Article 1 – Complete Care Model
3.1.1 Complete Care Model
-
Policy
-
California Correctional Health Care Services (CCHCS) in partnership with other California Department of Corrections and Rehabilitation (CDCR) divisions shall manage and deliver medically necessary health care services to the patient population. The Complete Care Model (CCM) is based on the industry standard known as the Patient-Centered Health Home. The CCM shall serve as the foundation for CCHCS health care services delivery. Within the CCM, staff shall utilize a Whole Person Care approach which recognizes that the best way to improve health outcomes is to consider the full spectrum of a patient’s needs – including medical, behavioral, socioeconomic, and beyond. This model improves patient care, reduces the need for hospitalizations and emergency services and enhances staff satisfaction. The CCM includes the following foundational principles and requirements:
-
Continuous Care. Health care systems and processes shall be structured to ensure that patients have a consistent relationship with a team of interdisciplinary staff accountable for their care, which allows Care Team members to know a patient’s history from experience, integrate new information and decisions from a whole-patient perspective, gain the confidence of their patients, and effectively advocate for patients.
-
CCHCS shall establish interdisciplinary Care Teams at each institution, accountable for the care of defined patient panels and the exchange of relevant clinical information between treatment teams.
-
Each patient shall be assigned a Care Team, and as much as possible, the patient’s primary care encounters shall occur with members of the assigned Care Team.
-
CCHCS shall take action to minimize unnecessary patient transfers from one Care Team to another and shall design effective systems and processes to ensure that patient needs are communicated prior to transfers and patients receive timely access to necessary services before, during, and after transfer.
-
-
Comprehensive Care. The health care system shall be designed to meet the patient’s health needs as a whole person, promote collaboration and coordination of services to address a single discipline, condition, or episode of care. CCHCS shall employ risk stratification, population management, and case management among the strategies used to achieve comprehensive care.
-
The Care Team shall be responsible for:
-
Assessing and periodically evaluating patient health needs;
-
Meeting health care needs, including prevention and wellness services, episodic care, chronic care, urgent or acute care, and end-of-life care; and
-
Assessing health care needs beyond the scope of the health care team and referring patients to appropriate providers and services.
-
-
CCHCS shall implement programs for patients by risk stratification; provide care management services to patients commensurate with their individual needs and risk levels; and identify and manage subpopulations of patients per evidence-based guidelines.
-
-
Coordination of Care. Patient services shall be coordinated and health information exchanged across all health care settings, levels of care, and specialty services.
-
The Care Team shall serve as the hub for organizing and scheduling health care services, facilitating appropriate delivery of health care services within and across systems, maintaining continuity of care, and managing exchange of information.
-
The Care Team shall establish reliable processes and systems to track the status and follow-up of specialty referrals, diagnostic studies, and treatment regimens from all disciplines including the Division of Rehabilitative Programs.
-
CCHCS shall establish standardized expectations and processes for clear and open communication between the Care Team and other care providers, which shall include:
-
Ensuring accountability for transitions in care;
-
Providing patient support and education before, during, and after transitions in care; and
-
Building relationships with other health care staff providing services to patients within the patient panel
-
-
-
Patient-Centered Care. Health care staff shall encourage patients to partner in their own care and to make informed decisions related to their health and health care choices. Health care staff shall incorporate the patients’ goals, preferences, and needs into treatment plans whenever feasible and appropriate.
-
The Care Team and other health care staff shall actively engage and empower patients to participate in care planning and delivery, supporting patients in learning to manage their own care between appointments with health care staff.
-
CCHCS shall implement programs to assess and improve patient health literacy and promote self-management planning and activities.
-
-
Preventive Care. Health care staff shall provide preventive care to the patient population based on age, gender, and other clinical recommendations from the United States Preventive Services Task Force Guide to Clinical Preventive Services where health care staff can focus on disease prevention and health maintenance. It includes early diagnosis of disease, discovery and identification of patients at risk of development of specific problems, counseling, and other necessary intervention to avert a health problem. Surveillance for infectious diseases, screening tests, health education, and immunization programs are common examples of preventive care.
-
Accessible Care. CCHCS shall ensure that patients receive timely access to the full range of necessary services, that communication with patients is delivered effectively, and adapted as necessary to the patient’s needs.
-
Scheduling systems and processes shall incorporate strategies to optimize access to care and reduce wait times, including a flexible appointment system that accommodates visit lengths, same-day visits, and scheduled follow-ups, as well as strategies to increase efficiency, such as consolidated/bundled appointments.
-
When possible, health care staff shall consider patient preferences regarding access, such as providing appointment times that do not interfere with the patient’s work shifts or classes.
-
-
Use of Health Information. Health care staff shall use health information systems to identify and manage individual patients and patient populations, apply evidence-based standards and guidelines, and to promote continuity and coordination of care and interoperability of health care documentation to improve patient outcomes.
-
Health records shall be completed timely, accurately, and thoroughly, and records from outside the electronic health record shall be readily available prior to patient encounters.
-
CCHCS shall develop connectivity via electronic or other information pathways to encourage timely and effective communication between providers caring for the same patient.
-
CCHCS staff shall integrate clinical decision support into electronic systems to promote the application of current guidelines or standards as appropriate in the course of patient care.
-
CCHCS shall produce reports for the management of individual patients and patient populations, such as patient registries, patient profiles, and patient summaries, and health care staff shall use these reports regularly for purposes of care management, population management, and other patients care activities.
-
-
Continuous Improvement. At all levels of the organization (statewide, regional, institution, and Care Team or program), leaders shall be responsible for establishing a culture of teamwork, continuous learning, and innovation. Activities to continuously evaluate and improve health care processes shall be incorporated into the day-to-day work of health care staff.
-
Leaders shall champion cultural change, as well as specific improvement strategies, such as initiatives from the institution’s annual improvement plan.
-
Responsibility for conducting improvement activities shall be shared by all staff, from leadership to team members.
-
CCHCS shall establish an effective communication system to keep staff at all levels of the organization appraised of improvement priorities, organizational goals, and performance evaluation findings.
-
CCHCS shall use data and statistical tools to provide Care Teams with feedback about their performance in critical health care processes and the health outcomes of patients within their assigned panel.
-
-
-
Purpose
-
To establish a standardized and integrated care model that organizes and delivers core primary care functions to improve:
-
Quality of care and patient outcomes.
-
Efficiency and value of care.
-
Patient and staff satisfaction.
-
Adherence to legal and regulatory requirements.
-
-
Responsibility
-
Statewide
-
CCHCS and CDCR departmental leadership, at all levels of the organization shall ensure administrative, custodial, and clinical systems are in place and appropriate tools, training, technical assistance, and levels of resources are available so that Care Teams can successfully implement the CCM.
-
The Director, Health Care Operations and Corrections Services, and Director, Health Care Services, are responsible for statewide planning, implementation, and evaluation of the CCM.
-
-
Regional
-
Regional Health Care Executives are responsible for the administration of this policy at the subset of institutions within an assigned region.
-
-
Institution
-
The Chief Executive Officer is responsible for implementation of this policy at the institution level.
-
-
-
References
-
United States Preventive Services Task Force
-
Agency for Healthcare Research and Quality – Patient Centered Medical Home Resource Center,
https://www.ahrq.gov/ -
California Association of Public Hospitals and Health Systems – Whole Person Care in California Public Hospitals and Health Systems
https://caph.org/publication/whole-person-care-in-californias-public-health-care-systems/ -
The Joint Commission Primary Care Medical Home Certification,
http://www.jointcommission.org/accreditation/pchi.aspx -
National Committee for Quality Assurance – Patient-Centered Medical Home Recognition,
http://www.ncqa.org/Programs/Recognition/Practices/PatientCenteredMedicalHomePCMH.aspx -
Commonwealth Fund – Safety Net Medical Home Initiative,
http://www.commonwealthfund.org/interactives-and-data/multimedia/videos/2011/safety-net-medical-home-initiative -
Robert Wood Johnson Foundation / Improving Chronic Illness Care – The Chronic Care Model, http://www.improvingchroniccare.org/index.php?p=About_US&s=6
-
Robert Wood Johnson Foundation / Improving Chronic Illness Care – Reducing Care Fragmentation, http://www.improvingchroniccare.org/downloads/reducing_care_fragmentation.pdf
-
-
Revision History
-
Effective: 07/2015
Revised: 08/2020
-
3.1.2 Scope of Patient Care Services
-
Procedure Overview
-
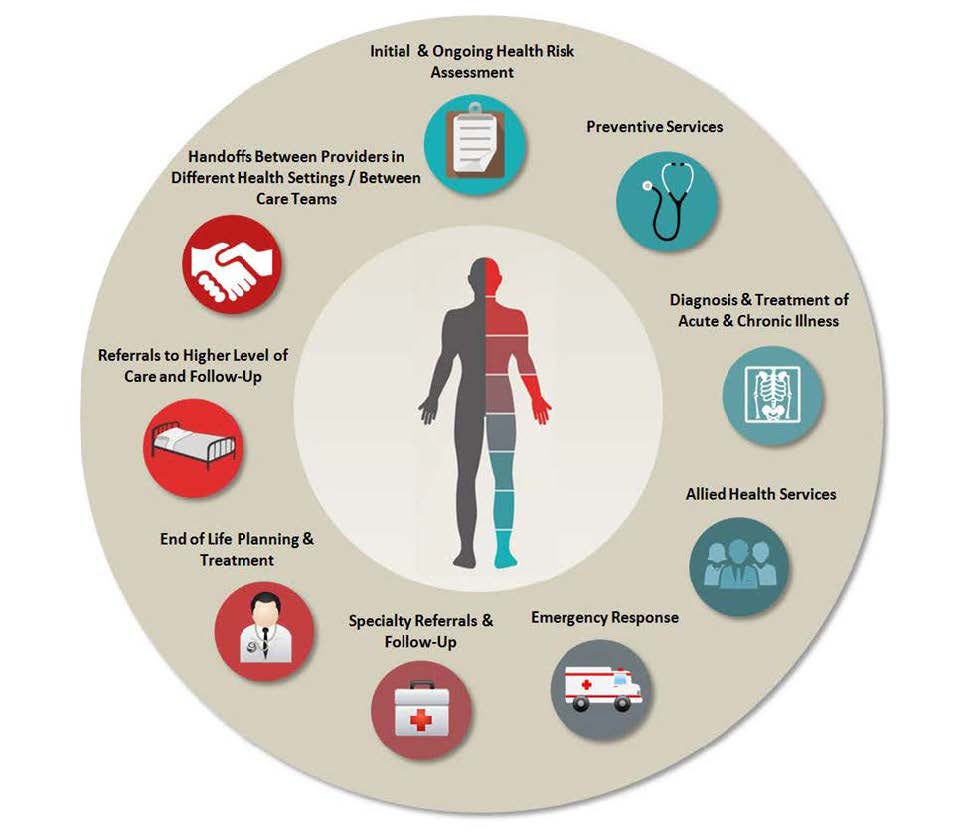
Under the Complete Care Model (CCM), an assigned Primary Care Team (PCT) serves as the center of each patient’s Health Home, directly delivering the majority of dental, medical, mental health, and nursing services and coordinating all care that falls outside the team’s scope of services (refer to the figure below). Applicable standards for the delivery and coordination of the services outlined in this procedure can be found in existing California Correctional Health Care Services (CCHCS) and Division of Health Care Services policies and procedures. PCTs remain responsible for adhering to these standards under the CCM (refer to Appendix 1, Services and/or Coordinated by the Primary Care Team and Associated Standards in the Health Care Department Operations Manual).
-
Complete Care Model Patient Care Services
-
-
This procedure incorporates existing policies and procedures that describe the scope of primary care services provided and coordinated by the PCTs to fulfill their role as the center of each patient’s Health Home by following the CCM. Refer to the Health Care Department Operations Manual and the Mental Health Services Delivery System Program Guide. In addition, the PCTs shall utilize existing decision support tools such as Care Guides, Nursing Protocols, Order Sets, Standing Orders, etc., when providing services.
-
-
Responsibility
-
Statewide
-
California Department of Corrections and Rehabilitation and CCHCS departmental leadership at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial, and clinical systems are in place and appropriate tools, training, technical assistance, and levels of resources are available so that Care Teams can successfully implement the Scope of Patient Care Services Procedure.
-
-
Regional
-
Regional Health Care Executives are responsible for implementation of this procedure at the subset of institutions within an assigned region.
-
-
Institutional
-
The Chief Executive Officer (CEO) has overall responsibility for implementation and ongoing oversight of a system to provide management of the Scope of Patient Care Services. The CEO delegates decision-making authority to designated Institutional Health Care Executives for daily operations of the Scope of Patient Care Services Procedure and ensures adequate resources are deployed to support the system.
-
The CEO and all members of the institution leadership team are responsible for ensuring all necessary resources are in place to support the successful implementation of this procedure at all levels including, but not limited to, the following:
-
Institution level
-
Patient panel level
-
Patient level
-
-
The CEO and all members of the institution leadership team shall ensure access to and utilization of equipment, supplies, health information systems, patient registries and summaries, and evidence-based guidelines.
-
The CEO and all members of the institution leadership team as a part of the Quality Management process on an ongoing basis shall:
-
Review and compare institutions’ PCT performance, including the overall quality of services, health outcomes, assignment of consistent and adequate resources, utilization of Dashboards, Master Registries, Patient Summaries, decision support tools and address issues pertaining to delivery of the Scope of Patient Care Services.
-
Provide PCT members with adequate resources, including protected time, staffing, physical plant, information technology, and equipment/supplies to accomplish daily tasks.
-
Work with custody staff to minimize unnecessary patient movement and ensure appropriate escort and transport.
-
-
The Chief Nurse Executive is responsible for the overall daily clinic operations and ensuring that the institution has designated supervisors to monitor clinic operations including, but not limited to:
-
Efficiency.
-
Coordination.
-
Supplies.
-
Equipment.
-
Physical plant issues.
-
Scheduling and access to care on a daily basis.
-
Identifying and addressing or elevating concerns regarding barriers.
-
-
-
-
Procedure
-
Services Delivered and/or Coordinated by the Primary Care Team
-
The PCT is responsible for coordinating care for patients within the assigned panel and pulling in other health care staff as necessary to meet the needs of the patient. The PCT identifies services that are outside of the team’s purview, coordinates patients’ access to necessary services, and ensures appropriate follow up after services have been provided by other providers in other health care settings.
-
The PCT provides the full scope of primary care services to patients within an assigned patient panel including, but not limited to:
-
Care coordination.
-
Initial and ongoing health risk assessment.
-
Preventive services, such as health screenings, health promotion, and health maintenance services.
-
Diagnosis and treatment of acute and chronic illness.
-
Allied Health Services required for diagnosis and treatment of acute and chronic illness such as diagnostic testing, medication administration, nutritional services, and health care equipment and supplies.
-
Emergency response.
-
Planning for end-of-life care such as advance directives, Physicians’ Orders for Life-Sustaining Treatment, and palliative care.
-
Specialty referrals and follow up.
-
Referrals to higher levels of care and follow up.
-
Facilitating handoffs between providers in different health care settings or between Care Teams to ensure the best possible care for the patient and continuity of planned care, pending appointments or services, medications, medical equipment and supplies, and all other necessary treatment.
-
-
-
Appendices
-
Appendix 1: Services Delivered and/or Coordinated by the Primary Care Team and Associated Standards in the Health Care Department Operations Manual
-
-
References
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.1, Complete Care Model
-
Health Care Department Operations Manual, Chapter 3, Article 3, Dental Care
-
California Department of Corrections and Rehabilitation, Mental Health Services Delivery System Program Guide, 2009 Revision
-
National Committee for Quality Assurance – Patient-Centered Medical Home Recognition http://www.ncqa.org/Programs/Recognition/Practices/PatientCenteredMedicalHomePCMH.aspx
-
The Joint Commission Primary Care Medical Home Certification
http://www.jointcommission.org/accreditation/pchi.aspx -
Agency for Healthcare Research and Quality – Patient Centered Medical Home Resource Center https://www.ahrq.gov/
-
Commonwealth Fund – Safety Net Medical Home Initiative
http://www.commonwealthfund.org/interactives-and-data/multimedia/videos/2011/safety-net-medical-home-initiative -
Robert Wood Johnson Foundation / Improving Chronic Illness Care – The Chronic Care Model http://www.improvingchroniccare.org/index.php?p=About_US&s=6 and Reducing Care Fragmentation http://www.improvingchroniccare.org/downloads/reducing_care_fragmentation.pdf
-
-
Revision History
-
Effective: 06/2016
Revised: 03/2017
-
-
Appendix 1: Services Delivered and/or Coordinated by the Primary Care Team and Associated Standards in the Health Care Department Operations Manual
Service Type Applicable Standards Initial and Ongoing Health Risk Assessment ∙ Reception Center (HCDOM Section 3.1.9)
∙ Health Care Transfer (HCDOM Section 3.1.10)
∙ Comprehensive Accommodation (HCDOM Section 3.6.2)
∙ Medical Classification System (HCDOM Section 1.2.14)
∙ Care Team and Patient Panels (HCDOM Section 3.1.3)
∙ Scheduling and Access to Care (HCDOM Section 3.1.6)
∙ Population and Care Management Services (HCDOM Section 3.1.7)Preventive Services ∙ Public Health and Infection Control (HCDOM Chapter 3, Article 8)
∙ Patient Health Care Education (HCDOM Section 3.1.4)
∙ Patient Care During Pregnancy and Childbirth (HCDOM Section 3.1.17)
∙ Dental Services (HCDOM Chapter 3, Article 3)Diagnosis and Treatment of Acute and Chronic Illness ∙ Scheduling and Access to Care (HCDOM Section 3.1.6)
∙ Medication Management (HCDOM Chapter 3, Article 2)
∙ Clinical Guidelines (HCDOM Section 1.2.2)
∙ Gender Dysphoria Management (HCDOM Section 4.1.7)
∙ Hepatitis C Management
∙ Nursing Services/Protocols
∙ Dental Services (HCDOM Chapter 3, Article 3)Allied Health Services ∙ Laboratory Services (HCDOM Section 3.1.15)
∙ Medical Imaging Services (HCDOM Section 3.1.14)
∙ Outpatient Dietary Intervention (HCDOM Section 3.1.12)
∙ Durable Medical Equipment and Medical Supply (HCDOM Section 3.6.1)
∙ Pharmacy Services (HCDOM Chapter 3, Article 5)Emergency Response ∙ Emergency Medical Response (HCDOM Chapter 3, Article 7) Specialty Referral and Follow-Up ∙ Outpatient Specialty Services (HCDOM Section 3.1.12)
∙ Utilization Management Program (HCDOM Section 1.2.15)
∙ Physician Orders for Life Sustaining Treatment (HCDOM Section 2.4.2)End-of-Life Planning and Treatment ∙ Palliative Care and Treatment (HCDOM Section 3.1.18)
∙ Advance Directive for Health Care (HCDOM Section 2.4.1)
∙ Physician Orders for Life Sustaining Treatment (HCDOM Section 2.4.2)Referrals to Higher Levels of Care and Follow-Up ∙ Health Care Transfer (HCDOM Section 3.1.10)
∙ Specialized Health Care Housing (HCDOM Section 3.1.11)Handoffs Between Providers in Different Health Settings/Between Care Teams ∙ Health Care Transfer (HCDOM Section 3.1.10) -
This list is not a complete listing of all associated policies and procedures.
-
3.1.3 Care Teams and Patient Panels
-
Procedure Overview
-
The Complete Care Model Policy maintains a Patient-Centered Health Home for each patient consisting of an interdisciplinary Care Team responsible for delivering comprehensive care for patients in accordance with their health care needs, directly providing the majority of clinical care services, and coordinating care when patients require services beyond what the Care Team provides.
-
This procedure defines interdisciplinary Care Teams, identifying the team members and outlining their roles and responsibilities. In addition, this procedure outlines the process for assigning each patient to a Care Team, presents the expectations for notification to patients and panel management, and introduces daily and twice-monthly forums that Care Teams shall use to monitor and manage both clinic operations and changes in the patient panel.
-
-
Responsibility
-
Statewide
-
California Correctional Health Care Services (CCHCS) and California Department of Corrections and Rehabilitation (CDCR) departmental leadership at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial, and clinical systems are in place and appropriate tools, training, technical assistance, and levels of resources are available to ensure the scheduling system is successfully implemented and maintained.
-
-
Regional
-
Regional Health Care Executives are responsible for the administration of this procedure at the subset of institutions within an assigned region.
-
-
Institutional
-
The Chief Executive Officer (CEO) has the overall responsibility for implementation and ongoing oversight of the scheduling system at the institution and patient panel level. The CEO delegates decision-making authority to the Chief Nurse Executive (CNE) for daily operations of the scheduling system and ensures adequate resources are deployed to support the system including, but not limited to, the following:
-
Ensuring access to and utilization of equipment, supplies, health information systems, patient registries, patient summaries, and evidence-based guidelines.
-
Assigning patients to a Care Team.
-
Maintaining a list of the core members of each Care Team which shall be available to all institutional staff. Patients shall be informed of their assigned Care Team members at intake and/or upon request.
