripts are provided to the field.
-
Attendance for MDs and DOs is reported directly to the Accreditation Council for Continuing Medical Education’s (ACCME) Program and Activity Reporting System and these MDs and DOs may access their CME activity transcript at any time via ACCME’s https://www.cmepassport.org/.
-
-
-
Record Retention
-
Documentation of committee recommendations and resulting decisions shall be retained for three years.
-
-
References
-
California Business & Professions Code, Division 2, Chapter 5, Article 10, Section 2190, et seq.
-
California Department of Corrections and Rehabilitation, Department Operations Manual, Chapter 1, Article 30, Section 15030.8, Program and Policy Coordination Section
-
California Medical Association, CME Accreditation, AMA PRA Category 1 Credits(s)™
-
California Code of Regulations, Title 16, Section 1336, et seq.
-
-
Revision History
-
Effective: 11/2021f
Revised: 01/06/2025
-
1.2.18 Health Care Ethics Committee
-
Policy
-
California Correctional Health Care Services (CCHCS) shall maintain an Ethics Committee at headquarters to support clinicians and improve patient care by:
-
Consulting on and reviewing cases where ethical dilemmas have been identified and staff, patients, or surrogates have questions or are in conflict.
-
Educating health care staff and patients using the wide body of literature in bioethics and published legal decisions related to clinical ethics.
-
-
Purpose
-
The CCHCS Ethics Committee shall provide multidisciplinary consultation, guidance, and education about the ethical aspects of providing health care within the California prison system.
-
The Ethics Committee supports ethical reflection, respectful dialogue, and critical analysis based on standard practices from the ethics literature to facilitate resolution of bioethical dilemmas.
-
Organizational benefits from the Ethics Committee include:
-
Enhancing patient care
-
Conserving resources/avoiding unnecessary costs
-
Improving accreditation review
-
Reducing risk of lawsuits
-
Sustaining organizational integrity
-
Encouraging professionalism of all staff within the organization
-
-
-
Applicability
-
The Ethics Committee is accountable to and reports to the Governing Body or a subcommittee which reports to the Governing Body.
-
-
Procedure
-
Ethics Committee Membership
-
Chairperson and Vice-chairperson
-
Chairperson shall be a clinician or nurse from headquarters or the field appointed by the Director, Health Care Operations or designee.
-
Vice-chairperson shall be a clinician or nurse administrator from headquarters appointed by the Director, Health Care Operations or designee.
-
-
General Membership
-
Members will be nominated by leaders of disciplines which support the Ethics Committee. Individuals nominated should have an interest in the area of health care ethics, be willing to serve a minimum of one year on the Ethics Committee, and attend monthly meetings on a regular basis. Membership is composed of approximately 20 members of a multidisciplinary group of CCHCS and California Department of Corrections and Rehabilitation (CDCR) representatives. Each of the following disciplines/groups shall be invited/encouraged to appoint a member to the Ethics Committee:
-
Medical Services
-
Nursing Services
-
Mental Health Services
-
Dental Services
-
Custody
-
CCHCS Office of Legal Affairs
-
CDCR Office of Legal Affairs
-
Chaplaincy
-
CDCR Division of Adult Institutions
-
Community members
-
-
Ethics Committee members shall be geographically designated throughout the state.
-
Members with expertise in issues relevant to a referred case will be specifically invited to attend meetings where these issues will be discussed (i.e., a mental health representative for a mental health issue, a dental representative for a dental case, etc.).
-
-
Consult Team
-
As time and support permit, a subset of committee members shall be trained to serve as clinical ethics consultants.
-
Selected individuals shall be among those who have expressed interest and possess the clinical knowledge necessary to triage consult requests.
-
These identified individuals shall receive additional training and mentoring including attending a comprehensive ethics course, if possible, at least once during their first year of service.
-
Attempts shall be made to designate Consult Team members geographically.
-
Designated trained and qualified Consult Team members shall triage Ethics Committee consultation requests from institutions and obtain additional information on each case as needed. In addition, these members may provide ethics education to the referring institution and further guidance if/when an Ethics Committee consultation is deemed not appropriate. Pending the formation of a Consult Team, the Ethics Committee Chairperson, Vice-chairperson, or designee, shall perform these functions.
-
Consultations provided by Consult Team members shall be reviewed at scheduled Ethics Committee meetings.
-
-
-
Meetings
-
The Ethics Committee shall meet no less than quarterly.
-
A quorum shall exist when at least six members are present. Each member shall have one vote.
-
Members of the Ethics Committee in any discipline may vote on Ethics Committee recommendations.
-
Ethics Committee action is approved with a majority vote. A record of Ethics Committee proceedings shall be kept in a secure location, in which all Ethics Committee actions and recommendations are described.
-
-
Ethics Committee Meeting Activities
-
The Ethics Committee consults on and reviews institutional cases referred by individuals from throughout the organization to the Ethics Committee wherein staff has identified ethical dilemmas and when staff, patients, or surrogates have questions or are in conflict.