-
Ensuring consistent Care Team staffing with a back-up system for core members.
-
Providing Care Team members with the information they need during huddles (e.g., Huddle Report).
-
Ensuring protected time for Care Teams to hold daily huddles.
-
Documenting and tracking huddle actions and attendance.
-
Ensuring that at least twice-monthly, each Care Team conducts a Population Management Working Session utilizing tools such as dashboards, patient registries, patient summaries, and Electronic Health Record System (EHRS) tools to address concerns related to potential gaps in care and improve patient outcomes.
-
Adequately preparing new Care Team members to assume team roles and responsibilities.
-
Assessing competence of existing Care Team members.
-
Updating procedures, roles and responsibilities as new tools and technology become available.
-
Reviewing/comparing institution Care Team performance, including the overall quality of services, health outcomes, assignment of consistent and adequate resources, utilization of dashboards, patient registries, patient summaries, and other decision support tools and address issues as necessary.
-
Providing Care Team members with adequate resources, including protected time, staffing, physical plant, information technology, and equipment/supplies to accomplish daily tasks.
-
Working with custody staff to minimize unnecessary patient movement that results in changes to a patient’s panel assignment.
-
Ensuring, in collaboration with the Warden, that the institution establishes a Local Operating Procedure by which priority health care ducats are issued and delivery by custody staff is verified and documented.
-
Requiring institution leadership to establish a back-up system to ensure scheduling queues are managed when Scheduling Support staff are on leave or otherwise unable to meet daily monitoring requirements.
-
-
TheCEO, or designee, and all members of the institution leadership team are responsible for establishing an organizational culture that promotes teamwork across disciplines.
-
The CNE, or designee, is responsible for:
-
The overall daily operations of the scheduling system for health care within the EHRS.
-
The coordination of health care between health care scheduling systems (e.g., outside specialty appointments).
-
Oversight and management of the scheduling processes and resources, including personnel.
-
Ensuring that the institution has a designated scheduling lead to monitor scheduling processes on a daily basis and identify and address or elevate barriers to access.
-
Ensuring that Scheduling Support staff is available for all clinical areas.
-
-
The Chief Medical Executive (CME), or designee, is responsible for the overall medical management of patients and ensures resources are available to meet the needs of the population.
-
The Supervising Registered Nurse and Chief Physician and Surgeon or CME, or designees, shall meet as needed to review the Care Team’s performance, including the overall quality of services, health outcomes, level of care utilization and shall utilize dashboards, patient registries, patient summaries, and all electronic decision support tools to address or elevate issues as necessary.
-
-
-
Procedure
-
Patient Panels
-
Institution leaders shall adopt methods to promote a consistent, ongoing relationship between patients and their Care Teams to achieve operational efficiency; ensure timely access to care; optimize movement and escort capabilities; balance workload; address patient acuity and complexity to support patients in the management and organization of their care.
-
Each patient shall be assigned to a Care Team and be notified of the Care Team assignment.
-
Assignment to a Care Team may be organized in a variety of ways, as dictated by the needs of patients and the institution including, but not limited to, assignment by the following:
-
Housing unit.
-
Alphabetical roster.
-
Last two digits of CDCR number.
-
Custodial factors.
-
Mental health program assignment.
-
Medical factors and other special patient needs.
-
-
All Care Teams shall have access to the master registry.
-
Institutions shall communicate any change in their strategy for panel assignment to headquarters to preserve the accuracy and reliability of the master registry.
-
The Care Team is responsible for tracking the status of the assigned patient panel and shall monitor the master registry and Huddle Report daily, identifying changes to the assigned patient panel and communicating changes to team members using the daily huddle or other appropriate forums.
-
-
-
Care Team Members
-
Care Team Composition
-
At a minimum, each Care Team shall consist of the following core members:
-
Primary Care Providers (PCPs).
-
Primary Care – Registered Nurse (RN).
-
Provider support staff (e.g., Certified Nursing Assistants, Medical Assistants, or Licensed Vocational Nurses [LVN]).
-
Medication Administration Nurse (LVN/Psychiatric Technician).
-
Care Manager – RN.
-
LVN Care Coordinator.
-
Administrative support staff.
-
Other members, as needed.
-
-
Other team members may be added to the Care Team on a per-patient basis. For example, the Care Team would include a dentist and other dental staff when planning, delivering, and coordinating services for a patient with complex dental needs. The range of possible Care Team members includes, but is not limited to, custody staff, pharmacy staff, dietitians, specialists, specialty nursing staff, laboratory or imaging staff, and therapists (e.g., occupational, recreational, respiratory, and other types of therapists).
-
Depending on the mission of the institution and the needs of the patient panel, members may be added to the Care Team as core members. For example, if there is a high proportion of patients with serious mental illnesses in a patient panel, a Primary Mental Health Clinician and/or Primary Psychiatrist may serve as members of the Care Team.
-
-
Continuity in Team Membership
-
Institutions shall avoid unnecessary changes in the membership of the Care Team to reduce disruptions in care. Individual changes in Care Team membership do not require formal notice to patients.
-
The institutions’ CME, or designee, shall ensure the Care Team has assigned and available Care Team members at all times with minimal disruptions to continuity.
-
The institution CEO, CME, and CNE, or designees, shall ensure each core member of the Care Team:
-
Is assigned and available.
-
Has a consistent back-up staff member.
-
Has a coordinated schedule to optimize continuity.
-
Has scheduled hours of work in alignment with clinic operational needs.
-
Has scheduled work hours and hours of clinic operation in alignment for the entire Care Team.
-
-
Contingency plans shall be in place to optimize continuity in the event of scheduled absences and, whenever possible, in the event of unscheduled absences. Back-up designations shall be included in the Care Team.
-
-
In recognition that communication and collaboration between Care Team members is greatly facilitated by being present in the same clinic space at the same time, institution leaders shall:
-
Review the schedules and work locations of Care Team members, at least annually, and take action to optimize the number of hours that core members work in the clinic together and have access to patients.
-
Ensure that Care Team members are located in close proximity to each other when they are providing services to patients, wherever possible.
-
-
-
Roles and Responsibilities of the Care Team
-
The entire Care Team shall be accountable for the outcomes of patients in the assigned patient panel, and each Care Team member shall be responsible to ensure efficiency and effectiveness of the Care Team (Refer to Appendix 1, Care Team Roles and Responsibilities).
-
All Care Team members shall be required to:
-
Establish and maintain professional, effective, and therapeutic relationships with patients.
-
Create a climate of mutual respect in which individual Care Team members feel comfortable sharing their concerns about unsafe, ineffective, or inefficient processes, systems, or operations, including the inappropriate management of individual patients.
-
Promote clear and frequent communication between Care Team members.
-
Participate fully in the Care Team’s collective efforts to manage the patient panel, including identifying necessary patient care activities and allocating work among Care Team members.
-
Maintain an up-to-date knowledge of trends, best practices, and guidelines in clinical practice and operations as relevant to each Care Team member’s respective licensure.
-
Evaluate the quality of clinic processes and services in the course of day-to-day work and collaborate with other Care Team members to investigate and resolve quality problems.
-
Promote a safe, effective, efficient, and collaborative work environment.
-
-
Documentation of patient care and the patients’ response to care is essential for effective communication between health care providers and providing quality health care. To ensure accurate recording of patient care activities and to ensure the transfer of information between the members of the interdisciplinary care team, health care staff shall:
-
Document all patient contacts, interventions, observations, care and treatments provided and the results of the care and treatment in the health record at the time of service.
-
Record documentation using the Subjective, Objective, Assessment, Plan, Education format or use other forms of documentation such as narrative charting, charting by exception, focused assessment, etc., as indicated by the clinical situation. However, all documentation shall contain subjective and objective patient care data at a minimum regardless of format.
-
Ensure that all documentation complies with the documentation standards contained in the Health Care Department Operations Manual, Chapter 2, Article 3, Health Information Management.
-
-
-
Daily Care Team Huddle
-
The Care Team shall convene each business day in a Care Team Huddle to:
-
Monitor changes to the patient panel, such as transfers to and from the panel, and take action to continue and/or coordinate care for these patients.
-
Discuss recent health care events, problems and trends that impact patients within the assigned patient panel, identify services that may need to be provided to patients, and determine how and when services will be provided including, but not limited to the following:
-
Unscheduled Triage and Treatment Area visits.
-
Medical holds.
-
Transfers to and from higher levels of care.
-
Pending consultations and specialty services requests.
-
New patients assigned to the Care Team. Once seen in the clinic, the RN or PCP shall order a follow up based on their chronic care conditions.
-
Abnormal laboratory findings.
-
High risk patient/care management issues.
-
Mental health issues (e.g., self-injurious behavior, suicidal/homicidal ideation, coordination of testing procedures).
-
Medication line issues, including specialty medications that require coordination with offsite (e.g., chemotherapy, Narcotic Treatment Program methadone).
-
Polypharmacy.
-
-
Manage day-to-day clinic operations, including preparation for that day’s encounters, conferring with custody, addressing security or construction impacts to clinic processes, and planning coverage of clinic services while staff are on leave.
-
Discuss daily clinical operational problems, such as the following:
-
Episodic care triage.
-
Same day and next day relevant health information availability (e.g., diagnostic study reports, consultation notes, and discharge summaries) and add-on appointments.
-
Review and resolution of scheduling concerns.
-
Potential barriers to care, including lockdowns, restricted movement, fog lines, backlogs, and other considerations.
-
Staffing issues, such as upcoming vacation, mandatory training, or other events affecting availability of staff.
-
Supply/resource issues.
-
Review and discussion of the Care Team’s performance with respect to targeted disease management and preventive service metrics.
-
Ongoing evaluation and improvement.
-
-
-
Institutional leadership shall establish a standard start time for Care Team Huddles to ensure that Care Team members have protected times for huddles and that huddles begin on time.
-
Huddle Preparation
-
Institutional leadership shall work with Care Team members to:
-
Incorporate the use of the Patient Summary.
-
Use a standard Daily Huddle Script and Daily Huddle Report that prompts Care Team members to address topics mandated in this procedure (Refer to the Daily Huddle Script and Daily Huddle Report).
-
Determine who shall be responsible to have the Huddle Report at each daily huddle, and what other information shall be provided to the Care Team in advance of the huddle.
-
-
Huddle Documentation
-
Care Teams shall document patients and issues discussed during the Primary Care Huddle and actions taken as a result, monitoring to ensure that necessary follow up has occurred.
-
Each Care Team shall be responsible for monitoring the Daily Huddle Script, and Training Participation Sign-in Sheet.
-
-
-
Monitoring and Sustainability
-
Institutional leadership shall designate a standing committee reporting to the local Quality Management Committee for oversight of the Complete Care Model monitoring activities. The Care Team shall:
-
Take corrective action to resolve and/or elevate concerns identified in the review.
-
Review and action shall be documented and forwarded to the designated committee.
-
-
The CEO and institutional leadership team shall establish an ongoing monitoring program to periodically assess the quality of Care Team services and adherence to this procedure including, but not limited to:
-
Accuracy and efficacy of panel assignment strategies.
-
Stability of Care Team staffing and use of back-up systems.
-
The amount of time each day that all Care Team members are working in the clinic together and any associated physical plant issues.
-
Inclusion of other team members/disciplines to manage patient care.
-
Care Team Huddle attendance.
-
Frequency, quality, and timeliness of daily Primary Care Huddles.
-
Documentation of Primary Care Huddle activities and necessary follow up.
-
Frequency and quality of Population Management Working Sessions.
-
Adverse events or barriers linked to Care Team processes described in this procedure.
-
-
The CEO and institutional leadership team shall utilize or implement a monitoring process to assess the Care Team members and staff supporting Care Team processes. The monitoring process shall include, but is not limited to, feedback about skills required to successfully provide or support primary care services such as:
-
Clinical skills (e.g., history-taking, physical examinations, assessment, and treatment planning).
-
Adherence to policy guidelines, protocols, and decision support tools.
-
Recognition of patient care needs that fall outside the scope of what is provided by the Care Team and appropriate and timely referral.
-
Management of handoffs as patients move from one Care Team to another or across levels of care.
-
Care management of patients who are high risk or otherwise clinically complex.
-
Population and panel management, including provision of preventive services and managing subpopulations with specific chronic diseases.
-
Self-management planning and patient education.
-
Effective communication.
-
Optimizing access to care through use of co-consultation, appointment bundling, same-day appointments, and other strategies.
-
Redesigning clinic processes to increase efficiency and use team members to the full extent of their licensure.
-
Identification, analysis, and resolution of quality problems, including use of data to evaluate performance and investigate problems.
-
Application of available patient management tools, including patient registries and EHRS.
-
Overall contribution to the Care Team and a culture that promotes teamwork.
-
-
-
Training and Decision Support
-
The CEO and institutional leadership team shall establish an orientation and training program to ensure that all staff serving as members of a Care Team or supporting Care Team functions fully understand their roles and responsibilities prior to assuming their duties. Elements of the program shall include, but are not limited to review of:
-
Expectations in this procedure.
-
Any changes to local Care Team processes.
-
National health care industry advances pertinent to the Patient-Centered Health Home.
-
New information systems or technology that may increase the efficiency or effectiveness of Care Team processes or forums.
-
Updates in clinical practice, including new CCHCS guidelines, standing orders, nursing protocols, industry best practices, and findings in clinical literature.
-
Training needs.
-
-
-
Appendices
-
Appendix 1: Care Team Roles and Responsibilities
-
-
References
-
Health Care Department Operations Manual, Chapter 2, Article 3, Health Information Management
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.6, Population and Care Management Services
-
The Joint Commission Primary Care Medical Home Certification, http://www.jointcommission.org/accreditation/pchi.aspx
-
National Committee for Quality Assurance – Patient-Centered Medical Home Recognition, http://www.ncqa.org/Programs/Recognition/Practices/PatientCenteredMedicalHomePCMH.aspx
-
Agency for Healthcare Research and Quality – Patient Centered Medical Home Resource Center, http://www.ahrq.gov/
-
Commonwealth Fund – Safety Net Medical Home Initiative, http://www.commonwealthfund.org/interactives-and-data/multimedia/videos/2011/safety-net-medical-home-initiative
-
Robert Wood Johnson Foundation / Improving Chronic Illness Care – The Chronic Care Model, http://www.improvingchroniccare.org/index.php?p=About_US&s=6, and Reducing Care Fragmentation, http://www.improvingchroniccare.org/downloads/reducing_care_fragmentation.pdf
-
-
Revision History
-
Effective: 06/2016
Revised: 12/2020
-
-
Appendix 1: Care Team Roles and Responsibilities
Care Team Roles and Responsibilities Primary Care Provider a. Attend and actively participate in the daily huddle.
b. Diagnose and manage the patients’ episodic illnesses, chronic conditions, preventive care, and their complex needs.
c. Order and coordinate patient care services including, but not limited to, specialty and higher level of care.
d. Support Transitional Services Team (Resource Registered Nurse [RN]) in transition planning for complex medical conditions.Mental Health Clinician and/or Psychiatrist a. When indicated, attend and actively participate in Primary Care Huddles to provide mental health input into patient behaviors, compliance, and treatment options as they relate to the patient’s mental health condition.
b. Coordinate mental health care, as needed.
c. Provide relevant mental health history.
d. Support Transitional Services Team (Resource RN) in transition planning for complex mental health conditions.Dentist a. When indicated, attend and actively participate in Primary Care Huddles to provide input concerning dental treatment needs.
b. Coordinate patient care services including, but not limited to, oral surgery services, lab tests, diagnostic imaging and diagnostic procedures.
c. Consult with other care team members on the patient’s episodic illnesses, chronic conditions, preventive care needs, and mental health conditions.
d. Provide input on dental infections/conditions, refusals of dental care and planned dental care that may affect other aspects of the patient’s overall health care needs.Primary Care Provider Support Staff a. Attend and actively participate in the daily huddle.
b. Prepare patients for visits (e.g., vital signs, weights, gathering specialty reports and diagnostic results, other health information preparation).
c. Conduct/perform Point-of-Care testing and administration of treatments in accordance with licensure/certification.
d. Assist with tracking and access to Durable Medical Equipment.Primary Care RN a. Attend and actively participate in the daily huddle.
b. Manage the patient’s episodic illnesses, chronic conditions, preventive care needs, and their complex care management using established protocols and other decision support.
c. Advocate for the patient.
d. Coordinate the patient care services for the designated patient panel.
e. Manage medication for patients assigned to the team.
f. Provide patient education.
g. Conduct/perform Point-of-Care testing.
h. Participate in discharge planning.Licensed Vocational Nurse Care Coordinator a. Attend and actively participate in the daily huddle.
b. Advocate for the patient.
c. Monitor designated patient panel registries and report any changes to the team members.
d. Coordinate the patient care services for the designated patient panel.
e. Manage medication for patients assigned to the team.
f. Provide patient education.
g. Conduct/perform Point-of-Care testing.
h. Participate in discharge planning.Supervising Registered Nurse (SRN) II a. Attend and actively participate in the daily huddle as indicated.
b. Oversight of key clinical processes including, but not limited to, scheduling and medication management, and management of refused orders inbox.
c. Audit compliance for a variety of nursing measures including, but not limited to, quality of care.
d. Identify opportunities for improvement.
e. Communicate staffing needs.
f. Coordinate with custody to mitigate barriers affecting access to health care.
g. Facilitate conflict resolution.
h. Provide clinical support as indicated.Primary Care Team Office Technician a. Attend and actively participate in the daily huddle.
b. Ensure all patients are appropriately scheduled.
c. Ensure access to care barriers are made known to the full Care Team.
d. Retain records from daily huddles.
e. Prepare information for daily huddles.
f. Maintain attendance records for daily huddles.
g. Schedule patients in the scheduling system in accordance with policy timeframes.
h. Ensure Care Team workload is balanced for scheduled patients.
i. Maintain a current and accurate schedule for the clinic.
j. Support improvements in the design of the clinic schedule to optimize efficiency and access to care, such as open access scheduling, or consolidation of multiple appointments for the same patient into a single encounter.Transitional Services Team (Resource RN) a. When indicated, attend the Daily Huddle to provide pertinent information to Care Team members regarding transitional planning for qualifying complex patients, and communicate any needed support from Primary Care Team (PCT) members.
b. Assessing identified patients with complex care needs across all health care domains, and liaise with other internal and external stakeholders to ensure care needs are addressed prior to release to the community, and when possible, prior to entry into the prison system.
c. Support the PCT when transition planning to the community involves specialty health care and rehabilitative services.
d. Coordinate with community health care and rehabilitative agencies to ensure continuity of care for qualifying complex patients.
e. Coordinate care with prisons and jails for patients departing on a temporary basis, such as transfers related to court appearances, higher levels of care for medical, mental health, or dental reasons.
f. Communicate identified needs with parole offices and probation offices and arranging specialize care services with community health care providers, when and where appropriate.Medication Administration Nurse a. Attend and actively participate in the daily huddle when possible.
b. Ensure timely delivery of prescribed medications to patients on the panel.
c. Alert the Care Team of adherence issues and adverse medication events.
d. Alert pharmacy or the SRN II when prescribed medications are unavailable.
e. Report medication errors.
f. Alert the SRN II to medication administration access issues.
g. Reconcile medication orders in the Electronic Health Record System.
h. Perform routine vitals that are associated with medications.
i. Conduct/perform Point-of-Care testing as associated with medication delivery.
3.1.4 Patient Education
-
Policy
-
California Correctional Health Care Services (CCHCS) staff shall provide clinical and health education to patients regarding disease prevention, recommended treatment modalities, and available health care resources at all stages of their confinement within any California Department of Corrections and Rehabilitation (CDCR) facility. Patient education shall be a continuous and ongoing process designed to educate and inform the patient beginning with the patient’s arrival at a Reception Center and continuing throughout incarceration.
-
The CCHCS/CDCR Patient Education Program supports the Complete Care Model by recognizing that the patient is an active partner in their own health care. The patient, their Primary Care Team, and other health care providers determine the most appropriate health care goals, interventions, and outcomes based on the patient’s health care needs and personal objectives with the understanding that an informed patient delivers an improved patient outcome and reduces overall morbidity and mortality.
-
Patient education within CCHCS consists of two main components which are Clinical Patient Education and Health Education. Clinical Patient Education is a planned, systemic, and sequential program of teaching provided to patients in a clinical environment based on the patient’s assessment, evaluation, diagnosis, prognosis, individual needs, and care requirements pursuant to the patients’ health status and desired outcomes. Health Education is provided to all patients to promote general health and wellness, disease prevention, and is designed to change and improve health behaviors within the patient population.
-
-
Purpose
-
To provide education that promotes wellness and empowers patients to actively participate in their disease management and prevention.
-
-
Responsibilities
-
Statewide
-
CDCR and CCHCS departmental leadership at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial, and clinical systems are in place, and appropriate tools, training, technical assistance, and levels of resources are available so that health care staff can successfully implement and maintain the Patient Education Program.
-
The Undersecretary, Health Care Services, CDCR, and the Directors of CCHCS are responsible for statewide planning, implementation, and evaluation of the Patient Education Program.
-
The Undersecretary, Health Care Services, CDCR, and the Directors of CCHCS shall designate a statewide committee with responsibility for the oversight of all aspects of the Patient Education Program within CCHCS/CDCR. The designated committee shall be multidisciplinary and consist of, at a minimum, the following members, or their designees, the Deputy Directors of Medical, Nursing, Mental Health and Dental Services, Pharmacy, Ancillary and Allied Health Services, and the Regional Health Care Executives (RHCEs). The committee shall be responsible for ensuring appropriate, standardized patient education material is developed and available for patients statewide and at all levels of care.
-
-
Regional
-
RHCEs are responsible for implementation of this policy and procedure at the subset of institutions within an assigned region.
-
-
Institutional
-
The Chief Executive Officer (CEO) has overall responsibility for implementation and ongoing oversight of a system to provide management of Patient Education Programs in their institution. The CEO delegates decision making authority to designated health care executives for daily operations of the Patient Education Program and ensures adequate resources are deployed to support the program.
-
The CEO and all members of the institution leadership team are responsible for ensuring resources are in place to support the successful implementation of this procedure at all levels, which include access to and utilization of equipment, supplies, health information systems, patient registries and summaries, and evidence-based guidelines.
-
Institution-specific local operating procedures shall be developed and implemented based upon the tools provided by statewide health care leadership.
-
Each institution shall designate a subcommittee in writing that has responsibility for the oversight and coordination of all Patient Education Programs within the institution. The designated subcommittee shall report to the institution’s Quality Management Committee. At a minimum, the designated subcommittee shall ensure that patient education (clinical and peer mentoring) is provided based on the identified needs of their patient population by:
-
Identifying high-risk groups within their patient population.
-
Facilitating health care events within CDCR and/or the Local Community (e.g., Substance Abuse Prevention, Influenza Campaigns, Heat Injury Prevention).
-
Reviewing clinical and health education needs identified during discussions with Patient Representative Groups (i.e., Men’s Advisory Council [MAC], Women’s Advisory Council [WAC], and Inmate Family Council [IFC]).
-
Identifying needs based on reviews of patient grievances.
-
Identifying needs based on internal and external audits and reviews (e.g., Patient Safety, Medication Administration Process Improvement Program, Office of Internal Affairs, or Prison Law Office visits).
-
Holding discussions with CDCR partners (i.e., custody, Inmate Education Services and Vocational/Prison Industry Authority [PIA] Training and Services).
-
-
Each institution shall ensure that clinical education, health education, and Patient Education Programs are:
-
Coordinated.
-
Mutually supportive and meet the needs of the patient population as a whole.
-
Address the needs of identified high-risk patient populations as well as the health care needs and goals self-identified by the patient population.
-
-
-
-
Procedure Overview
-
This procedure provides guidelines for the development, utilization, provision, and documentation of health care education to CDCR/CCHCS patients. The purpose of the Patient Education Program is to promote wellness and empower patients to actively participate in disease prevention and management. This program aims to reduce morbidity/mortality and overall health care costs.
-
Patient education shall be provided to each patient within CDCR on a continuous and ongoing basis using processes designed to educate and inform the patient beginning with arrival at a Reception Center (RC) and continuing throughout incarceration. The CCHCS/CDCR Patient Education Program consists of two main components which are Clinical Patient Education and Health Education, both of which are an integral part and support of the Complete Care Model.
-
CCHCS/CDCR Patient Education Program shall support the goals of the Public Safety and Rehabilitation Act of 2016 (Proposition 57) through clinical and peer mentor programs designed to encourage and enable patients to understand and take responsibility for their health care needs and decisions, gain insight, and actively and fully participate in rehabilitative programs in preparation for their reintegration into the community once they complete their incarceration and transition to supervision.
-
-
Procedure
-
General Requirements
-
The Patient Orientation to Health Care Services Handbook shall be available in each institution law library and shall be provided to each patient within 14 business days upon arrival and upon patient request by the Receiving & Release (R&R) or Primary Care Nurses at any CDCR institution.
-
Each Standardized Nurse Protocol/Procedure and Care Guide shall include a patient education component, including printed material that shall be provided to the patient.
-
All patient education material shall be provided in a manner that can be used by the patient population to which it will be distributed.
-
The Statewide Patient Education Committee shall ensure printed materials are developed in both English and Spanish to the greatest extent possible. Languages other than English or Spanish shall have patient education material translated into the identified language.
-
To the extent possible, printed materials shall also be provided in formats, or by methods, accessible by patients with visual impairments in accordance with the Health Care Department Operations Manual (HCDOM), Section 2.1.2, Effective Communication Documentation. Institutions where the identified visually impaired population (DPV) exceeds 10% shall coordinate with the headquarters Patient Education Committee to have selected patient education material published in a large print version.
-
-
-
Reception Centers
-
Patient education shall begin with the patient’s arrival into CDCR custody at the RC. The RC R&R nursing staff shall:
-
Identify barriers to learning and ensure accommodations are documented in the health record. Examples include, but are not limited to:
-
Language.
-
Learning difficulties as documented in the patient’s transfer records or reported by the patient such as necessary effective communication (EC) accommodations (visual, hearing, speech).
-
-
Accommodations shall be provided based on the patient’s reported needs until formal testing can be performed (i.e., Hearing Testing or Test for Adult Basic Education testing, or Developmental Disabilities Program [DDP] screening).
-
-
Clinical Education Programs – Verbal and Written Patient Education
-
As described in (e)(1) above, CDCR/CCHCS shall develop and/or provide written material designed to support patient education, develop health literacy, improve the overall quality of life and health care outcomes, and reduce morbidity and mortality.
-
Individual face-to-face patient education
-
The most significant and effective method of patient education is direct verbal contact with a health care provider. Within CCHCS/CDCR, most patient education is performed during face-to-face clinical encounters. CCHCS/CDCR staff shall utilize every patient visit as an opportunity for therapeutic intervention and education.
-
CCHCS/CDCR staff who provide patient education shall document in the health record the education provided, the patient’s understanding of the information provided, and EC accommodations used (if needed).
-
-
-
Clinical Education Program – Institutional General Requirements
-
Each institution shall develop a Patient Education Program tailored to the identified needs of their institution. At a minimum, the program shall include the following elements:
-
R&R patient education (i.e., Patient Orientation to Health Care Services Handbook, Sick Call Process for the institution, the conduct of medication lines, access to health care processes, etc.)
-
Patient education during clinical contacts as needed
-
Nursing-led Therapeutic Groups (NL-TGs)
-
Orientation to available self-management and substance abuse programs
-
Peer Mentor Program
-
Woman’s Health Program (Central California Women’s Facility, California Institution for Women, and Folsom State Prison-Women’s Facility)
-
-
The patient education process shall begin upon arrival at the institution. The R&R nurse shall ensure that each patient requiring accommodation has access to the Durable Medical Equipment necessary for effective learning and communication (e.g., glasses, hearing aids, and batteries).
-
Patient education shall be a component of each health care visit. Documentation in the health record may include the following, as applicable:
-
Patient education provided
-
Printed material provided
-
Patient’s understanding of the education provided
-
EC process used (if applicable)
-
Topics include, but are not limited to:
-
Wellness & Prevention
-
Newly Diagnosed Disease (Episodic or Chronic)
-
Treatment Plan
-
Patient Goals
-
Medications and Treatments (Therapies)
-
Procedures, Diagnostic Tests, and Preventative Screening
-
Compliance/Adherence
-
Men’s/Women’s Health to include disease prevention and family planning
-
-
-
During Primary Care Team Huddles, Population Management Working Sessions, and/or mental health (MH) Interdisciplinary Treatment Team (IDTT), health care staff shall identify therapeutic groups which would be beneficial to include in a patient’s treatment plan or plan of care. This may include NL-TGs, mental health groups, self-management, etc. Any discussions and recommendations shall be documented in the health record.
-
Patient education for patients participating in the Mental Health Services Delivery System (MHSDS) shall receive patient education as described in the MHSDS Program Guide.
-
Patient education provided under this section shall supplement the clinical patient education provided under this procedure and be designed to meet the patient’s unique mental health needs as identified in their MH Interdisciplinary Treatment Plan.
-
The patient’s MH treatment team shall coordinate with the patient’s primary care team to meet the full range of clinical education needs through the development of adaptations to the educational process necessitated by clinical and mental health diagnosis (e.g., adapting IDTT plan based on visual problems, or Chronic Care Plan based on MH diagnosis, [i.e., schizophrenia or DDP status]).
-
-
Dental patients shall receive patient education as described in the HCDOM, Chapter 3, Article 3, Dental Care. Dental staff shall coordinate with the patient’s primary care and MH Health Treatment Teams (as applicable) to coordinate education activities and necessary adaptations to the standard dental education program and/or materials.
-
Where indicated, health care staff shall coordinate with other institutional staff to coordinate Patient Education Programs based on identified needs for rehabilitation and success such as:
-
Division of Rehabilitative Program staff for educational, vocational, and Cognitive Behavioral Therapy programs.
-
PIA to develop works skills.
-
Community Transition Program staff to coordinate pre-release activities and possible parole needs, etc.
-
-
-
Clinical Education Programs – Nursing-led Therapeutic Groups
-
Each patient shall be provided the opportunity to participate in nursing-led group activities that provide education on disease processes, positive health behaviors and health improvement, therapeutic interventions (clinical and self-directed), and are designed to improve the patients overall quality of life and health status.
-
Content development
-
An approved set of NL-TGs shall be developed to meet identified patient needs on a statewide basis.
-
Content and curricula shall be developed and standardized statewide. A multidisciplinary team shall develop NL-TGs under the direction of a Headquarters Chief Nurse Executive (HQ CNE).
-
Each NL-TG shall meet established guidelines and quality metrics as determined by the Statewide Patient Education Committee.
-
The HQ CNE shall designate a Nurse Consultant Program Review (NCPR) to lead the NL-TG development process. The NCPR shall collaborate with other disciplines to ensure content is accurate, relevant, and evidence-based.
-
NL-TGs will be written in a manner to qualify for Milestone, Rehabilitative Achievement, or other incarcerated person participation credits as delineated in California Proposition 57.
-
NL-TGs shall be separated into broad categories. Each category may have multiple individual lesson plans which support the overall category patient education goal. A list of approved NL-TGs shall be maintained under the direction of the designated HQ CNE.
-
-
NL-TG Scheduling
-
The institution CNE shall coordinate with the Community Resource Manager as outlined in the established workflow and collaborate with medical, mental health, dental, and custody staff to create a Master Schedule of Groups offered within the institution.
-
The institution CNE is responsible for approving and signing the Nursing Master Schedule and ensuring that it is included in the designated subcommittee’s discussion and minutes.
-
The institution CNE shall build upon the Nursing Group schedule utilizing the needs of the patient population being served to ensure the quality and variety of the NL-TGs as well as their relevance to the patient population.
-
NL-TGs may be scheduled and offered seven days per week on both second and third watch (See Appendix 1).
-
Groups shall be considered for all patients including those in the general population, patients with physical disabilities, cognitive impairments or substance use disorders, and all participants in the DDP and/or MHSDS.
-
Individual patient factors to be considered may include:
-
Patient classification and/or housing
-
Groups available on the master schedule
-
Times groups are scheduled
-
Any current behavior issues or concerns, any precipitating event
-
Perceived knowledge deficit
-
-
Institutional factors to be considered may include:
-
Whether classifications of patients (i.e., DDP and Enhanced Outpatient Program patients) are permitted to mix for therapeutic purposes
-
Physical plant limitations – available space
-
Custody support
-
-
-
-
Based on the category, the Primary Care Registered Nurse (PCRN) or Mental Health Registered Nurse (MHRN) shall determine specific groups to be provided from those that are made available in the statewide nursing education library available on Lifeline. When possible, suggestions for classes shall be discussed with the patient before scheduling. This discussion shall be documented in the health record.
-
The PCRN or MHRN shall place an order in the health record for each category of the group.
-
The patient shall be scheduled for a group encounter via the Health Care Priority Ducat Scheduling System by category and specific group content desired.
-
At the conclusion of each therapeutic group session, the nurse facilitator shall document participation, attainment of goals, and other pertinent information in each attendee’s health record. Attendance shall also be documented in the patient scheduling system.
-
-
Health Care Education – Peer Education Programs
-
Peer Health Care Education (PHE) is an effective means of providing health care education in a manner that is relevant and relatable for the individual patient. Each institution shall develop a Patient Education Program designed to improve overall health literacy based on the needs identified by their patient population.
-
PHE shall be developed collaboratively with input from each health care discipline and institutional stakeholders (e.g., custody, MAC/WAC, IFC).
-
Patients may be referred to a PHE group by any CDCR/CCHCS staff member, or they may request enrollment by submitting a CDCR 22, Inmate/Parolee Request for Interview, Item or Service, and/or CDCR 7362, Health Care Services Request, per the local operating procedure. The patient shall be notified of the results of the request in writing via institutional mail within 14 business days of the request.
-
General Requirements
-
The CDCR/CCHCS Patient Education Program is designed to provide ongoing peer mentoring and informal education for the management of chronic health issues (e.g., diabetes mellitus, pain, weight control) using an evidence-based curriculum.
-
The CDCR/CCHCS Patient Education Program is a partnership between health care, institutional staff, and the patients, each of whom are equally involved in the program’s development and implementation.
-
-
Incarcerated Peer Mentors (IPMs) provide their peers with structured health information and education which will help create the kind of cultural change that benefits the incarcerated, supervised persons, their families, and communities through a common frame of reference and set of shared experiences relevant to the patient.
-
Each institution shall coordinate with institutional custody leadership to establish a sufficient number of paid IPM positions to meet the identified needs of the institution. IPMs shall be assigned, monitored, supervised, and evaluated in compliance with the requirements set forth in California Code of Regulations, Title 15, and the Department Operations Manual for participation in the Inmate Work Incentive and Training Program (IWTIP). Nursing Supervisors may be designated as supervisors for the IPMs working in patient education.
-
Trained staff shall facilitate the Patient Education Program. While the primary support for the program shall be provided by nursing staff, each discipline shall provide expertise as necessary.
-
Each institution shall designate one Lead PHE Facilitator and a sufficient number of PHE coordinators to coordinate and, as necessary, conduct activities related to the implementation and administration of the Patient Education Program.
-
These positions do not necessarily need to be licensed clinical staff; however, if they are not, a Registered Nurse shall be designated as a resource for the Lead PHE Facilitator.
-
-
Program Development (Curricula)
-
A multidisciplinary team shall develop each Peer Health Group curriculum under the direction of a HQ CNE.
-
PHE classes shall include, but not be limited to:
-
Vaccines
-
STDs
-
HIV
-
Hepatitis C
-
Norovirus
-
Healthy Lifestyle
-
Depression
-
Grief
-
Substance Use
-
-
Nursing staff shall collaborate with other health care professionals and interact with patients to ensure that health-related information discussed in peer mentoring meetings is of reasonable accuracy so as to promote health maintenance.
-
-
Each institution shall develop a program in which IPMs are randomly observed to ensure the quality of material and to support the IPMs in group facilitation activities.
-
Each IPM shall be trained and their “mastery” of the material verified prior to their conducting of any peer health care education activities. Training, competency, and periodic observations shall be documented in the IPM’s IWTIP files.
-
A local community connection is a valuable resource for the IPMs. It is important that each institution coordinate their Patient Education Program with programs offered by community-based organizations. Community-based organization shall be encouraged and recruited to participate in facilitating the program training patients and in the sharing of resources, expertise, and follow up upon release from CDCR custody.
-
-
-
Appendices
-
Appendix 1: Sample NL-TG Schedule
-
References
-
California Penal Code, Part 3, Title 2, Chapter 3, Sections 3407 and 3409
-
California Proposition 57: The Public Safety and Rehabilitation Act of 2016
-
Health Care Department Operations Manual, Chapter 2, Article 1, Section 2.1.2, Effective Communication Documentation
-
Health Care Department Operations Manual, Chapter 3, Article 3, Dental Care
-
The Joint Commission, (2018). Retrieved from: https://www.jointcommission.org/Topics/
-
U.S. Department of Health and Human Services (2017). Agency for Healthcare Research and Quality: Guide to patient and family engagement in hospital quality and safety. Retrieved from:
https://www.ahrq.gov/professionals/systems/hospital/engagingfamilies/index.html -
Revision History
-
Effective: 01/2006
Revised: 07/2019 -
Appendix 1: Sample Nurse Lead Treatment Group Schedule
-
Institution: Anywhere State Prison
Day of Week Hours Title Location Monday 0900-1000 Cancer Awareness A5-102 1030-1200 Medication Management A5-102 1230-1400 Anger Management (Module 1) A3-101 1415-1545 Victim Awareness A5-102 1830-2000 Self Care Skills (Module 1) A3-101 Tuesday 1300-1400 Anger Management (Module 2) A5-102 1415-1545 Men’s Health A5-102 Wednesday 0900-1000 Cancer Awareness A5-102 1030-1200 Medication Management A5-102 1230-1400 Anger Management (Module 1) A3-101 1415-1545 Victim Awareness A5-102 1830-2000 Self Care Skills (Module 1) A3-101 Thursday 0900-1000 Stress Management A5-102 1030-1200 Men’s Health A5-102 1230-1400 Diabetes’s Education & Spt. Gp A3-101 1415-1545 Asthma Education & Spt. Gp A5-102 Friday 0900-1000 Cancer Awareness A5-102 1030-1200 Medication Management A5-102 1230-1400 Anger Management (Module 1) A3-101 1415-1545 Victim Awareness A5-102 1830-2000 Self Care Skills (Module 2) A3-101
3.1.5 Scheduling and Access to Care
-
Policy
-
California Correctional Health Care Services (CCHCS) and California Department of Corrections and Rehabilitation (CDCR) shall utilize systems and processes to optimize access to care and maintain an effective and efficient scheduling system to ensure timely patient access to health care services. This includes a flexible appointment system that accommodates various encounter appointment types, encounter lengths, same-day encounters, and scheduled follow-ups as well as strategies to increase efficiency, such as consolidated appointments. This procedure also specifies roles and responsibilities for key staff involved in the scheduling system.
-
-
Responsibility
-
Statewide
-
CCHCS and CDCR departmental leadership at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial, and clinical systems are in place, and appropriate tools, training, technical assistance, and levels of resources are available to ensure the scheduling system is successfully implemented and maintained.
-
-
Regional
-
Regional Health Care Executives are responsible for the administration of this procedure at the subset of institutions within an assigned region.
-
-
Institutional
-
The Chief Executive Officer (CEO) has overall responsibility for implementation and ongoing oversight of the scheduling and access processes at the institution. The CEO delegates decision-making authority to the institution leadership team for daily operations to ensure adequate resources are deployed to support the process including, but not limited to the following:
-
Ensuring access to and utilization of equipment, supplies, health information systems, patient registries, patient summaries, and evidence-based guidelines.
-
Assigning patients to a Care Team.
-
Maintaining a list of the core members of each Care Team, which shall be available to all institutional staff. Patients shall be informed of their assigned Care Team members at intake or upon request.
-
Ensuring consistent Care Team staffing with a plan to designate back-up staff.
-
Providing Care Team members with the information they need during huddles (e.g., communication of on-call information).
-
Ensuring protected time for Care Teams to hold daily huddles and twice-monthly population management working sessions.
-
Documenting and tracking huddle actions, follow-up deliverables and attendance.
-
Ensuring that at least twice-monthly, each Care Team conducts a Population Management Working Session utilizing tools such as dashboards, patient registries, patient summaries, and other tools to identify potential gaps in care and opportunities to improve safe, appropriate, timely and cost-effective services.
-
Adequately preparing new Care Team members to assume team roles and responsibilities.
-
Assessing competence of existing Care Team members.
-
Updating institution procedures, roles, and responsibilities as new tools and technology become available.
-
Reviewing or comparing institution Care Team performance including the overall quality of services, health outcomes, assignment of consistent and adequate resources; utilization of dashboards, patient registries, patient summaries, and decision support tools; and addressing issues as necessary.
-
Providing Care Team members with adequate resources including staffing, physical plant, information technology, and equipment or supplies to accomplish daily tasks.
-
Working with custody staff to minimize unnecessary patient movement resulting in changes to a patient’s panel assignment.
-
Requiring institution leadership to establish a back-up system to ensure that scheduling queues are managed when Scheduling Support Staff are on leave or otherwise unable to meet daily monitoring requirements.
-
Ensure Local Operating Procedures are followed by applicable staff under the CEO and Warden’s direction.
-
-
The CEO and all members of the institution leadership team are responsible for establishing an organizational culture that promotes teamwork among Care Team members and across disciplines.
-
The CEO and institution leadership team shall review institution-wide scheduling and access to care data monthly in the context of local Quality Management Committee and subcommittee meetings.
-
To ensure accuracy of scheduling system data, the institution leadership team shall:
-
Periodically evaluate the reliability of scheduling system data through comparison with independent data sources, such as movement or ducat reports and progress notes, or audits for abnormal or incomplete entries.
-
Take effective action to remedy unreliable data, including creating or revising decision support, updating desk procedures, and redesigning orientation and training strategies.