-
Participation of referring individuals is encouraged when discussing referred, institutional cases.
-
The Ethics Committee also reviews and discusses cases reviewed by Consult Team members.
-
Meeting agenda includes:
-
New active case consultations, presentations, and discussions.
-
Consults managed in the past month by a committee member.
-
Review of recommendations and topics of interest.
-
Journal club, ethics related topics.
-
-
The Ethics Committee may use the four-box method of ethics consultation for all recommendations. (Refer to Appendix 1).
-
The Ethics Committee does not make health care treatment decisions as these are between providers and their patients. The suggestions presented by the Ethics Committee to the referring institution are not institutionally binding, nor are they legally binding. The Ethics Committee shall make recommendations in specific cases in a prospective manner, but shall not judge the ‘ethics’ of past events or decisions.
-
The Ethics Committee provides relevant education and training on ethics topics to Ethics Committee members and to staff via webinar or other appropriate distribution methods.
-
-
Confidentiality
-
The proceedings and records of the CCHCS Ethics Committee shall be confidential and protected from discovery to the extent permitted by law.
-
-
-
Appendices
-
Appendix 1: Four Box Method of Ethics Consultation
-
-
References
-
California Civil Code, Division 1, Part 2.6, Section 56, et seq., Confidentiality of Medical Information Act
-
Albert Jonsen, Mark Siegler, William Winslade; Clinical Ethics, A Practical Approach to Ethical Decisions in Clinical Medicine, 4th Edition. McGraw-Hill, Inc., New York, 1998
-
-
Revision History
-
Effective: 12/2013
-
-
Appendix 1: Four Box Method of Ethics Consultation
Medical Indications Patient Preferences The Principles of Beneficence and Nonmaleficence
1. What is the patient’s medical problem? History? Diagnosis? Prognosis?
2. Is the problem acute? Chronic? Critical? Emergent? Reversible?
3. What are the goals of treatment?
4. What are the probabilities of success?
5. What are the plans in case of therapeutic failure?
6. In sum, how can this patient be benefited by medical and nursing care, and how can harm be avoided?The Principle of Respect for Autonomy
1. Is the patient mentally capable and legally competent? Is there evidence of incapacity?
2. If competent, what is the patient stating about preferences for treatment?
3. Has the patient been informed of benefits and risks, understood this information, and given consent?
4. If incapacitated, who is the appropriate surrogate? Is the surrogate using appropriate standards for decision-making?
5. Has the patient expressed prior preferences,
e.g., Advance Directives?
6. Is the patient unwilling or unable to cooperate with medical treatment? If so, why?
7. In sum, is the patient’s right to choose being respected to the extent possible in ethics and law?Quality of Life Contextual Features The Principles of Beneficence and Nonmaleficence and Respect for Autonomy
1. What are the prospects, with or without treatment, for a return to normal life?
2. What physical, mental, and social deficits is the patient likely to experience if treatment succeeds?
3. Are there biases that might prejudice the provider’s evaluation of the patient’s quality of life?
4. Is the patient’s present or future condition such that his or her continued life might be judged undesirable?
5. Is there any plan and rationale to forgo treatment?
6. Are there plans for comfort and palliative care?The Principles of Loyalty and Fairness
1. Are there family issues that might influence treatment decisions?
2. Are there provider (physicians and nurses) issues that might influence treatment decisions?
3. Are there financial and economic factors?
4. Are there religious or cultural factors?
5. Are there limits on confidentiality?
6. Are there problems of allocation of resources?
7. How does the law affect treatment decisions?
8. Is clinical research or teaching involved?
9. Is there any conflict of interest on the part of the providers or the institution? -
Albert Jonsen, Mark Siegler, William Winslade; Clinical Ethics, A Practical Approach to Ethical Decisions in Clinical Medicine, 4th Edition. McGraw-Hill, Inc., New York, 1998
1.2.19 Headquarters Durable Medical Equipment and Medical Supply Committee and Interdisciplinary Team
-
Policy
-
The Headquarters Durable Medical Equipment and Medical Supply Committee (HDMEC) shall provide statewide oversight and management of Durable Medical Equipment (DME) and medical supplies provided by California Correctional Health Care Services (CCHCS) to California Department of Corrections and Rehabilitation (CDCR) patients. Institutions can request changes to the Durable Medical Equipment and Medical Supply Formulary and appeal denials of nonformulary DMEs to the HDMEC.
-
Responsibility
-
Statewide
-
CCHCS and CDCR departmental leadership at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial, and clinical systems are in place, and appropriate tools, training, technical assistance, and levels of resources are available so that care teams can successfully implement the HDMEC policy.
-
The Deputy Director (DD) of Medical Services and the Deputy Medical Executive of Utilization Management are responsible for the statewide planning, implementation, and evaluation of the HDMEC policy.
-
-
Regional
-
Regional leadership is responsible for this policy at the subset of institutions within an assigned region.