-
Re-validate problematic data monthly until the data reliability issue is resolved.
-
-
Local quality improvement committees shall act as appropriate to investigate quality problems and develop interventions to improve access.
-
The Chief Nurse Executive (CNE) is responsible for:
-
The overall daily operations of the scheduling system for medical care.
-
The daily coordination of health care services between health care scheduling systems.
-
Daily oversight and management of scheduling processes and resources including personnel.
-
Ensuring that the institution has a designated Scheduling Supervisor to monitor scheduling processes daily and identify and address or elevate barriers to access.
-
Ensuring that Scheduling Support Staff is available daily for all clinical areas.
-
-
The Chief Medical Executive (CME) is responsible for the overall medical management of patients and ensures provider resources are available to meet the needs of the population.
-
At least monthly, the CME and CNE shall review the effectiveness of local scheduling processes including, but not limited to, the following in each Primary Care Clinic to determine if adjustments need to be made to the overall clinic operations plan to meet patient care needs in an efficient manner:
-
Scheduling Reports.
-
Utilization of the consolidated patient provider calendar.
-
Utilization of open access time and co-consultation.
-
Number of additional “add-on” appointments.
-
Current backlog.
-
Wait times.
-
Refusal rates.
-
-
The Supervising Registered Nurse (RN) and Chief Physician and Surgeon shall meet to review the Care Teams’ performance including the overall quality of services, health outcomes, and level of care utilization and shall utilize dashboards, patient registries, patient summaries, and decision support tools to address or elevate issues as necessary.
-
The Scheduling Supervisor over Clinics and the Clinic Manager shall:
-
Review select information daily to identify and immediately address scheduling system problems.
-
Determine whether all Scheduling Support Staff, Nursing Staff, Primary Care Providers (PCPs) and Medical Assistants, attend their respective clinics that day and shall verify that appropriate back-up has been provided if any of these staff are unavailable.
-
Review scheduling management reports daily including, but not limited to, the following:
-
Scheduling system diagnostic data to identify data entry errors and appointment trends.
-
Scheduling queues not managed properly.
-
Duplicate appointments and orders.
-
Unscheduleable appointments.
-
Other scheduling system issues.
-
-
Review clinic scheduling processes to ensure utilization of strategies such as open access, encounter consolidation, scheduling conflict resolution, and co-consultation to optimize access.
-
Improve communication processes within the Care Team and across health care settings that impact scheduling and access, including daily huddles.
-
Provide frequent feedback to health care staff involved in the scheduling system on their individual performance based upon findings from daily observation of scheduling processes.
-
-
The Care Team
-
At least monthly, the Care Team shall evaluate the effectiveness and efficiency of scheduling processes and overall access to care. The Care Team shall consider trends in the following:
-
Adherence to access timeframes.
-
Proportion of appointments seen as scheduled and reasons patients were not seen as scheduled.
-
Episodic Care referral rates to the PCP.
-
Effectiveness of scheduling strategies, such as open access, encounter consolidation, scheduling conflict resolution, and co-consultation.
-
Design of clinic schedules (e.g., number of open access slots, allotting certain time blocks for different appointment types).
-
Productivity.
-
Demand management, including episodic care, chronic care, chronos, medication refusals and other types of non-adherence counseling, and grievances.
-
Allocation of work across team members.
-
Clinic closures.
-
Specialty provider network issues.
-
Completeness and accuracy of scheduling data.
-
Security and construction impact to access.
-
Population management health care alerts.
-
-
The Care Team shall take corrective action to resolve and elevate concerns identified in the review. The Care Team review and corrective action shall be documented and forwarded to the designated committee.
-
-
Health care staff shall be trained in scheduling and access to care concepts and principles. Targeted training shall be provided to those who have specific roles in the scheduling process (e.g., providers, nurses, schedulers). A system for the orientation, mentoring, and cross-training of all critical positions in the scheduling system shall be maintained.
-
Each institution shall ensure all Scheduling Support Staff have a desk procedure with guidance on how to employ the scheduling system accurately and effectively with information tailored to different work locations and scheduling functions. The desk procedure shall be updated as scheduling processes change.
-
Each institution shall develop or adopt decision support tools (e.g., forms, checklists):
-
Prompting clinic staff to communicate clearly to Scheduling Support Staff.
-
Giving tips on how to enter data in a way that is recognized by the scheduling system.
-
Reminding Scheduling Support Staff and clinic staff of new scheduling procedures and updated access to care timeframes.
-
-
Staff involved in the scheduling system shall receive training on changes to scheduling processes and tools as they evolve and periodic refresher training on their roles and responsibilities.
-
-
-
Procedure
-
General Scheduling Concepts
-
Standardized Scheduling System
-
All institutions shall use the standardized statewide scheduling system.
-
-
Scope of the Scheduling Process
-
The scheduling process shall begin upon a patient’s arrival at CDCR and continue throughout the patient’s stay.
-
-
Scheduling System User Designations and Accessibility
-
Staff shall submit a Solution Center ticket to add or change a provider or location.
-
-
-
Access to Health Care Services
-
Hours of Access
-
All CDCR incarcerated persons shall have access to medically necessary health care services seven days per week, 24 hours per day.
-
RNs shall be onsite at the institution seven days per week, 24 hours per day.
-
A Provider-on-call or Medical Officer of the Day shall be available 24 hours a day,7 days a week to provide consultation and onsite care as necessary.
-
Medical, mental health, and dental services shall be available at any time.
-
STAT laboratory tests listed on the Approved STAT Testing Menu are available seven days per week, 24 hours per day.
-
-
Each institution shall establish hours of operation for Primary Care Clinics, generally at least eight hours per day, Monday through Friday, excluding state holidays.
-
-
Methods of Access
-
Licensed Health Care Initiated Appointments
-
Access to care includes planned health care encounters scheduled at appropriate intervals and initiated by licensed health care staff as part of ongoing treatment planning and care management to address health care needs.
-
-
Patient Request for Services:
-
Access to care also includes episodic encounters requested by patients either through written request, verbal report, or demonstration of urgent or emergent health care needs.
-
At any time, patients with health care needs may submit a CDCR 7362, Health Care Services Request Form. Patients with urgent health care needs may complete a CDCR 7362 or notify any institutional staff, including correctional staff for assistance. Patients with life-threatening conditions shall receive immediate medical attention.
-
If a patient is unable to complete a CDCR 7362, health care staff shall complete the form on behalf of the patient. Health care staff shall document the complaint and the reason the patient did not personally complete the CDCR 7362 and shall sign and date the CDCR 7362.
-
Institutions shall ensure the CDCR 7362 is available to patients in the housing units, clinics, and Reception Centers. Housing unit staff and health care staff shall make the CDCR 7362 available upon request. Each institution shall have at least one locked box on each yard and facility designated for patients to deposit the CDCR 7362, which shall be accessible to patients daily.
-
-
Initial Review and Triage of a CDCR 7362
-
On normal business days:
-
A designated health care staff member on each yard or facility shall collect the CDCR 7362s from the designated areas, document the date and time of pickup, and deliver the forms to the Primary Care RN (PCRN) for review.
-
Upon receipt of the CDCR 7362, the PCRN shall:
-
Review and triage patient health needs based on information documented on the CDCR 7362.
-
Determine whether the patient requires emergent, urgent, or routine care.
-
Immediately refer emergent, or urgent medical, mental health, and dental needs to the appropriate clinician for evaluation consistent with established program guidelines.
-
Indicate which discipline the patient is being referred to, and whether the health care request is symptomatic (to include any Substance Use Disorder [SUD] complaint) or asymptomatic.
-
Document the following on each CDCR 7362: date and time reviewed by RN; print or stamp name, signature and title.
-
Based on the RN’s review in triage, the RN shall immediately contact the appropriate department when indicated and document the name and title of the licensed clinician notified and the date and time of notification on the CDCR 7362.
-
Copy and deliver/forward requests for services for more than one area (e.g., medical, mental health, and dental) to the requested service areas as soon as possible.
-
-
-
On non-business days:
-
All CDCR 7362s shall be sent to the Triage and Treatment Area (TTA) RN for review and triage.
-
Upon receipt of the CDCR 7362s, the TTA RN shall follow the procedure set forth in Section (c)(2)(B)3.a.2)a)-b) and d)-g) each.
-
The TTA RN shall determine whether the patient requires emergent, urgent, or routine care and shall take direct action to coordinate care for patients with emergency or urgent conditions.
-
The TTA RN shall ensure that the routine CDCR 7362s are delivered to the PCRN that is assigned to that patient by the beginning of the next business day.
-
-
-
Emergency Care Required
-
Patients with life-threatening medical symptoms shall receive immediate medical attention pursuant to the Health Care Department Operations Manual (HCDOM), Chapter 3, Article 7, Emergency Medical Response.
-
The PCRN shall ensure immediate transportation of the patients to the designated area for evaluation and treatment.
-
For patients with a potential mental health or dental emergent condition during normal business hours, the PCRN shall immediately assess the patient and communicate findings directly with designated mental health or dental staff.
-
Patients with a potential mental health emergency (e.g., danger to self or others or significant impairment or dysfunction due to mental disorder) shall remain under continuous observation until the patient is evaluated by a mental health clinician or by TTA medical staff.
-
When a patient is referred to the mental health program, the CDCR 7362 shall be forwarded, and a Mental Health Consultation ordered by the PCRN.
-
-
Urgent Care Required
-
Patients with urgent medical symptoms shall be scheduled for a same day face-to-face encounter with the PCRN and other members of the Care Team as indicated by symptoms.
-
Any dental or mental health needs that are deemed urgent may be directly referred to the appropriate clinicians for evaluation, as availability allows.
-
In such cases, the RN shall immediately contact the appropriate department and document the name and title of the licensed clinician notified and the date and time of notification on the CDCR 7362.
-
The referral shall also be documented in the health record.
-
-
For patients with urgent symptoms involving more than one clinical discipline, the PCRN shall ensure any urgent medical, dental, or mental health conditions are evaluated.
-
-
Symptoms One Business Day Required
-
The PCRN, using clinical judgement, shall assess any patient who describes symptoms or a potentially harmful situation within one business day.
-
If symptoms or conditions are consistent with a standardized procedure, the PCRN shall implement the interventions within the standardized procedure.
-
If the symptoms or conditions are not consistent with a standardized procedure, the PCRN shall determine (based upon their clinical judgement) the need for co-consult with the PCP to meet the needs of the patient.
-
In addition to the required documentation by the PCRN, the PCP shall document the communication with the PCRN and the medical plan of care in the health record preferably on the day the co-consultation occurs. The PCP shall assess whether an in-person PCP evaluation or follow up are clinically necessary.
-
-
SUD Requests One Business Day Required
-
The PCRN shall assess any patient who submits a CDCR 7362 related to SUD that is not emergent or urgent within one business day.
-
If the SUD request is emergent or urgent, follow sections above (Emergency Care Required or Urgent Care Required).
-
The PCRN may co-consult with the PCP and if needed, co-consult with the Addiction Medicine Central Team.
-
-
-
-
Asymptomatic Requests
-
The PCRN shall separately address routine CDCR 7362s that do not include symptoms or a potentially harmful situation within 14 calendar days, routing them to appropriate staff or, if clinically indicated, have a face to face encounter with the patient.
-
CDCR 7362 requests for programs (dental, mental health, etc.) that do not describe symptoms shall be delivered the same day to the designated program representative on normal business days.
-
-
PCP Referrals
-
When the PCRN determines a PCP referral is necessary, the patient shall be seen based on the following timeframes:
-
Emergency – immediately
-
Urgent – within 24 hours
-
Routine – within 14 calendar days
-
-
-
-
Scheduling Strategies
-
CCHCS staff shall use strategies such as open access, encounter consolidation, co-consultation, and collaborative planning of the clinic schedule to optimize access to medical services.
-
Services that Require Appointments
-
Health care encounters shall be considered appointments and shall be ordered and scheduled within the Electronic Health Record System including, but not limited to, the following encounter reasons:
-
Episodic care encounters, including PCRN encounters and provider referrals.
-
Well patient encounters.
-
Chronic care follow-up appointments.
-
Specialty services.
-
Care management encounters.
-
Interdisciplinary treatment planning sessions.
-
Recurring patient monitoring or follow-up appointments, such as dressing changes and blood pressure checks.
-
Injection appointments.
-
Public health screening and treatments.
-
Patient education and non-adherence counseling.
-
Special situations such as hunger strike evaluations and monitoring.
-
Follow up after return from a higher level of care.
-
Health care grievances.
-
Release Planning – Care Coordination.
-
-
In the event a patient transfers to another institution, the receiving Care Team shall ensure that existing health care appointments, including specialty referrals, are reordered at the receiving institution as indicated. All members of the Care Team shall ensure that follow-up appointments are continued in Cross Encounter Reconciliation including, but not limited to, the following:
-
TTA encounters.
-
Receiving and Release intake.
-
Discharge from a higher level of care.
-
Integrated Substance Use Disorder Treatment (ISUDT) Behavioral Health appointments.
-
Chronic Care PCP appointments.
-
Addiction Medicine Central Team orders.
-
-
-
Translation Services
-
Translation services, including sign language, shall be made available to patients as necessary via certified bilingual health care staff, certified bilingual CDCR staff, or by utilizing a certified interpretation service. Each institution shall maintain a contract for certified interpretation services pursuant to the HCDOM, Section 2.1.2, Effective Communication Documentation.
-
-
Scheduling
-
General Requirements
-
Health care staff shall:
-
Communicate to custody staff no later than one business day prior to the scheduled encounter.
-
Minimize scheduling conflicts for patients, including avoiding conflicts between health care appointments and other programming such as visitation, Board of Parole Hearings (BPH), and school or job assignments by using the Consolidated Patient/Provider Calendar.
-
-
Each institution shall establish a procedure by which health care ducats are issued as priority ducats and delivery by custody is verified and documented. This procedure shall include the following:
-
The method by which priority health care ducats are delivered to each patient.
-
The individual responsible for issuing priority health care ducats.
-
Verification by custody staff that the priority health care ducats were issued to the patient.
-
A method of re-routing priority health care ducats to patients and documentation of the re-routing.
-
-
The patient is responsible to report to the health care appointment at the time indicated on the priority health care ducat.
-
Developmental Disability Program or Disability Placement Program designated patients shall be provided specific instruction regarding the time and location of their scheduled appointment. The custody staff delivering the priority health care ducats shall communicate effectively and appropriately based upon the patient’s ability to understand to ensure that the patient arrives at the designated appointment location
-
-
Custody staff shall deliver priority health care ducats to patients prior to their scheduled appointment.
-
Failure to Report for a Medical or Dental Appointment
-
If the patient (including patients who are in the Mental Health Services Delivery System [MHSDS]) fails to report to a scheduled medical or dental appointment, the assigned health care access clinic officer shall immediately contact the designated housing unit, or work or program assignment to locate the patient and have them escorted or have the patient report to the scheduled medical or dental appointment.
-
Custody staff shall locate the patient and escort the patient to the appointment or direct the patient to report to the scheduled medical or dental appointment. If necessary, custody staff shall order the patient to comply with the instructions on the priority ducat.
-
If the reason the patient did not report as ducated was beyond the patient’s control (e.g., out to court), custody staff shall advise health care staff of this fact. Health care staff shall document the cancellation and reorder, if clinically necessary.
-
If the patient continues to refuse, custody staff shall advise the patient that they are in violation of Title 15, Section 3014, Calls and Passes, which states “Inmates must respond promptly to notices given in writing, announced over the public address system, or by any other authorized means.”
-
If the reason the patient did not report as ducated was due to the patient refusing to report as directed, custody staff shall escort the patient to the health care area for health care staff to discuss the implications of refusing health care treatment. Licensed health care staff shall counsel the patient and have the patient sign the CDCR 7225, Refusal of Examination and/or Treatment if the patient continues to refuse treatment after the counseling. The CDCR 7225 shall be filed in the health record.
-
Patients who are insistent in their refusal to report shall not be subject to cell extraction or use of force to gain compliance with the priority health care ducat. In these instances, licensed health care staff shall respond to the patient’s housing unit to provide the necessary patient education regarding the refusal. Custody staff cannot accept refusals on behalf of the patient, nor can refusals be taken over the phone.
-
The reason for the failure to report shall be documented by health care staff in the health record.
-
Custody staff may issue a CDC 115, Rules Violation Report, if the patient refuses to present to the clinic.
-
-
Medical appointments shall be rescheduled as clinically indicated.
-
Dental appointments shall be rescheduled according to the HCDOM, Section 3.3.5.1, Priority Health Care Services Ducat Utilization.
-
-
Failure to Report for a Mental Health Appointment
-
If a patient in the MHSDS refuses to report for a mental health appointment in person, custody staff shall not complete a CDC 115 or a Counseling Only Rules Violation Report (formerly known as a CDCR 128A, Custodial Counseling Chrono).
-
Refer to the CDCR Mental Health Services Delivery Systems Program Guide and current mental health policies for additional procedures regarding mental health appointment refusals.
-
-
-
Lockdown and Other Security Concerns
-
Health care services shall continue to be provided during alarms or incidents not occurring on the impacted clinic yard. For alarms or incidents occurring on the clinic yard, clinic services shall resume as soon as safely possible during and following the alarms or incidents.
-
During a facility or prison lockdown, health care staff shall coordinate with custody staff to facilitate continuity of care. Custody personnel shall escort patients to scheduled clinic appointments; lockdown shall not prevent the completion of critical functions such as medication administration and scheduled or unscheduled health care appointments in or out of the institution.
-
A system shall be maintained to provide patient access to health care services in restricted housing units and facilities or housing units. Access to health care services shall continue to be managed by the CDCR 7362 process and shall be accomplished via health care staff alerting all patients of the collection of the CDCR 7362 when entering a housing unit for the purpose of retrieving completed CDCR 7362s. This shall be done at least daily in each restricted housing unit and in any housing unit where patients are confined to their cells or the building and have no ability to submit access to care requests. The collection of the CDCR 7362 shall be documented by health care staff in the housing unit logbook.
-
-
Security Precautions During Health Care Encounters
-
Health care encounters shall be provided in a manner that affords both auditory and visual confidentiality consistent with security and safety concerns of patients and health care providers.
-
Health care staff shall carry a whistle and, where available, a personal alarm and position themselves to have a clear egress route from the treatment room while performing assigned duties.
-
Health care screenings, evaluations, interviews, and treatment shall be held in a private setting unless the security of the institution or safety of staff will be compromised, or unless health care staff in the presence of the patient requests the presence of custody staff. As a default, custody staff are not required during a health care encounter with a patient who is not maximum custody or whose current behavior does not present a threat to the safety of staff or other patients.
-
A patient shall not be placed in mechanical restraints during a health care encounter unless they are a safety concern for staff or others as determined by custody staff. For mental health treatment, the use of mechanical restraints shall comply with existing mental health policies.
-
Health care staff may ask custody staff to leave the room if they are comfortable with the patient and custody staff shall respect the request of health care staff and leave the room.
-
If health care staff asks custody staff to exit the room and leave the door propped open, custody staff shall be in control of the door to remain in compliance with State Fire Marshall requirements.
-
-
A treatment module shall be utilized for the duration of encounters with patients who are a safety and security risk. For patients receiving treatment as a part of the MHSDS and requiring the use of a module for safety and security risks, a therapeutic treatment module shall be utilized.
-
Upon removal of the mechanical restraints, the front port on the module shall be closed during the encounter.
-
Health care staff shall not put their face in or near the opening of a cuff or food port.
-
If it is necessary to perform a procedure, the patient shall be removed from the treatment module and placed in waist restraints while being treated outside the module.
-
-
When health care staff are in housing units or on the tiers, custody staff shall maintain visual surveillance.
-
Visual surveillance shall not interfere with the privacy of the encounter except for cell front medication distribution.
-
When unscheduled clinical encounters need to occur within a housing unit, health care staff shall conduct the encounter in a confidential setting with custody staff maintaining visual observation when necessary.
-
-
-
Clinic Closure or Cancellation of Scheduled Appointments
-
Any modification of clinic hours, clinic closure, and cancellation or rescheduling of scheduled appointments requires the approval of the CEO or a designated clinical executive.
-
-
Timeframes
-
Under the Complete Care Model, the goal of all Care Teams is to provide timely access to care and to allow immediate access to necessary services.
-
To ensure that patients are not exceeding acceptable thresholds for timely care, access to care timeframes shall be viewed as the maximum allowable timeframe that a patient may be seen and not as a guideline for scheduling.
-
Scheduling Support Staff shall set appointments several days in advance of the acceptable threshold.
-
-
Patients with chronic conditions shall have follow-up encounters according to the timeframes in the applicable care guides. If there is no applicable care guide, the follow-up shall be as ordered or no less frequently than every 365 days.
-
For follow-up appointments with the PCP after specialty services, refer to the HCDOM, Section 3.1.11(c)(7).
-
Patients discharged to an outpatient setting from the TTA who experienced a high-risk event including, but not limited to, suspected drug overdose that responded to naloxone resuscitation or skin/soft tissue infection shall be seen by their PCP within five calendar days of discharge from the TTA.
-
Patients discharged to an outpatient setting from the TTA who did not experience a high-risk event, which led to the TTA encounter and do not require PCP follow up within five calendar days, shall be seen by their PCRN or PCP within timeframes that are clinically indicated.
-
Patients discharged to an outpatient setting from an unplanned community hospitalization or emergency department encounter, or from a CDCR specialized medical bed stay not related to a mental health crisis or PIP stay shall be seen by their PCP within five calendar days of discharge.
-
Patients in the MHSDS shall be scheduled for appointments in accordance with the Mental Health Program Guide and current mental health policies.
-
Patients screened positive for SUD with a National Institute on Drug Abuse Quick Screen, or otherwise determined to need completion of SUD assessment shall be referred for an ISUDT Behavioral Health assessment within the following timeframes:
-
Patients rapidly induced on medication-assisted treatment (MAT) medications shall be seen within seven calendar days.
-
Patients identified in all other circumstances shall be seen within 30 calendar days.
-
-
Patients with a completed SUD assessment indicating Opioid Use Disorder or Alcohol Use Disorder shall be seen by a provider within 30 calendar days for evaluation for MAT.
-
Patients releasing to the community who require pre-release care coordination shall have follow-up appointments as dictated by the Earliest Possible Release Date. Refer to the Whole Person Care – Community Discharge Planning/Pre-Release Workflow.
-
Patients induced on a MAT medication shall be seen for a Post Induction MAT Medication Evaluation.
-
In the outpatient setting, patients shall have MAT Medication Evaluation completed by an RN, Licensed Vocational Nurse, or Licensed Psychiatric Technician within three calendar days post induction.
-
In the inpatient setting, patients shall have MAT Medication Evaluation completed by an RN on the day after the induction.
-
-
-
Scheduling Queues and Building the Clinic Schedule
-
Health care staff shall place orders for appointments that need to be scheduled, which will flow into various request queues in the scheduling system. Scheduling Support Staff are responsible for monitoring the appropriate request queue for each Care Team and clinic location daily with particular focus on scheduling appointments for patients within several days of the relevant threshold date.
-
-
Increasing Patient Show Rates and Clinic Efficiency
-
When scheduling patients, health care staff shall consider patient preferences regarding access, such as providing appointment times that do not interfere with the patient’s existing health care appointment and the patient’s assigned programming (e.g., BPH, job, school, or other rehabilitative programming).
-
-
Recurring Appointments
-
Scheduling Support Staff shall use the recurring appointment function when a provider or clinician’s order will result in a series of appointments with a specified frequency.
-
-
Rescheduling
-
Scheduled appointments shall be rescheduled as needed, and efforts shall be made to reschedule within the original order compliance date.
-
-
Cancelling Orders for Appointments and Scheduled Appointments
-
Health care staff are prohibited from cancelling or discontinuing orders for appointments and scheduled appointments from the scheduling system unless there is documentation explaining the reason for cancellation, and the health care staff are authorized to perform cancellations.
-
-
Tracking “Reasons Not Seen”
-
Health care staff shall record and track reasons that patients are not seen as scheduled. Health care staff shall use the standard “Reasons Not Seen” as listed in the scheduling system.
-
-
Confirmed Appointments Already Seen
-
The Primary Care Scheduler, or designee, is responsible to contact members of the Care Team to obtain any missing information or address discrepancies.
-
-
Open Access
-
Institutions shall use open access slots to ensure that patients are seen in an efficient manner, in a clinically appropriate setting, and within all mandated timeframes. Approximately 20 percent of Primary Care Clinic appointment slots shall remain open and available for same-day or next-day urgent clinical issues or appointments with short, mandated timeframes.
-
Primary Care Clinics shall designate specific times each day as open access times for the Care Team.
-
During daily huddles, the Care Team shall identify patients that need to be scheduled into the same-day or next-day open access times and ensure that this information is communicated to the Scheduling Support Staff if they are not present at the huddle.
-
Appointments that may be appropriate for open access slots include, but are not limited to, the following:
-
Follow-up on abnormal diagnostic results or other critical abnormal clinical findings.
-
Return from higher level of care follow-up.
-
TTA follow-up.
-
High priority specialty referral follow-up.
-
High-risk or complex patients new to the Care Team.
-
Patients whose condition has become clinically complex.
-
Other urgent referrals.
-
-
If open access slots remain available even after all urgent follow-ups are addressed, these slots may be used to schedule other routine appointments.
-
Except for certain clinics (e.g., Restricted Housing Unit) where patient need and health care staff coverage may vary, clinic schedules shall be booked 14-30 calendar days out (except for “Open Access” slots).
-
-
Encounter Consolidation (Bundling)
-
To increase clinic efficiency and timely access, Scheduling Support Staff shall review all pending appointments for possible bundling and discuss with the Care Team at the daily huddle to determine the total time required for the patient.
-
-
Co-Consultation
-
Throughout the day, the Care Team shall look for opportunities to collaborate using co-consultation strategies to resolve issues in one encounter that would likely result in a referral to another member of the Care Team, thus eliminating the need for the patient to return to the clinic for a second time.
-
-
Provision of Additional Health Care Staff During Examinations
-
An additional health care staff shall be present during all examinations of patients involving genital, rectal, or breast examinations.
-
Upon patient request, an additional health care staff may be present during other examinations.
-
-
-
-
Appendices
-
Appendix 1: Appointment Timeframes
-
-
References
-
Code of Federal Regulations, Title 45, Parts 160 and 164. Health Insurance Portability and Accountability Act
-
California Code of Regulations, Title 15, Division 3, Chapter 1, Subchapter 4, Article 2, Section 3270, General Policy
-
California Code of Regulations, Title 15, Division 3, Chapter 1, Article 1, Section 3014, Calls and Passes
-
Health Care Department Operations Manual, Chapter 2, Article 1, Section 2.1.1, Patients’ Rights
-
Health Care Department Operations Manual, Chapter 2, Article 1, Section 2.1.2, Effective Communication Documentation
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.6, Population and Care Management Services
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.9, Health Care Transfer
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.11, Outpatient Specialty Services
-
Health Care Department Operations Manual, Chapter 3, Article 7, Emergency Medical Response
-
California Department of Corrections and Rehabilitation, Mental Health Services Delivery System Program Guide
-
Agency for Healthcare Research and Quality – Patient Centered Medical Home Resource Center, https://www.ahrq.gov/ncepcr/research/care-coordination/pcmh/index.html
-
The Joint Commission Primary Care Medical Home Certification, http://www.jointcommission.org/accreditation/pchi.aspx
-
National Committee for Quality Assurance – Patient-Centered Medical Home Recognition, http://www.ncqa.org/Programs/Recognition/Practices/PatientCenteredMedicalHomePCMH.aspx
-
-
Revision History
-
Effective: 06/2016
Revised: 05/19/2025 -
Appendix 1
-
Scheduling Timeframes
-
-
3.1.6 Population and Care Management Services
-
Procedure Overview
-
The Complete Care Model recognizes that patients have varied health care needs, and the delivery system needs to be flexible enough to serve a wide range of patients – from young, healthy patients without any history of chronic disease, to patients with multiple serious mental health, dental, and medical co-morbidities who require intensive services coordinated across multiple providers. The delivery system shall also include a wide range of social supports to maintain health and functioning.
-
California Correctional Health Care Services (CCHCS) shall provide systematic assessment, risk stratification, monitoring, and care management of identified groups of patients at the statewide, regional, institutional, panel, and individual levels to meet the needs of a diverse patient population. Population management includes the evaluation of resources, processes and outcomes and is an integral component of the Complete Care Model and the Primary Care Team (PCT) function.
-
PCTs and other health care staff are expected to provide services to patients commensurate with each patient’s risk level and complexity to protect patients at risk for poor outcomes and ensure the most cost-effective allocation of scarce health care resources. Patients at higher risk utilize the most resources and represent the smallest percentage of the total population. Patients with complex health care needs exposed to the highest risk of permanent disability or death utilize the highest concentration of care management services (refer to figure below).
-
Risk Levels and Actual Adverse Outcomes/Resource Use
-
-
-
CCHCS provides PCTs with guidelines, tools, and reports to support the provision of evidence-based care at all risk levels.
-
This procedure describes the organization’s risk stratification system, specifies the services generally appropriate for patients at different risk levels, and outlines processes for monitoring and managing patient populations, individual patients, and the components of the population and care management services delivery system.
-
-
Responsibility
-
Statewide
-
California Department of Corrections and Rehabilitation (CDCR) and CCHCS departmental leadership at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial, and clinical systems are in place and appropriate tools, training, technical assistance, and levels of resources are available so that Care Teams can successfully implement the Population and Care Management Services Procedure.
-
-
Regional
-
Regional Health Care Executives are responsible for implementation of this procedure at the subset of institutions within an assigned region.
-
-
Institutional
-
The Chief Executive Officer (CEO) has overall responsibility for implementation and ongoing oversight of a system to provide population management which includes risk stratification and care management. The CEO delegates decision-making authority to designated Institutional Health Care Executives for daily operations of the population and care management services system and ensures adequate resources are deployed to support the system.
-
The CEO and all members of the institution leadership team are responsible for ensuring all necessary resources are in place to support the successful implementation of this procedure at all levels including, but not limited to the following:
-
Institution level
-
Patient panel level
-
Patient level
-
-
The CEO and all members of the institution leadership team shall ensure access to and utilization of equipment, supplies, health information system, patient registries and summaries, and evidence-based guidelines.
-
The CEO and all members of the institution leadership team shall ensure protected time at least twice monthly, where each PCT shall conduct a Population Management Working Session utilizing tools such as Dashboards, Master Registries, Patient Summaries to address concerns related to potential gaps in care in order to improve patient outcomes including, but not limited to (refer to the Population Management Working Session Facilitator’s Guide):
-
Trends in access to and quality of care.
-
Patient risk stratification.
-
High risk/complex patients.
-
Patient safety alerts.
-
Resource Management (e.g., contract, supplies, equipment, space, environment).
-
Scheduling Reports.
-
Surveillance of communicable diseases.
-
-
The CEO and all members of the institution leadership team as part of the Quality Management process on an ongoing basis shall:
-
Review and compare institutions’ PCTs performance, including the overall quality of services, health outcomes, assignment of consistent and adequate resources, utilization of Dashboards, Master Registries, Patient Summaries, and decision support tools and address issues as necessary.
-
Provide PCT members with adequate resources, including protected time, staffing, physical plant, information technology, and equipment/supplies to accomplish daily tasks.
-
Work with custody staff to minimize unnecessary patient movement that results in changes to a patient’s panel assignment.
-
-
The Chief Medical Executive (CME) is responsible for overall medical management of patients and ensures resources are available to meet the medical needs of the population.
-
The Chief Nurse Executive (CNE) is responsible for the overall daily clinic operations and ensuring that the institution has designated supervisors to monitor clinic operations including, but not limited to:
-
Efficiency.
-
Coordination.
-
Supplies.
-
Equipment.
-
Physical plant issues.
-
Scheduling and access to care on a daily basis.
-
Identifying and addressing or elevating concerns regarding barriers.
-
-
The designated Supervising Registered Nurse (SRN) and the CME, or designee, shall meet weekly to review the PCTs’ performance, including, but not limited to, the overall quality and efficiency of services, health care outcomes, and level of care utilization. The review shall utilize tools such as Dashboards, Master Registries, Patient Summaries, and decision support tools to address or elevate issues as necessary.
-
-
-
Procedure
-
Determining Patient Risk
-
Automated Risk Classification System
-
CCHCS shall maintain an automated system that continuously updates a patient’s risk status based on the most current clinical information, including demographic, diagnostic, medication, specialty services, and inpatient data.
-
Within two calendar days of arrival at a Reception Center, all incarcerated persons shall be placed into the appropriate risk categories and be made available on the Master Registry. The Master Registry risk information shall be updated daily and made available to PCTs and other health care staff with need-to-know information (refer to Appendix 1, Clinical Risk Stratification Criteria).
-
-
Risk Verification
-
Each PCT shall be responsible for verifying that a patient has been placed at the appropriate risk level.
-
Any time a PCT member determines that a patient may have been placed in the wrong risk category, the team member shall elevate the issue using the established process. Refer to the Clinical Risk Verification Process available on the Lifeline Health Care Department Operations Manual Resources tab, for more detail on the communication process used to elevate discrepancies in risk classification.
-
-
-
Population Management Services Overview
-
Service Types and General Eligibility: CCHCS offers four levels of population management services (refer to Appendix 2, Population Risk and Resource Stratification Matrix):
-
Primary Prevention: Services to promote health, prevent onset of disease, and maintain current health status such as immunization and screening.
-
Secondary Prevention: Services to treat one or more well controlled diseases to avoid serious complications.
-
Tertiary Prevention: Services to treat the advanced stages of one or more diseases and minimize disability and includes complex care management services.
-
Catastrophic/Complex Care: Services may range from restoring health to only providing comfort care and includes complex care management services.
-
-
Primary Prevention Services Overview
-
PCTs are responsible for providing patients within the assigned patient panel services to promote, maintain and improve health, prevent disease, and identify and manage the early onset of disease. This level of services may be provided by Licensed Vocational Nurses, Psychiatric Technicians, and Registered Nurses (RNs) functioning within their respective scopes of practice, in collaboration with other members of the PCT. To fulfill this role, team members shall:
-
Remain current on preventive services guidelines, eligibility criteria, and the priority prevention areas listed in the State Health Care Services Performance Improvement Plan.
-
Use eligibility criteria, available registries and reports to identify and monitor subpopulations within the patient panel.
-
Directly provide preventive services including administering immunizations as well as screening and identifying and addressing risk factors by early interventions such as medications, lifestyle modifications, self-management tools, patient education, and other strategies to promote and maintain health.
-
-
Applicable Guidelines
-
PCTs shall provide Primary Prevention Services to the patient population based on age-gender recommendations from the United States Preventive Services Task Force Guide to Clinical Preventive Services and consistent with Care Guides.
-
PCTs shall utilize approved Nursing Protocols, Order Sets, Standing Orders, Care Guides, and other decision support tools when providing services.
-
-
-
Secondary Prevention Services Overview
-
PCTs are responsible for identifying and managing patients within the assigned panel who have one or more stable chronic diseases by providing services to halt progression and prevent serious complications. This level of services may be provided by RNs functioning within their scope of practice, in collaboration with other members of the PCT. To fulfill this role, team members shall:
-
Remain current on Care Guides issued by the Clinical Guidelines Committee, Nursing Protocols, Standing Orders, and other applicable evidence-based standards of care.
-
Be aware of identified priority chronic disease management areas listed in the Statewide Health Care Services Performance Improvement Plan.
-
Use available registries and reports to identify and monitor subpopulations within the patient panel eligible for disease management services.
-
Directly provide disease management services, including but not limited to, self-management planning and tools, patient education, routine ongoing evaluation of patient’s health status and progress toward self-management goals, and adjustment of treatment strategies and interventions.
-
-
Applicable Guidelines
-
PCTs shall utilize approved Nursing Protocols, Order Sets, Standing Orders, Care Guides, and other decision support tools when providing services.
-
PCTs shall reference other evidence-based guidelines if a Care Guide is not available for a specific condition.
-
-
-
Tertiary Prevention Services Overview
-
PCTs are responsible for identifying and managing patients within the assigned panel who are in advanced stages of one or more chronic diseases to stabilize current disease state, slow progression and to mitigate further complications as well as minimize disability and maximize functioning and independence. This level of services may be provided by RNs functioning within their scope of practice, in collaboration with other members of the PCT. To fulfill this role, team members shall:
-
Remain current on Care Guides issued by the Clinical Guidelines Committee, Nursing Protocols, Standing Orders, and other applicable evidence-based standards of care.
-
Use available registries and reports to identify and monitor subpopulations within the patient panel eligible for tertiary prevention services.
-
Directly provide tertiary prevention services, including but not limited to, routine ongoing evaluation of patient’s health status, adjustment of treatment strategies and interventions, reinforcing self-management planning and tools, patient education, and progress toward mutually agreed upon treatment goals.
-
-
Applicable Guidelines
-
PCTs shall utilize approved Nursing Protocols, Order Sets, Standing Orders, Care Guides, and other decision support tools when providing services.
-
PCTs shall reference other evidence-based guidelines if a Care Guide is not available for a specific condition.
-
-
-
Catastrophic/Complex Care Management Services for High Risk and Clinically Complex Patients Overview
-
High risk and clinically complex patients are at an exponentially higher risk for adverse health outcomes than the average incarcerated person and require more intensive assessment, monitoring, and treatment planning services to mitigate risk. Providing intensive services to this population is also an important utilization management (UM) strategy. While these patients comprise a small proportion of the total patient population (roughly ten percent), they consume more than half of the available pharmaceutical, specialty, and inpatient services. Although all PCT members shall have a role in complex care management, the Primary Care RN shall be the primary coordinator for complex care management services.
-
PCTs are responsible for identifying and managing patients within the assigned panel who have a very severe illness or condition and potentially significant risk factors. Services provided for these patients may have high costs with limited or no opportunity for improvement, stabilization, or cost control (e.g., end of life care, premature labor pregnancy complications). To fulfill this role, team members shall:
-
Remain current on Care Guides issued by the Clinical Guidelines Committee, Nursing Protocols, Standing Orders, and other applicable evidence-based standards of care.
-
Use available registries and reports to identify and monitor subpopulations within the patient panel eligible for catastrophic care services.
-
Directly provide catastrophic care services including, but not limited to, high intensity, direct, total and/or specialized care of complex, complicated, unstable or high risk patients.
-
Coordinate this level of services with the UM Nurse, CNE, CME, and Chief of Mental Health, or their respective designees, and ensure services are provided by a multi-disciplinary team of health care providers including specialists and specialized care settings.
-
-
Applicable Guidelines
-
PCTs shall utilize Care Guides and California Code of Regulations, Title 22, when providing services.
-
PCTs shall reference other evidence-based guidelines if a Care Guide is not available for a specific condition.
-
-
Eligibility for Services
-
Patients with the following risk designations shall be provided complex care management services:
-
High Risk 1.
-
High Risk 2.
-
Clinically Complex.
-
-
The PCT may elect to offer complex care management services to additional patients, including, but not limited to:
-
Medium risk patients with deteriorating health status.
-
Patients unwilling or unable to accept/participate in treatment.
-
Patients submitting multiple health care services requests for the same or similar complaints.
-
-
-
Multi-Disciplinary Plan of Care
-
All patients receiving health care services shall have a multi-disciplinary Plan of Care.
-
The Plan of Care shall be developed by the PCT; at a minimum, this includes the Primary Care RN and PCP. Other disciplines shall be included as indicated by the patient care needs.
-
Detailed discipline-specific plans shall be developed as indicated by patient needs and integrated with the overarching Plan of Care (e.g., Wound Care Plan, Pain Management, and Mental Health Treatment Plan).
-
The Plan of Care shall be documented and maintained in the health record.
-
-
-
Population Management Working Sessions
-
PCTs are responsible for providing the bulk of population management services for patients within their assigned panel.
-
The CME or Chief Physician and Surgeon and the CNE or SRN III, and others as appropriate to the institution’s mission and particular performance issues, shall hold working sessions conjointly with each institution Care Team at least twice monthly to ensure that teams have protected time to identify patient subpopulations and take action to address patient needs.
-
At a minimum, core PCT members shall attend the Population Management Working Session.
-
-
These working sessions offer a forum to:
-
Update PCTs regarding new clinical guidelines and organizational changes relevant to primary care delivery.
-
Identify barriers to care and resource needs.
-
Provide PCTs regular feedback about their performance in managing clinic work, coordinating patient services, and evaluating Care Team performance.
-
Assist Care Teams in managing subpopulations within the patient panel and improving patient outcomes.
-
-
Topics shall include, but are not limited to:
-
Patient registry flags and alerts indicating abnormal clinical findings and/or missing documentation (e.g., CDCR 1845, Disability Placement Program Verification, CDCR 7410, Comprehensive Accommodation Chrono, Medical Classification Chrono, Medical Hold).
-
Trends in the PCT’s performance on key Health Care Services Dashboard metrics as compared to statewide performance objectives, the statewide average, and the performances of other Care Teams at the institution.
-
Utilization of resources including, but not limited to, supplies, equipment needs, and contracts.
-
Access to care data and statistics.
-
Scheduling Reports (e.g., Aging Report, To Be Scheduled Report).
-
Potentially avoidable hospitalizations for any patients within the panel.
-
New patients and patients leaving the panel in the context of their impact on the overall risk stratification of the patient panel (workload management and resource demand).
-
Mission changes that impact the patient panel (e.g., yard conversions, constructions, new programs).
-
-
-
Sustainability of Population Management Services
-
Key Roles in Population Management and Patient Panel Allocation
-
Institution leadership shall periodically review the roles and responsibilities of staff providing oversight or delivering population management services including, but not limited to:
-
PCT members.
-
Clinic operations supervisor.
-
Public Health Nurses.
-
UM Nurses.
-
Unit supervisors.
-
Penal Code 2602 coordinators.
-
Enhanced Outpatient Program coordinators.