-
-
Institutional
-
The Chief Executive Officer, or designee, has overall responsibility for adherence to this policy at the institution.
-
-
-
Procedure
-
HDMEC
-
Membership
-
The Chair shall be a Deputy Medical Executive or Assistant Deputy Medical Executive appointed by the DD, Medical Services. The Chair shall assist the DD, Medical Services, in the selection of other members of the HDMEC.
-
Voting Members (and alternate designees) are appointed by the DD, Medical Services, from among CCHCS and CDCR staff nominated by relevant leaders for each discipline:
-
Two or more physician executives or managers, or their designees, from Medical Services.
-
Two or more institution or regional physician executives or managers.
-
Two or more executives or managers, or their designees, from Nursing Services.
-
Two or more institution or regional nursing executives or managers.
-
The Chair may designate any voting member to chair the HDMEC in their absence.
-
-
Non-Voting Members
-
Members may include one executive or managerial representation from:
-
Dental Services.
-
Psychiatry Services.
-
Procurement Services.
-
Direct Care Contracts.
-
Health Care Correspondence and Appeals Branch.
-
Corrections Services.
-
Division of Adult Institutions.
-
CCHCS Office of Legal Affairs
-
Fiscal Management Section
-
-
The Chair is a non-voting member unless needed to reach a quorum, as noted below.
-
-
-
Meetings
-
The HDMEC shall meet as often as necessary but not less frequently than annually.
-
A quorum shall consist of at least 50% of voting members in attendance. In reaching the quorum requirement, alternate designees of the same clinical discipline may be counted toward a quorum, and the Chair may serve as a member for their clinical discipline, if necessary, to reach a quorum.
-
In the event of a tie amongst voting members, the Chair has the option to serve as the tiebreaker, if not acting as a voting member to reach a quorum, or request additional information and review the case a second time at a future HDMEC meeting with the same voting members present.
-
-
A record of the proceedings shall be kept and shall record activities, recommendations of the committee, and attendance.
-
The proceedings and records of the HDMEC shall be confidential and protected from discovery to the extent permitted by law.
-
-
Scope of Review
-
The HDMEC shall report to the Headquarters Utilization Management Committee.
-
The HDMEC is responsible for the following:
-
Review all approved requests for nonformulary DME from institutions.
-
Review and resolve provider appeals of denied nonformulary DME requests.
-
Review and resolve issues pertaining to security concerns versus health care needs.
-
Provide a written response to the requestor, if applicable.
-
Determine the necessity of DMEs and maintain the Durable Medical Equipment and Medical Supply Formulary.
-
-
-
Requests to HDMEC
-
To request an addition, change, or deletion of item(s) on the DME and Medical Supply Formulary, institutional staff shall submit a Form OBS 5002.
-
If a request for non-formulary DME or medical supply is denied at the institutional level the primary care provider may request a higher-level review by the HDMEC. The Chief Medical Executive, or designee, shall then forward the denied request for service through the “HQ DME Committee” message pool for further review.
-
-
-
Medical Equipment and Supply Interdisciplinary Team
-
Membership
-
The members shall be selected so as to represent the functional areas of the institution that are necessary for the appropriate and coordinated delivery of DMEs and medical supplies, including a minimum of two physicians and two nurses.
-
-
Meetings
-
The Medical Equipment and Supply (MES) Interdisciplinary Team shall meet at least monthly.
-
A record of the proceedings shall be kept and shall record activities, recommendations of the MES Interdisciplinary Team, and attendance.
-
-
Scope of Review
-
The MES Interdisciplinary Team shall:
-
Report to the HDMEC.
-
Utilize the Health Care Services Dashboard to monitor and review the timely distribution of DME.
-
Review and address out-of-compliance orders.
-
Review and address discrepancies of DME orders in the Electronic Health Records System and Strategic Offenders Management System.
-
Review and disseminate new statewide guidelines, policies, procedures, and recommendations.
-
Provide oversight and management of institutional practices and processes of DME and medical supplies that is consistent with CCHCS and CDCR standards.
-
-
-
-
References
-
Code of Federal Regulations, Title 42, Chapter 7, Subchapter XVIII, Part E, Section 1395 x(n), Durable Medical Equipment
-
California Civil Code, Division 1, Part 2.6, Section 56, et seq., Confidentiality of Medical Information Act
-
California Evidence Code, Division 9, Chapter 3, Section 1157
-
California Code of Regulations, Title 15, Division 3, Chapter 2, Article 1, Section 3999.98, Definitions
-
California Code of Regulations, Title 22, Division 3, Subdivision 1, Chapter 3, Article 2, Section 51160, Durable Medical Equipment
-
Armstrong Remedial Plan, Armstrong vs. Newsom, U.S. District Court of Northern California, Case No. C94-2307 CW, Amended January 3, 2001
-
California Department of Corrections and Rehabilitation, Department Operations Manual, Chapter 5, Article 43, Section 54030.6, Liability
-
-
Revision History
-
Effective: 07/22/2024
-