-
-
Institution leadership shall ensure procedures, templates, and roles and responsibilities are updated as new tools and technology become available.
-
Institution leadership shall periodically review the composition of patient panels, particularly relative to the number and proportion of patients that fall into each risk category, to ensure available staff resources are distributed in order to provide the required population management services.
-
-
Training and Decision Support
-
The CEO and institution leadership team shall establish an orientation and training program to ensure all staff serving as members of a PCT or supporting Care Team functions fully understand their roles and responsibilities prior to assuming their duties including, but not limited to:
-
Review of the expectations in this procedure.
-
Changes to local population management processes.
-
National health care industry advances pertinent to the Patient-Centered Health Home.
-
New information systems or technology that may increase the efficiency or effectiveness of Care Team processes or forums.
-
Updates in clinical practice, including new CCHCS clinical guidelines, standing orders, nursing protocols, industry best practices, and findings in clinical literature.
-
-
During twice-monthly Population Management Working Sessions and in other forums, institutions shall provide staff involved in population management with training and information to support staff in providing high quality care, including, but not limited to:
-
New guidelines and procedures.
-
Use of new technology and tools.
-
Effective processes and strategies in preventive care, disease management, and complex care management.
-
Best practices in the health care industry relative to population management, especially in the area of handoffs/communication across different health care settings.
-
Common system lapses in the area of population management and ways to prevent these lapses from occurring.
-
Group review of specific patient cases for problem-solving and education.
-
-
Institutions shall assess the competence of staff performing population management work at least annually. Results shall be documented in the employee file and staff development training file.
-
-
Performance Evaluation and Improvement
-
Designated Standing Improvement Committee
-
Institution leadership shall designate an existing interdisciplinary standing committee reporting to the local Quality Management Committee (QMC) for oversight of the population management system monitoring activities.
-
-
Evaluation
-
The committee shall review population management performance trends and take action to improve care at least monthly. At a minimum, the committee shall review Health Care Services Dashboard information but may also consider monitoring reports, internal audits and surveys, and reviews by stakeholders such as court experts, the Prison Law Office, and the Office of the Inspector General. Beyond trends in performance metrics, the committee shall also consider the quality and effectiveness of program infrastructure, including, but not limited to:
-
Culture of excellence and teamwork.
-
Communication between PCT members, PCTs, health care staff and custody, and with providers in other health care settings.
-
Health information flow, including registry and Patient Summary usage.
-
Resource allocation in accordance with panel composition.
-
The extent to which daily huddle, Population Management Working Sessions, and Care Plan Conferences satisfy the purpose and requirements outlined in this procedure.
-
Competency of staff in key population management roles.
-
Decision support.
-
System for orienting and developing staff.
-
Program monitoring and staff competency testing.
-
-
Committee Actions
-
The committee may take a number of actions to improve program performance, including, but not limited to:
-
Identifying and prioritizing areas for improvement in population management.
-
Setting performance objectives.
-
Establishing improvement teams and/or directly managing improvement initiatives.
-
Applying nationally-recognized improvement techniques to analyze quality problems and develop and test solutions.
-
Monitoring the progress of improvement initiatives at least monthly and intervening as necessary when initiatives stall or show a decline in performance.
-
Identifying best practices and disseminating them across the institution.
-
Documenting improvement activity and results.
-
Regularly reporting performance trends and improvement activities to the QMC.
-
Ensuring that staff working in population management has the knowledge and skills necessary to contribute to improvement activities.
-
-
-
-
Appendices
-
Appendix 1, Clinical Risk Stratification Criteria
-
Appendix 2, Population Risk And Resource Stratification Matrix
-
-
References
-
California Code of Regulations, Title 22, Division 5, Chapter 12, Correctional Treatment Centers
-
California Penal Code, Part 3, Title 1, Chapter 3, Article 1, Section 2602
-
United States Preventive Services Task Force Guide to Clinical Preventive Services
http://www.ahrq.gov/professionals/clinicians-providers/guidelines-recommendations/guide/index.html -
Agency for Healthcare Research and Quality – Patient Centered Medical Home Resource Center, Patient-Centered Medical Home | ACT Center (act-center.org)
-
The Joint Commission Primary Care Medical Home Certification, http://www.jointcommission.org/accreditation/pchi.aspx
-
National Committee for Quality Assurance – Patient-Centered Medical Home Recognition, http://www.ncqa.org/Programs/Recognition/Practices/PatientCenteredMedicalHomePCMH.aspx
-
Commonwealth Fund – Safety Net Medical Home Initiative, http://www.commonwealthfund.org/interactives-and-data/multimedia/videos/2011/safety-net-medical-home-initiative
-
Robert Wood Johnson Foundation & John Hopkins Bloomberg School of Public Health/Chronic Care: Making the Case for Ongoing Care: Chronic Care: Making the Case for Ongoing Care (jhu.edu)
-
Accelerating Care Transformation (ACT) Center, Kaiser Permanente Washington Health Research Institute: Reducing Care Fragmentation: A Toolkit for Coordinating Care: https://www.act-center.org/application/files/7016/3112/2157/Toolkit_Reducing_Care_Fragmentation.pdf
-
-
Revision History
-
Effective: 06/2016
-
-
Appendix 1: Clinical Risk Stratification Criteria
Risk Category Definition Complex Care High Risk 1 Patients who trigger at least 2 of the following high risk selection criteria:
∙ High Risk Diagnosis/Condition: associated with current or future risk for adverse health event
∙ Multiple Higher Level of Care Events:
– Medical – 2 or more community hospital admissions in the past 12 months
– MH – 3 or more MH higher level of care admissions in the past 12 months
∙ Prolonged Medical Bed Stay: patients in CRC, OHU or a SNF≥50% of the past 90 days
∙ Polypharmacy: patients on 10 or more medications
∙ High risk specialty consultations (e.g., oncologist, vascular surgeon)- 2 or more in past 6 months
∙ 65 years of age or older
∙ Co-Morbid Medium Risk Diagnoses/Conditions: a combination of “medium risk” conditions which can be additive to increase the risk for future adverse health care events. (e.g., CKD with DM)Patients who meet one or more of the following criteria:
∙ Any mental health level of care higher than CCCMS
∙ Risk level of High Risk 1 or 2
∙ 2 or more admissions to a mental health higher level of care in the past 6 months
∙ Polypharmacy (taking 10 or more medications)
∙ Any hospitalizations in the past 3 months
∙ On medical hold
∙ Special Outpatient Program statusHigh Risk 2 Patients who trigger at least one of the high risk selection criteria listed under “High Risk 1” above. Medium Risk Patients with at least 1 chronic condition who do not meet the selection criteria for high risk; includes patients enrolled in the Mental Health Services Delivery System and patients with permanent disabilities (under ADA) affecting placement. Low Risk Healthy patients who do not meet any of the selection criteria for medium or high risk; includes a subset of patients with well-managed/stable chronic illness. -
Appendix 2: Population Risk and Resource Stratification Matrix
Category LEVEL 1:
Primary Prevention
(Low Resource Use)LEVEL 2:
Secondary Prevention
(Moderate Resource Use)LEVEL 3:
Tertiary
(High Resource Use)LEVEL 4: Catastrophic/Complex
(Very High Resource Use)Clinical Risk Low Risk Medium Risk High Risk 2 High Risk 1 Goal To prevent onset of disease and maintain current health status. To treat a disease and avoid serious complications. To treat the late or final stages of a disease and minimize disability. May range from restoring health to only providing comfort care. Healthy patients with no known diagnosis or complex treatments; however, may demonstrate warning signs or potentially significant risk factors.
Example:
∙ Healthy
∙ Blood glucose and lipids rising, but still within desired parameters.
∙ BMI elevated
∙ Smoker
Includes some patients with medical or mental health conditions considered to be well controlled or at low risk for adverse health event.
Example:
Otherwise healthy patients, including:
∙ Those who use ≤ 2 SABA dispenses in 12 months and not on an ICS
∙ Those with all HgA1C < 7.7 in 12 months and not on insulin
∙ Those who only receive monotherapy for blood pressure management
∙ Those who are receiving treatment for LTBI and have AST < 2Xs normal elevation
∙ CCCMS without medications or on a KOP SSRI only
Basic, uncomplicated nursing care of largely well population; prevention and wellness; stable, uncomplicated chronic disease; episodic care of acute injury or illness; routine care in primary care clinic; annual or semi-annual patient service plans (PSP).
Example:
∙ Independent ADLs
∙ Short term (no more than 4 weeks duration) conditions
∙ KOP medications or medication line no more frequent than BID
∙ Vital signs monthly, vital signs weekly for short period of time (no more than 4 weeks duration)
∙ Labs/procedures/treatments performed no greater than monthly except for situations as specified for short-term (no more than 4 weeks duration) medical conditions
∙ Independent wheelchair user and engaged in self management without complications.
Note: incarcerated person should be in an ADA designated facility.
∙ Independent prosthetic devices and engaged in self management without complications.
Note: incarcerated person should be in an ADA designated facility.
∙ Attend activities independently and willingly
∙ No thought disorder; no withdrawn or intrusive behavior
∙ Oriented, interacts appropriately
∙ Manages incontinence including indwelling catheters
[Most low risk patients will fall into this level/category.]Have diagnosis and/or complex treatment; at higher risk for complications or potentially significant risk factors.
Example:
∙ Blood sugar and lipids not within desired parameters
∙ No support system
Includes patients with at least one chronic condition or mental health condition at higher risk for adverse health event.
Example:
∙ One or more chronic illness, based upon prescribed medications and/or laboratory tests.
∙ CPAP with oxygen at night only
∙ Episodic oxygen therapy for acute asthma or respiratory condition no greater than twice a month
∙ CCCMS on NA/DOT medications
∙ MH High Utilization
∙ Permanent ADA with history of/or current complications
∙ Pregnancy
Low intensity nursing care of stable, chronic disease; functional limitations compensated by adaptive equipment; maintenance of status; prevention of exacerbation; symptom control and pain management; uncomplicated wound care (time-limited); uncomplicated chemo/radiation therapy; Quarterly patient service plans (PSP).
Example:
∙ Independent in ADLs
∙ Stable, chronic disease
∙ Routine Medication Line: NA/DOT, injectable and or transdermal medications. May also have KOP medications.
∙ PRN Medications: requires oral PRN medications including narcotics for significant physical symptoms.
∙ Vital signs daily for short period of time (not to exceed 2 weeks) to ascertain stability excluding vital signs required for certain medication such as pulse before administering Digoxin.
∙ Vital signs no greater than weekly excluding vital signs taken as required for certain medication such as pulse before administering Digoxin.
∙ Labs/procedures/treatments performed no greater than weekly for stable chronic disease excluding diabetic checks.
∙ Prosthetic devices with minimal assistance.
∙ Thought disturbance, effectual disturbance, withdrawn or intrusive behaviors requiring only redirection.
∙ Independent management of colostomies, incontinence including catheterization, and tracheotomies
∙ Wound care: uncomplicated, time-limited wound care or chronic stasis ulcers with independent dressing changes.
[Most medium risk patients will fall into this level/category.]Has diagnosis, complex treatment, and complications or potentially significant risk factors-goal is to prevent further complications.
Example:
∙ Has diabetes with early renal disease, coronary artery disease, failing eyesight and no support system.
∙ Three ER visits and two hospitalizations in past year
∙ Needs assistance with ADLs
Includes patients who are high risk priority 2 and trigger only 1 flag from the selection criteria below:
Example:
∙ Medications associated with important diagnoses which, if not taken, may lead to a serious adverse event (e.g., immunosuppressant, chemotherapy)
∙ 2 or more inpatient admissions in a 12 month period
∙ 2 or more appointments to “high risk” specialist(s) (e.g., oncologist, vascular surgeon) in a 6 month period
∙ 65 years of age or older
∙ 3 or more Mental Health Higher Level of Care admissions in the last 12 months
∙ In the CTC, OHU or SNF for 50% or more of the last 90 days
∙ Prescribed 10 or more medications
Medium intensity nursing care of complex, stable or at risk patients; uncomplicated post-surgical care; dementia, paraplegia, or hemiplegia able to participate in self-care; uncomplicated wound care (high risk for skin breakdown); Outpatient Housing Unit (OHU) placement; Monthly or every 2 month patient service plans (PSP).
Example:
∙ Requires some assistance with ADLs (bathing, feeding, dressing, toileting, etc.)
∙ Unstable, chronic disease, may require OHU placement
∙ Routine Medication Line: 3 or more NA/DOT, injectable and or transdermal medications. May also have KOP medications.
∙ PRN Medications: requires oral PRN medications including narcotics for significant physical symptoms.
∙ Colostomy and/or Foley catheter care requiring nurse intervention. If patient is stable and treatment is ongoing consider for placement in LTC facility.
∙ Frequent incontinency requiring nursing intervention – criteria for LTC
∙ Episodic incontinence including colostomies and indwelling catheters requiring nursing intervention – criteria for unstable chronic disease.
∙ Initiation of involuntary medications
[Most high risk priority 2 patients will fall into this level/category.]Have very severe illness or condition and potentially significant risk factors. May have high costs with limited or no opportunity for improvement, stabilization, or cost control (i.e., end of life care, premature labor pregnancy complications).
Example:
∙ Diagnosed with lung cancer
∙ Recent myocardial infarction
∙ Progression to ESRD with renal dialysis
∙ Amputation of one leg
∙ Blind
Includes patients who are high risk priority 1 and trigger at least 2 flags from the selection criteria below:
Example:
∙ Medications associated with important diagnoses which, if not taken, may lead to a serious adverse event (e.g., immunosuppressant, chemotherapy)
∙ 2 or more inpatient admissions in a 12 month period
∙ 2 or more appointments to “high risk” specialist(s) (e.g., oncologist, vascular surgeon) in a 6 month period
∙ 65 years of age or older
∙ 3 or more Mental Health Higher Level of Care admissions in the last 12 months
∙ In the CTC, OHU or SNF for 50% or more of the last 90 days
∙ Prescribed 10 or more medications
High intensity, direct, total and/or specialized nursing care of complex, complicated, unstable or high risk patients; daily care plan updates; significant dementia, paraplegia, hemiplegia, or quadriplegia unable to participate in self-care; Care management required; Inpatient level of care.
Example:
∙ Requires significant assistance or total care with ADLs (bathing, dressing, feeding, toileting, turning and positioning, ambulation and range of motion)
∙ Level of Care: Acute medical or mental health inpatient, skilled nursing facility, LTC facility, Hospice or end of life care.
∙ NA/DOT medications only
∙ IV therapy, blood and blood product transfusion, IV meds
∙ Daily vital signs, procedures or treatments for acute and unstable chronic disease excluding diabetic checks.
∙ Severe ill effects from chemotherapy and/or radiation therapy
∙ Complicated wound care to include use of wound vac.
∙ Frequent suctioning
∙ Tracheostomy with extensive nursing intervention
∙ NG tube or G-tube feedings requiring total nursing intervention
∙ Routine incontinence requiring total nursing intervention.
∙ Colostomy and/or Foley care that must be done by nurse
∙ Medical restraint (posey, soft wrist restraints, etc.) required for protection of self or to stabilize medical devices/dressings/tubes
∙ Confusion and disorientation secondary to dementia
∙ Thought disturbance, effectual disturbance, withdrawn or intrusive behavior that requires seclusion or restraint for protection of self and others
∙ Self-injurious behavior, 1:1 observation (acute mental health only)
∙ High Risk Pregnancy
[Most high risk priority 1 patients will fall into this level/category.]
3.1.7 Care Management/Care Coordination
-
Procedure Overview
-
This procedure describes the systems and processes California Correctional Health Care Services (CCHCS) staff shall utilize to assist patients in reaching an optimum level of wellness and functional capability as a means of achieving patient wellness and autonomy through advocacy, communication, education, identification of service resources, and service facilitation. Care Management/Care Coordination services are best offered in a climate that allows direct communication between the Care Manager/Care Coordinator, the patient, and appropriate service personnel in order to optimize the outcome for all concerned. This procedure also specifies roles and responsibilities for key staff involved in the care coordination system.
-
-
Responsibility
-
Statewide
-
California Department of Corrections and Rehabilitation (CDCR) and CCHCS departmental leadership at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial, and clinical systems are in place and appropriate tools, training, technical assistance, and levels of resources are available to ensure the Care Management/Care Coordination system is successfully implemented and maintained.
-
-
Regional
-
Regional Health Care Executives are responsible for implementation of this procedure at the subset of institutions within an assigned region.
-
-
Institutional
-
The Chief Executive Officer (CEO) has overall responsibility for implementing and ongoing oversight of the Care Management/Care Coordination system at the institution and patient panel level. The CEO delegates decision-making authority to the Chief Nurse Executive (CNE) for daily operations of the Care Management/Care Coordination system and ensures adequate resources are deployed to support the system including, but not limited to, the following:
-
Access to and utilization of equipment, supplies, health information systems, patient registries and summaries and evidence-based guidelines.
-
Ensure that at least twice monthly, each Primary Care Team (PCT) conducts a Population Management Working Session utilizing tools such as Dashboards, Master Registries, and Patient Summaries to address concerns related to potential gaps in care and improve patient outcomes including, but not limited to, ensuring:
-
Patients are linked to necessary services through Care Management/Care Coordination.
-
All patients have timely access to appropriate care.
-
Patients who are at increased risk for developing serious health complications and patients with chronic health care needs are identified and monitored.
-
Preventive services are provided.
-
Health education, wellness, and self-management are provided.
-
-
Adequately prepare new Care Team members to assume team roles and responsibilities with regard to Care Management/Care Coordination.
-
Assess competence of existing Care Team members.
-
Update procedures, roles and responsibilities as new tools and technology become available.
-
Work with custody staff to minimize patient movement that results in changes to a patient’s panel assignment in complex cases.
-
Annual Review of Adult Immunization Schedule Chart and the Preventive Services Matrix. At a minimum, nursing leadership shall arrange a meeting with the Chief Physician and Surgeon annually to review these documents and update them as needed based on current recommendations from CCHCS headquarters (HQ), the Centers for Disease Control and Prevention, and the United States Preventive Services Task Force.
-
-
The CEO shall:
-
Establish relationships with community agencies, specialists, hospitals, and others to ensure connectivity within a network of service delivery points.
-
Understand the requirements of each of the service delivery points to ensure coordination of care.
-
Convey information about correctional health care and our processes to the service delivery network.
-
-
The CNE is responsible for the overall daily operations, oversight, and management of the Care Management/Care Coordination systems, processes, and resources, including personnel.
-
The CEO and all members of the institution’s leadership team are responsible for establishing an organizational culture that promotes teamwork across disciplines.
-
The Chief Medical Executive is responsible for the overall medical management of patients and ensures resources are available to meet the needs of the population.
-
The Supervising Registered Nurse and Chief Physician and Surgeon shall meet to review the Care Teams’ performance, including the overall quality of services, health outcomes, and level of care utilization and shall utilize Dashboards, Master Registries, Patient Summaries, and decision support tools to address or elevate issues as necessary.
-
Institutional leadership shall develop a Local Operating Procedure to address standing orders for immunization, preventive screenings, and preventive medications based on current recommendations from CCHCS HQ, the Centers for Disease Control and Prevention, and the United States Preventive Services Task Force. As part of care management, the nursing staff may administer immunizations, order certain screening tests, and start preventive medications at the time of a Care Management/Care Coordination visit as long as there is a valid standing order.
-
-
-
Procedure
-
Responsibilities of the Nurse Care Manager/Care Coordinator
-
All members of the PCT assume responsibility for all aspects of patient care across the continuum and throughout the course of the patient’s daily activities. Members work to anticipate patient care needs, develop treatment plans, and coordinate care to ensure that services are provided without interruption or delay. Each member of the PCT is accountable for developing relationships and networking with other members of the health care community to ensure the overall health care needs of the patient are met. The PCT shall follow the patient through the systems of care to ensure their needs are met and improve patient outcomes.
-
The Registered Nurse shall:
-
Provide overall direction for the assigned Care Team patient panel. Assess, plan, implement, monitor, and evaluate patient care for an assigned patient group composed of patients mostly in the secondary prevention and tertiary groups.
-
Collaborate with the patient one-on-one to develop and maintain their treatment plan.
-
Interface with, and refer patients to other supportive services as appropriate.
-
Review data pertaining to the entire patient panel and coordinate patient care activities and education.
-
Direct the members of the care coordination team (e.g., Licensed Vocational Nurse [LVN], Psychiatric Technician [PT], and Office Technician) when coordinating the care of their patient panel.
-
-
The LVN/PT shall:
-
Perform patient care activities within the assigned primary care patient panel. The LVN/PT may also be assigned a subpanel of patients composed of patients in the primary prevention group for more comprehensive targeted nursing care (e.g., wound management, medication compliance).
-
Collaborate with patients one-on-one regarding their treatment plan. The LVN/PT may provide education and services to patients in both one-on-one and group settings. The LVN/PT interfaces with and refers patients to other supportive services, as appropriate.
-
Use their skills according to their scope of practice, including collecting data, documentation, communicating patient information to the Registered Nurse (RN) as needed, and providing patient education.
-
Assist in the development of a treatment plan based on the information gathered. The development of the treatment plan shall include self-management goals in conjunction with the Care Team and implementation of interventions.
-
Work with the RN to ensure prioritization of patient care management services.
-
-
Scheduling Support Staff shall:
-
Schedule appointments as necessary to meet the needs of the patient’s treatment plan.
-
Assist in the completion of requested audits for the Care Team.
-
Run reports from the Chronic Care Master Registry and other databases as needed by the Care Team.
-
Use and maintain a system to track scheduling and completion of visits, tests, studies, consults, and educational training.
-
-
-
Care Management Process
-
Care management begins when the patient enters into CDCR and continues throughout their stay within CDCR. The process is continuous and it transitions across institutions and patient care settings. The care coordination process is also instrumental in ensuring that the patient’s treatment plan transitions to the community upon parole or discharge.
-
Initial Care Management Visit (Receiving & Release)
-
All patients shall have an initial nurse care management visit within 30 days of arrival receiving & release.
-
On a daily basis, the Primary Care Nurse identifies new arrivals to the panel, evaluates their health care needs through the use of the Master Registries, Patient Summaries, existing treatment plans, and other documents, and determines the priority and timeline of initial care management visit.
-
The Primary Care Nurse collaborates with the PCT and other disciplines to initiate the coordination of the patient’s health care services.
-
-
Before the Care Management/Care Coordination Visit
-
The Primary Care Nurse/Care Coordinator shall:
-
Review the patient summary sheet, health record, and other available documents to identify the preventive services, immunization, and medication the patient has previously received or is currently prescribed.
-
Document this information in the treatment plan.
-
Identify recommended immunization and preventive services to discuss during the nursing care management scheduled visit.
-
Identify chronic disease history and current status.
-
Identify current risk stratification level.
-
Identify any effective communication barriers/disabilities and prepare a plan to meet those needs during the visit.
-
Prepare for any upcoming specialty care visits by collaborating with Specialty Services nurses to identify specific patient education materials, need for procedure preparation, and after care needs.
-
-
During the Care Management/Care Coordination Visit
-
The Primary Care Nurse/Care Coordinator shall provide the following services to the patient as directed by the patient’s current needs, goals and their treatment plan.
-
Perform comprehensive or focused assessment depending on nature of care management visit.
-
A comprehensive assessment shall be completed during initial and annual visits.
-
A focused assessment shall be completed during all other visits.
-
-
Ask the patient to describe their health status and progress since the last visit.
-
Discuss assessment findings.
-
Compare to previous status.
-
Discuss observed progress or regression from last patient objectives/goals.
-
Ask the patient to explain their goals for this visit.
-
Discuss nurse-suggested goals.
-
Discuss disease process including pathophysiology appropriate to the patient’s understanding.
-
Discuss treatment and medication compliance (e.g., self-care, Keep-on-Person).
-
Discuss outcomes of any test results or studies.
-
Solicit for patient questions, concerns, and other factors in their life that they think might have an effect on their health care, psychosocial response to illness or health situation (e.g., anxieties/fears, whether rational or not). Respond with active listening and information.
-
Discuss the need for any future tests or treatments.
-
Educate on warning signs and symptoms as well as how to seek emergency medical care.
-
Discuss opportunities for behavioral changes.
-
Make and document new goals with the patient.
-
Discuss patient status relative to the care management level, including celebrating improvements and goal achievements.
-
Discuss nursing and treatment plan implications of changing the care management level due to today’s assessment findings.
-
-
After the Care Management/Care Coordination Visit
-
The Primary Care Nurse/Care Coordinator shall:
-
Document the interventions provided in the appropriate locations in the health record (e.g., Patient Immunization Record, Problem List, Interdisciplinary Progress Note, treatment plan) such as:
-
The topic and counseling provided (e.g., smoking cessation).
-
Immunization administered.
-
Medication and/or treatments ordered.
-
Request For Services or laboratory slip for recommended screening.
-
The referrals to the PCP regarding recommended screenings and preventive medications.
-
The referral to other health care disciplines (e.g., mental health, dental).
-
The follow-up Care Management/Care Coordination visit.
-
-
Communicate any nurse-suggested changes to the treatment plan, solicit for the patient’s input on changes to the treatment plan, and engage the patient in a discussion of how to reconcile and synthesize the two input sources (nurse and patient).
-
Ask the patient which objectives they want to commit to working on until the next visit. Note the patient’s priority objectives and discuss steps to reach the objectives.
-
Update the treatment plan and provide a printed copy to the patient.
-
Confirm the next appointment interval with the patient and schedule an appointment.
-
Communicate the results of the visit and any new plans to the PCT, and other health care staff and/or disciplines to ensure coordination (e.g., during huddle, ad-hoc clinical discussions, or Population Management Working Sessions).
-
-
-
Management and Sustainability
-
Care Team
-
At least monthly as part of the Population Management Working Sessions, the Care Team shall evaluate the effectiveness and efficiency of the Care Management/Care Coordination process. The Care Team shall:
-
Review population management performance trends and take action to improve care.
-
At a minimum, review Health Care Services Dashboard information but may also consider monitoring reports, internal audits and surveys, and reviews by stakeholders such as court experts, the Prison Law Office, and the Office of the Inspector General.
-
Evaluate patients’ responses and the effectiveness of current treatment plan(s) and adjust plan(s) as appropriate. The Care Team shall endeavor to anticipate the educational, treatment and/or diagnostic needs of the patient based on identified trends, and adjust treatment plan(s) to improve outcomes for individual patients and patient populations.
-
Review patient registry flags and alerts indicating abnormal clinical findings, specialty services reports, community hospital reports, sentinel events, etc.
-
Review access to care data and statistics.
-
Review potentially avoidable hospitalizations for any patients within the panel.
-
Review new patients and patients leaving the panel in the context of their impact on the overall risk stratification of the patient panel (e.g., workload management and resource demand).
-
-
Institution Leadership Team
-
Institution leadership team shall periodically review the composition of patient panels, particularly relative to the number and proportion of patients that fall into each risk category, to ensure available staff resources are distributed in order to provide the required population management services and procedures, templates, and roles and responsibilities are updated as new tools and technology become available.
-
-
System Monitoring
-
The CEO and the institution leadership team shall review institution-wide care coordination data monthly in the context of the Quality Management Committee and subcommittee meetings. To ensure the efficiency of the care coordination system, the institution leadership team shall:
-
Periodically evaluate the care coordination program through methods such as a review of trends in:
-
Possible avoidable hospitalizations.
-
Sentinel events.
-
Utilization of episodic care.
-
Emergency Department or Triage and Treatment Area visits.
-
-
Take effective action to remedy problems by including, but not limited to, creating or revising decision support tools, updating desk procedures, and redesigning orientation and training strategies.
-
Re-validate problematic data monthly until the program goals are met.
-
-
Training and Decision Support
-
The CEO and the institution leadership team shall establish an orientation and training program to ensure that all staff serving as members of a Care Team or supporting Care Management/Care Coordination functions fully understand their roles and responsibilities prior to assuming their duties. Elements of the program shall include, but are not limited to, the following:
-
Reviewing expectations described in this procedure.
-
Training in care coordination for all institution staff. A system for the orientation, mentoring, and cross-training of all critical positions in the care coordination team shall be maintained.
-
Developing or adopting decision support tools (e.g., desk procedures) to prompt health care staff in different roles in Care Management/Care Coordination to fulfill their roles and responsibilities, including prompting clinic staff to communicate clearly to other members of the Care Team and reminding staff of new disease management procedures.
-
Providing ongoing training for staff involved in Care Management/Care Coordination regarding changes to the primary and chronic care programs and processes as they evolve as well as periodic refresher training on their particular roles and responsibilities.
-
Utilizing new information systems or technology that may increase the efficiency or effectiveness of Care Management/Care Coordination processes or forums.
-
Incorporating updates in clinical practice, including new CCHCS guidelines, standing orders, nursing protocols, industry best practices, and findings in clinical literature.
-
-
-
-
References
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.1, Complete Care Model
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.3, Care Teams and Patient Panels
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.6, Population and Care Management Services
-
Agency for Healthcare Research and Quality – Patient Centered Medical Home Resource Center https://www.ahrq.gov/
-
National Committee for Quality Assurance – Patient-Centered Medical Home Recognition
http://www.ncqa.org/Programs/Recognition/Practices/PatientCenteredMedicalHomePCMH.aspx -
Robert Wood Johnson Foundation – Improving Chronic Illness Care
https://www.rwjf.org/content/dam/farm/books/books/2007/rwjf12990 -
Institute of Medicine – Crossing the Quality Chasm – A New Health System for the 21st Century
http://iom.nationalacademies.org/Reports/2001/Crossing-the-Quality-Chasm-A-New-Health-System-for-the-21st-Century.aspx -
Centers for Disease Control and Prevention – Adult Immunization Schedule by Age
https://www.cdc.gov/vaccines/schedules/hcp/imz/adult.html -
United States Preventive Services Task Force – A & B Recommendations
https://www.uspreventiveservicestaskforce.org/uspstf/recommendation-topics/uspstf-a-and-b-recommendations
-
-
Revision History
-
Effective: 06/2016
Revised: 05/16/2023
-
3.1.8 Reception Center
-
Procedure Overview
-
The California Correctional Health Care Services (CCHCS) and California Department of Corrections and Rehabilitation (CDCR) shall conduct a Reception Center (RC) health care assessment (RC-HCA) as part of the RC initial intake process for each person newly committed to the CDCR custody. The goal of the RC-HCA process is to evaluate newly arriving patients in a timely manner, identify appropriate provider resources and patient acuity, expedite the transfer of high-risk patients to endorsed institutions, initiate necessary health care interventions, and ensure processing is based on the health care needs of the patient.
-
The RC-HCA shall be conducted at specifically designated RC institutions; however, under exigent circumstances, patients may be transferred to other institutions in order to have specific needs addressed, and this procedure shall be followed.
-
-
Responsibility
-
Statewide
-
CCHCS and CDCR departmental leadership at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial, and clinical systems are in place to fully maintain the RC-HCA process.
-
-
Regional
-
Regional Health Care Executives are responsible for the administration of this procedure at the subset of institutions within an assigned region.
-
-
Institutional
-
The Chief Executive Officer (CEO) is responsible for the ongoing oversight and maintenance of a system to provide management of the patient care services, including but not limited to, the RC-HCA process, at each designated institution.
-
The CEO delegates decision-making authority to designated institutional health care executives for daily operations and ensures adequate resources are deployed to support the RC-HCA process.
-
The CEO and all members of the institution’s leadership team are responsible for ensuring necessary resources are in place to support the success of this procedure at all levels of the institution.
-
The CEO shall ensure mechanisms (e.g., forums) are in place to coordinate the RC-HCA process with the CDCR staff responsible for the patient’s correctional RC process. A standing committee reporting to the Institution Quality Management Committee shall be designated to oversee implementation, sustainability, and continuous improvement of the RC initial intake process, and the CEO shall ensure that this subcommittee is operating in accordance with policies, procedures, and departmental rules.
-
The Chief Nurse Executive, Chief Medical Executive, Chief of Mental Health, Chief Psychiatrist, Supervising Dentist, and Chief Support Executive shall maintain a multidisciplinary approach to ensure that health care staff participating in the RC-HCA process shall have, at a minimum:
-
Training in the policies and procedures during orientation; whenever new policies, procedures, or equipment are issued; and as needed.
-
Demonstrated competency in the tasks necessary to complete the RC-HCA process prior to their performance of the tasks outlined in this procedure.
-
An established training file containing documentation of health care staff training, and initial and ongoing competency evaluations or professional practice evaluations for health care staff who perform any task outlined in this procedure.
-
-
-
-
Procedure
-
Reception Center Initial Health Screening and Triage
-
Newly committed individuals to CDCR shall have an RC initial health screening and triage conducted by licensed nursing staff upon arrival at the RC. The purpose of the screening is to identify immediate needs and to ensure continuity of care including medications, treatments, and accommodations.
-
The RC initial health screening and triage shall be accomplished prior to the patient being placed in housing. The RC initial health screening and triage serves as the basis for the RC Focused Health Care Assessment completed by the Primary Care Provider (PCP).
-
The RC initial health screening and triage of the patient includes the following elements at a minimum; additional assessments may be conducted as indicated by the patient’s clinical presentation and identified health care needs.
-
Health information gathering
-
Each patient shall have a face-to-face interview conducted by a licensed nurse which shall include, at a minimum:
-
A review of health records arriving with the patient.
-
A review of the patient’s immunization records.
-
A brief health history taken and documented in the health record.
-
A review of the patient’s medication history as documented in health records. If the arriving records are missing, incomplete, or inconsistent with the patient’s reported medication history, nursing staff shall contact the sending facility/agency and document the findings in the health record.
-
-
Vital signs (including blood pressure, temperature, pulse, and respirations).
-
Actual measurements of weight and height. In unusual circumstances (i.e., the patient refuses), stated measurements can be taken and shall be clearly documented in the health record as “stated by the patient.
-
”Tuberculosis screening using the procedures outlined in the current CCHCS Care Guide: Tuberculosis, for symptom screening.
-
For patients presenting with symptoms of opioid withdrawal, utilize the Clinical Opioid Withdrawal Scale (COWS). Patients with a score of eight and greater shall be referred to the Triage and Treatment Area (TTA) for withdrawal management (Refer to the current CCHCS Care Guide: Intoxication & Withdrawal).
-
For patients presenting with signs of alcohol withdrawal, utilize the Clinical Institute Withdrawal Assessment of Alcohol-Revised (CIWA-AR). Patients with a score of 10 and greater shall be referred to the TTA for withdrawal management (refer to the current CCHCS Care Guide: Intoxication & Withdrawal).
-
Finger stick blood sugar shall be recorded for each patient with a stated history or recorded diagnosis of diabetes.
-
A pain assessment shall be performed on each patient whose history indicates a recent inpatient admission, procedure, or upon self-report of pain.
-
-
If the RC initial health screening and triage is conducted by licensed nursing staff who is not a Registered Nurse (RN) and the patient answered “yes” to any questions, an RN shall review the data collected, conduct an assessment, determine the appropriate disposition of the patient pursuant to this policy, and document in the health record.
-
Diagnostic Screening Tests and Assessments
-
Each patient shall be offered the following screening tests based on the Opt-Out screening method:
-
Men less than or equal to 44 years old shall be screened for Chlamydia and Gonorrhea.
-
Women less than or equal to 44 years old shall be screened for Chlamydia, Gonorrhea and Trichomonas.
-
Human Immunodeficiency Virus (HIV) antibody screening.
-
Serum pregnancy test for females less than 60 years old.
-
Varicella Immunoglobulin G (IgG).
-
Coccidioidomycosis (cocci) delayed-type hypersensitivity skin test for males 18 to 64 years of age, unless prior documented positive result, history of cocci disease, or a medical condition that would otherwise restrict placement in Cocci 1 area (e.g., immunocompromised).
-
Rapid Plasma Reagin (RPR) syphilis test.
-
Papanicolaou test (cervical cytology screen) for all females as clinically appropriate (i.e., cervix intact).
-
Hepatitis C Virus (HCV) antibody with reflex to HCV viral load.
-
Interferon-Gamma Release Assays (IGRA) blood test.
-
Hemoglobin A1C (HbA1c) for patients with a history of diabetes.
-
Hepatitis B surface antigen (HBsAg), surface antibody (HBsAb) and Hepatitis B core antibody (HBcAb).
-
Urine Drug Screen (UDS).
-
-
Each patient arriving to an institution on Medication Assisted Treatment (MAT) shall have the following laboratory tests ordered:
-
Complete Blood Count without Differential (CBC, w/o Diff).
-
Comprehensive Metabolic Panel (CMP).
-
UDS.
-
Electrocardiogram (EKG) for individuals arriving on methadone.
-
-
Prior to the laboratory performing the tests, the patient shall be provided with education about the tests and informed that testing is also available upon patient request throughout incarceration.
-
Special Requirements for High-Risk Disease Screening/Testing.
-
Cocci delayed-type hypersensitivity skin test screening, administration or declination, and results shall be reported and documented in the health record and the Cocci Screening and Surveillance System (refer to the current CCHCS Care Guide: Coccidioidomycosis).
-
Patients shall be informed that the purpose of the cocci skin test is to ensure those at higher risk of cocci disease (negative result) will not be housed in the institutions with the highest risk.
-
Patients shall be offered an IGRA blood tests (refer to the current CCHCS Care Guide: Tuberculosis).
-
-
If the patient declines a screening test, the CDCR 7225, Refusal of Examination and or Treatment, shall be signed by the patient, and the refusal documented in the health record. If the patient refuses to sign the form, the refusal shall be documented in the health record with two witness signatures.
-
Nursing staff shall identify any recommended preventive services and immunizations based on the current recommendations from the Centers for Disease Control and Prevention and the United States Preventive Services Task Force’s recommendations on immunizations, and document in the health record.
-
A CDCR 7385, Authorization for Release of Protected Health Information, shall be presented for the patient’s signature in order to obtain previous health records (e.g., substance use disorder treatment records or HIV test results).
-
Nursing staff shall educate and provide the patient with information about how to access health care services at the institution. This education shall be documented in the health record and shall include, at a minimum, the education of the following topics:
-
The Patient Orientation to Health Care Services Handbook.
-
Assignment to a patient care team, some care team members provide care via telehealth.
-
Provisions of telehealth.
-
Patients’ rights.
-
How to submit a CDCR 7362, Health Care Services Request Form.
-
Over-the-counter products available from the canteen.
-
Durable Medical Equipment (DME) as applicable.
-
Contraception information for female patients.
-
-
Nursing staff shall initiate the orders for diagnostic and preventive services identified in Sections (c)(1)(C)3.a. through h. above.
-
-
Durable Medical Equipment
-
Patient DME needs shall be properly addressed in accordance with the HCDOM, Section 3.6.1, Durable Medical Equipment and Medical Supply.
-
-
Disposition
-
An RN shall review relevant data for each person newly committed to determine a disposition and if a referral to a provider or a higher level of care is required. The review and disposition shall be documented in the health record.
-
Each patient’s priority disposition shall be determined based upon responses to questions on the RC initial health screening and triage, which are used to create automated clinical rules within the Electronic Health Record System, as either high or low priority.
-
Medical Emergent: For patients with identified emergent medical needs, staff shall initiate an emergency medical response.
-
Medical Urgent: Patients with identified urgent medical needs shall be referred to a PCP in the reception center and/or transported to the TTA or higher level of care for immediate evaluation and treatment.
-
Mental Health Emergent: Patients identified as having an emergent mental health condition, such as suicidal ideation or current self-harm, shall be referred immediately to mental health services and transported to the TTA for further evaluation and consultation with a mental health clinician or PCP.
-
Mental Health Urgent: Patients identified as having an urgent mental health condition shall be referred to mental health services and evaluated as required by the Mental Health Services Delivery System Program Guide.
-
Dental Services Emergent: Patients identified as having an emergent dental condition for which evaluation and treatment are immediately necessary to prevent death, severe or permanent disability, or to alleviate or lessen disabling pain shall be immediately referred to dental services. If outside of normal business hours, the patient shall be transported to the TTA for further assessment in accordance with the HCDOM, Chapter 3, Article 3, Section 3.3.5.9, Dental Emergencies.
-
Dental Services Urgent: Patients identified as having an urgent dental condition shall be referred to dental services within one business day.
-
Arriving on MAT: Patients identified as arriving on MAT shall be referred to:
-
A Licensed Clinical Social Worker within 14 calendar days.
-
An Addiction Medicine Central Team (AMCT) provider within 14 calendar days.
-
A Narcotic Treatment Program for a consult that must take place within four calendar days for patients arriving on methadone for MAT.
-
A PCP shall be contacted to place a medical hold.
-
A PCP shall be contacted to place bridge orders for all MAT medications to ensure continuity of care (e.g., if a patient arrives on methadone, a 3-day bridge order is required).
-
-
An AMCT provider by immediately calling the AMCT for pregnant women with a COWS score greater than four.
-
-
Patients who have been receiving prescription medications shall have their prescription medications ordered within eight hours of arrival to prevent an interruption in receiving medication.
-
Patients with a health care condition not requiring an emergent or urgent referral shall be scheduled for an appointment with the appropriate health care provider using the timeframes outlined below:
-
PCP: Focused Health Care Assessment within five working days.
-
Mental Health: Mental Health Screening within five working days.
-
Dental: RC Dental Screening within 60 calendar days.
-
-
-
-
-
Reception Center Focused Health Care Assessment
-
Each person newly committed to the CDCR shall have a Focused Health Care Assessment performed by a PCP within five working days of arrival at the RC. The purpose of this assessment is to identify patients who are acutely ill, infectious, or those with clinically significant health care needs to ensure continuity of care.
-
If there is not enough time to complete all health care assessments for patients who are designated high priority the day that patients arrive at the RC, the assessment shall be completed the following business day.
-
The Focused Health Care Assessment shall include, at a minimum, the following:
-
A review of the RC initial health screening and triage.
-
A review of the patient’s immunization records through Cerner CA Immunization Registry or other means.
-
A review of available health records including, but not limited to, a review of diagnostic testing.
-
A consultation of the Controlled Substance Utilization Review and Evaluation System database pursuant to California Health and Safety Code, Section 11165.4.
-
A face-to-face interview with the patient. The purpose of the interview shall be to identify:
-
Current or recent symptoms, treatment, and medications.
-
Significant past medical history, to include surgical history.
-
Significant medical, family, and social history.
-
Risk factors for chronic disease or adverse health outcomes (e.g., history of tobacco use, history of substance use).
-
Significant disabilities and the need for reasonable accommodations or DME.
-
-
A physical examination shall be targeted based on the review of the records, the history obtained during the face-to-face interview, and identified or stated risk factors. If there are no identified or stated risk factors after a review of records and history, at a minimum, an exam of the heart and lungs shall be completed and documented in the health record.
-
The PCP shall initiate a treatment and care plan based on the information obtained from the diagnostic test and assessments which shall include, at a minimum, the following:
-
Orders for diagnostic screening tests and assessments, if not ordered during the RC initial health screening and triage (refer to Sections (c)(1)(C)3.a.1) through 10) and (c)(1)(C)3.b.1) through 3) above for listing). If the patient opted-out of any of the screening tests or assessments during the RC initial health screening and triage, the PCP shall provide patient education and document the patient’s response in the health record.
-
Orders for additional clinically indicated diagnostic testing based on the health history and assessment performed by the PCP.
-
Routine preventive services (e.g., age-based lipid screening, immunizations, cancer screens [i.e., based on age, sex and other risk factors]) and routine screening related to chronic conditions when no symptoms are present (e.g., retinal and podiatric foot exams for diabetic patients) shall be performed at the endorsed institutions.
-
Initiation of the patient’s problem list in the health record.
-
Completion of a Request for Service (RFS) for any clinical condition that requires an emergent or high priority specialty consultation in accordance with the HCDOM, Section 3.1.11, Outpatient Specialty Services.
-
Routine and medium priority referrals for specialty services, in general, shall be deferred as clinically indicated, until the patients are transferred to their endorsed institutions.
-
Patients with pending high priority specialty services shall be placed on a medical hold to prevent transfer and discontinuity of care in accordance with the HCDOM, Section 1.2.14, Medical Classification System.
-
-
Orders for follow-up appointments as clinically indicated for the care and treatment of the patient’s identified health care needs.
-
-
The PCP shall provide patient education as indicated, which at a minimum, shall include the following:
-
Review of lab results, physical exam findings, and plan of care with the patient.
-
How to access health care and return to the clinic as needed.
-
-
Identification of the patient’s medical classification factors and completion of a Medical Classification Chrono in accordance with the HCDOM, Section 1.2.14, Medical Classification System.
-
If laboratory results or other diagnostic results are received after the RC Focused Health Care Assessment has been conducted, the PCP shall evaluate the result and determine if a follow-up appointment is needed. The PCP, or designee, shall notify the patient of diagnostic test results in accordance with the HCDOM, Section 3.1.14, Laboratory Services.
-
The PCP shall ensure that each of the items above is documented in the health record.
-
-
-
Transfer to an Endorsed Institution
-
Continuity of health care shall be maintained pending the patient’s assignment and transfer to an endorsed institution. Each RC patient shall be assigned to a Primary Care Team (PCT) while awaiting transfer to an endorsed institution. The PCT shall be responsible for ensuring timely access to health care services, including, but not limited to:
-
Carrying out the plan outlined in the RC Focused Health Care Assessment including follow-up of RFS that were ordered during the patient’s RC Focused Health Care Assessment.
-
Review of, and action on, laboratory, diagnostic, and screening test results.
-
Provision of episodic and ongoing chronic health care.
-
Providing care management and care coordination services for the patient’s chronic conditions.
-
Providing appropriate preventive care services such as immunizations, cancer screening with mammography, and fecal occult blood tests, as well as care related to chronic conditions when no symptoms are present, such as retinal screens for patients with diabetes, as clinically indicated.
-
Routine health care services provided by the PCT shall not delay the patient’s transfer to an endorsed institution.
-
-
Continuity of mental health or dental care for each RC patient shall be provided, in accordance with established policies and procedures, pending the patient’s transfer to an endorsed institution.
-
-
-
References
-
Armstrong Remedial Plan, Armstrong v. Newsom, U.S. District Court of Northern California, Amended January 3, 2001
-
Clark Remedial Plan, Clark v. California, United States District Court of Northern California, March 1, 2002
-
California Health and Safety Code, Division 10, Chapter 4, Article 1, Section 11165.4
-
California Code of Regulations, Title 15, Division 3, Chapter 1, Article 1, Section 3002(b)(4)
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.14, Medical Classification System
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.1, Complete Care Model
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.5, Scheduling and Access to Care
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.6, Population and Care Management Services
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.11, Outpatient Specialty Services
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.14, Laboratory Services
-
Health Care Department Operations Manual, Chapter 3, Article 2, Section 3.2.6, Medication Continuity with Patient Movement: Transfer/Parole/Release
-
Health Care Department Operations Manual, Chapter 3, Article 3, Section 3.3.2.1, Initial Health Screening – Receiving and Release
-
Health Care Department Operations Manual, Chapter 3, Article 3, Section 3.3.5.9, Dental Emergencies
-
Health Care Department Operations Manual Chapter 3, Article 7, Section 3.7.1, Emergency Medical Response System
-
Health Care Department Operations Manual, Chapter 3, Article 8, Section 3.8.6, Tuberculosis Program
-
California Department of Corrections and Rehabilitation, Mental Health Services Delivery System Program Guide, and associated updates and policies
-
Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices, United States Preventive Services Task Force
-
California Correctional Health Care Services, CCHCS Care Guide: Tuberculosis-Surveillance
-
California Correctional Health Care Services, Patient Orientation to Health Care Services Handbook
-
-
Revision History
-
Effective: 12/2017
Revised: 07/28/2023
-
3.1.9 Health Care Transfer
-
Procedure Overview
-
The transfer of care between health care providers is a high-risk, complex, and multifaceted health care process. The goal of the California Department of Corrections and Rehabilitation (CDCR) is to perform each patient transfer in a manner that ensures the continuity of high-quality, safe care for each patient within CDCR. A transfer of care includes not only the processes and activities required to transport the patient, but also those activities, discussions, processes, and tasks required for admissions and discharges from differing locations, care settings, and levels of care within CDCR and external to CDCR. Transfers of care are accomplished by verbal communication between the sending and receiving care teams, with written documentation accompanying the patient as an adjunct to the conversation between health care providers.
-
Under the Complete Care Model, the health care transfer procedure is designed to ensure seamless continuity of patient care through the timely and complete communication of information between members of the patient’s care team using a series of standardized systems and processes across the continuum of patient care activities. These procedures and processes mitigate risk, promote patient safety, maintain continuity of care, improve access, and enhance professionalism, teamwork, and the formation of new patient-provider care relationships.
-
-
Responsibility
-
Statewide
-
CDCR and California Correctional Health Care Services (CCHCS) departmental leadership at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial, and clinical systems are in place, and appropriate tools, training, technical assistance, and levels of resources are available to ensure that the Health Care Transfer Procedure is successfully implemented and maintained.
-
CDCR and CCHCS departmental leadership at all levels of the organization, within the scope of their authority, are responsible for ensuring appropriate services are available for patients statewide, coordination of care, providing access to the appropriate level of care (LOC), development of decision support and technological tools, contact lists, and reducing the risks associated with handoff and transfer of patients between care teams, institutions, and levels of care.
-
The Headquarters Utilization Management Committee (HUMC) shall be responsible for ensuring processes are in place for continuity of care of high-risk medical patients and those with scheduled care needs as they transition from CDCR custody to the community (i.e., parole, probation, or discharge). CDCR and CCHCS Utilization Management (UM) collaborates with institutional staff, Division of Adult Parole Operations (DAPO), Post Release Community Supervision (PRCS), and community providers and agencies to ensure appropriate placement and services for patients who require ongoing care for chronic diseases after their release from CDCR facilities.
-
The statewide mental health program shall be responsible for ensuring processes are in place for continuity of care of mental health patients.
-
Health Care Placement Oversight Program (HCPOP), in coordination with the HUMC or the statewide mental health program (as applicable), is responsible for the endorsement of patients between health care facilities if the institution cannot provide appropriate, medically necessary health care treatment to the patient. HCPOP facilitates the transfer of a subset of complex, high-risk patients between institutions in collaboration with clinical services.
-
-
Regional
-
Regional Health Care Executives are responsible for implementation of this procedure at the subset of institutions within an assigned region and within the scope of their authority shall:
-
Ensure administrative, custodial, and clinical systems are in place, and appropriate tools, training, technical assistance, and levels of resources are available to ensure that this procedure is successfully implemented and maintained.
-
In coordination with the Regional Quality Management Support Unit (QMSU), monitor and analyze the transfer process metrics outlined in Section (b)(3)(D)1-2 for their subset of institutions in order to identify trends, process lapses, and opportunities to mitigate risk to patient care.
-
Regional QMSU shall report trended data, analysis, and process improvement activities to the designated statewide committee, no less than quarterly. Identified patient safety issues shall be addressed through the Health Care Incident Reporting System within the timeframes specified in the Health Care Department Operations Manual (HCDOM), Section
,1.2.6, Statewide Patient Safety Program.
-
-
Assist local leadership in the development of process improvement activities, best practices, and recommendations for improvement in the transfer process.
-
-
-
Institutional
-
The Chief Executive Officer (CEO) has overall responsibility for implementation and oversight of a system to provide management of the transfer of health care services within their institution. The CEO delegates decision-making authority to designated health care executives and leadership for daily operations of the health care transfer process, and ensures adequate resources are available to support the system.
-
The CEO and members of the local leadership team are responsible to ensure:
-
Resources are in place to support the successful implementation of this procedure at all levels including, but not limited to:
-
Institution level
-
Patient panel level
-
Patient level
-
-
Access to and utilization of equipment, supplies, health information systems, Patient Registries, Patient Summaries, and evidence-based guidelines.
-
Monitoring to assess the transfer process, which includes, but is not limited to, feedback to successfully ensure that continuity of care is achieved such as utilization of available patient management tools, including Patient Registries and the Electronic Health Record System (EHRS).
-
An orientation and training program is established and maintained at a local level to ensure that staff serving as members of an interdisciplinary care team or supporting health care functions understand their roles and responsibilities prior to assuming their duties. Elements of the program shall include, but are not limited to, the review of:
-
Expectations in this procedure.
-
Any changes to local transfer processes.
-
New information systems or technology that may increase the efficiency or effectiveness of transfer processes or forums.
-
Updates in clinical practice, including new CCHCS guidelines, standing orders, nursing protocols, industry best practices, and findings in clinical literature.
-
Training needs.
-
-
-
The institution’s health care transfer Local Operating Procedures (LOP) shall contain provisions for the implementation, structure, and operation of a quality assurance process.
-
Each institution shall designate a subcommittee in writing that has responsibility for the administration of the health care transfer process. The designated subcommittee shall report to the Institution Quality Management Committee (IQMC).
-
This subcommittee shall, at a minimum, address the following operational elements throughout the transfer process.
-
Communication of health care information.
-
Timely access to the appropriate LOC.
-
Continuity of care.
-
Access to necessary medications, Durable Medical Equipment (DME) and medical supplies, as ordered by the provider.
-
Health Information Management (HIM), including EHRS.
-
Identification of potential or actual risks to the patient as a result of the transfer process as well as risk mitigation strategies necessary to prevent potential or identified risks.
-
Performance improvement.
-
-
In addition, this subcommittee shall:
-
Review health care transfers to ensure that continuity of care is achieved for each patient.
-
Take corrective action to resolve or elevate concerns identified in the reviews.
-
Be responsible for reviewing, taking action, documenting, and forwarding best practices and recommendations for improvement to the IQMC.
-
-
-
-
-
Procedure
-
Patient Handoff Overview
-
Each transfer of care within and outside of CDCR shall be facilitated through a handoff process where information is provided by the designated sending care team member to the designated receiving care team member.
-
Transfers of care can occur between points of care (locations) (i.e., facility to facility), providers of care (i.e., Primary Care Registered Nurse [PCRN] to PCRN or Primary Care Provider [PCP] to PCP), or between levels of care (i.e., Clinic to Correctional Treatment Centers [CTC], or Mental Health Crisis Beds [MHCB] to Enhanced Outpatient Programs [EOP]).
-
Transfer between levels of care shall include verbal communication between the designated sending and receiving RN.
-
For transfers of care between points of care, the patient’s Primary Care Team (PCT) shall ensure that all outstanding primary care has been provided, ordered, and communicated to the receiving care team.
-
-
Patient handoffs include, but are not limited to:
-
A transfer from one area of the institution to another, resulting in a new care team (i.e., intra–facility transfers).
-
A transfer from one medication point of service (e.g., medication window) to another requiring the transfer of medications to the new point of service.
-
A permanent (i.e., inter-facility transfers) or temporary (i.e., Medical/Psychiatric and Return) transfer to another CDCR institution.
-
An urgent/emergent (i.e., through an Emergency Department [ED]) or planned (i.e., a medical appointment or planned treatment or admission) outside facility transfer.
-
Transfers to or from a different LOC within CDCR including, but not limited to:
-
Mental health levels of care
-
Correctional Clinical Case Management System (CCCMS)
-
MHCB
-
EOP
-
Acute Care Facility (ACF), including transfers to or from the Department of State Hospitals (DSH)
-
Intermediate Care Facility (ICF), including transfers to or from DSH
-
-
Medical levels of care
-
Ambulatory Care
-
Specialized Health Care Housing (e.g., Outpatient Housing Unit [OHU], CTC, Skilled Nursing Facility [SNF])
-
-
-
Transfers to or from CDCR Division of Adult Institutions control
-
Intake – Reception Center
-
Release from custody
-
Temporary transfer to community custody (e.g., out-to-court)
-
-
-
All patient handoffs shall:
-
Be tailored to the circumstances necessitating the transfer of care and the individual patient’s care needs.
-
Occur prior to the time of transfer, allowing for sufficient time for the sending and receiving care teams to ensure that all necessary supplies, equipment, medications, and other items necessary to provide care to the patient are available.
-
Include a verbal discussion and be documented in the patient’s health record to ensure the care needs are communicated to the receiving provider.
-
-
For transfers involving a change in institution, the Receiving and Release (R&R) Nurse shall:
-
Screen the health record prior to transfer for indications of potential or scheduled health care appointments.
-
Coordinate with the patient’s PCP, mental health clinician or dental provider to ensure that continuity of care is maintained before, during, and after the transfer.
-
-
-
Transfer of Information During the Patient Handoff
-
While each transfer of care may be unique, the transfer of information shall include, but is not limited to, as clinically indicated, the following:
-
Diagnosis
-
The patient’s primary diagnosis including the reason for the transfer of care.
-
Other significant diagnoses that may impact patient care during the transfer process.
-
Diagnoses listed on the patient’s current problem list.
-
Mental health LOC
-
Current suicide risk, self-harm risk, or precaution status
-
Behavioral problems and effective interventions
-
-
-
Current Physical Status
-
Vital signs
-
Objective data
-
Fall Risk, if applicable
-
-
Pertinent past medical history
-
Recommendations for care, if applicable
-
Pre-release information, if applicable
-
Current medications
-
Current treatments
-
Allergies
-
Significant flags, if applicable. Examples of flags include, but are not limited to:
-
Advance Directive for Health Care
-
Physician Orders for Life-Sustaining Treatment (POLST)
-
Coccidioidomycosis restrictions
-
Clozapine restrictions
-
Suicide watch and precautions
-
Public health concerns
-
Infection control needs
-
Medication alerts including Penal Code (PC) 2602 medications
-
PC 2604 information, if applicable
-
-
Limitations and accommodations
-
DME
-
Effective communication needs
-
-
-
Designated health care staff shall prepare a transfer packet for the patient depending on the location of the transfer (e.g., transfer to a location on the same yard may not require a transfer packet). Contents may vary based on the patient’s condition, the urgency of the transfer, and method of transportation. The transfer packet shall include information necessary to ensure continuity of care which may include, but is not limited to, the following, as applicable:
-
Transfer-Bus Content
-
Patient summary sheet
-
CDCR 7465, Physician Orders for Life-Sustaining Treatment (POLST)
-
CDCR 7421, Advance Directive for Health Care
-
First Responder Data Collection Tool
-
Emergency Care Flow Sheet
-
Emergent Transfer Report
-
Inpatient Discharge Summary
-
Medications (e.g., Nurse Administered [NA], Direct Observation Therapy [DOT], Keep-On-Person [KOP])
-
DME and Medical Supplies
-
-
-
Patient Transfer Process
-
Inter-facility Transfer (Institution to Institution) – Sending Institution
-
Custody staff shall notify health care staff via a bus list of a patient’s imminent transfer at least seven calendar days prior to the date of transfer.
-
The R&R Nurse shall:
-
Screen the health record for contraindications to transfer (e.g., inpatient, medical holds, potential medical holds, dental holds, specialty appointments).
-
Communicate with the patient’s care team to resolve issues and concerns. The PCP shall update the Medical Classification Chrono and initiate a medical hold if necessary, pursuant to the HCDOM Section, 1.2.14, Medical Classification System.
-
Communicate with the pharmacy to identify transfer medications and establish the supply that shall be sent with the patient (e.g., high cost, nonformulary). Refer to the HCDOM, Section 3.5.20, Medication Continuity with Patient Movement: Transfer/Parole/Discharge/Re-entry Program.
-
Communicate with the Supervising Dentist, or designee, to identify if conditions requiring a dental hold can be addressed at the proposed receiving institution
-
Notify the Classification and Parole Representative (C&PR) or designated custody representative if there is a contraindication to the patient’s transfer.
-
Complete the Inter-facility Transfer Screening.
-
-
The provider shall communicate verbally or electronically with the receiving institution’s PCP, R&R Nurse, or Triage and Treatment Area (TTA) Nurse regarding patients with special clinical requirements including, but not limited to, medications, treatments, or significant medical issues that may affect housing placement at the receiving institution.
-
Designated health care staff shall prepare the transfer packet for the patient as stated in Section (c)(2)(B).
-
Within 24 hours prior to the transfer, the R&R or TTA Nurse shall conduct a face-to-face interview and assess the patient for contraindications to transfer.
-
The evening prior to transfer, the medication nurse shall administer medication(s) to the patient per provider orders and deliver patient-specific NA/DOT medications to R&R for transfer with the patient.
-
On the day of transfer the R&R Nurse shall:
-
Provide required medications.
-
Verify receipt of the patient’s NA/DOT and KOP medication.
-
Verify the patient’s possession of DME.
-
Ensure items are placed in the white transfer packet with the transfer documents.
-
Provide rescue medications to the patient for holding during the transportation process.
-
-
The R&R Nurse shall complete the Pre-Boarding and ensure required handoff documentation is contained in the transfer packet.
-
The R&R Nurse shall verbally communicate to the receiving institution all information necessary to ensure the smooth transfer of care between institutions.
-
-
Inter-facility Transfer (Institution to Institution) – Receiving Institution
-
The R&R or TTA Nurse shall complete the Initial Health Screening before the patient is physically housed.
-
If the Initial Health Screening is completed by anyone other than an RN, and the patient answers “yes” to any questions, health care staff shall contact an RN for assessment and disposition of the patient. Health care staff, other than an RN, shall document the referral to the RN on the Initial Health Screening in the health record.
-
The RN shall document their assessment and disposition of the patient in the health record.
-
-
Patients shall be screened for Tuberculosis (TB) and Coccidioidomycosis according to current public health guidelines.
-
Pending specialty orders and other information shall be communicated to the UM RN, Specialty RN, and PCP via the Cerner Specialty message pool and the designated care team’s message pool.
-
The PCP shall complete order reconciliation by the close of the next business day.
-
The UM RN shall process Request for Service (RFS) orders that are in “pending”, “overdue”, or “in process” status in the UM Multi-Patient Task List (MPTL) per current guidelines.
-
The UM RN shall reconcile the completed non-formulary DME RFS.
-
If the UM RN or PCP identifies a high priority RFS is pending, they shall ensure a medical hold is placed, in accordance with the HCDOM, Section 3.1.11, Outpatient Specialty Services.
-
-
The receiving institution shall ensure the patient is scheduled for an initial new arrival assessment encounter as clinically indicated, as follows:
-
High Risk: PCP encounter within seven calendar days.
-
Medium or Low risk patients with one or more chronic conditions with prescribed medications: PCP or PCRN encounter within 30 calendar days or as ordered by the provider.
-
Medium or Low risk patients without known chronic conditions with prescribed medications shall be seen by a care team member as needed, or based on applicable care guides.
-
Mental health LOC patients: Initiation of a Mental Health PowerPlan previously ordered in a planned state. If a Mental Health PowerPlan has not been ordered in a planned state, the patient shall be referred to mental health via the institution’s LOP.
-
-
-
Non-CDCR Institution Transfers (Out-to-Court, Release from Custody)
-
Prior to the patient’s transfer, the R&R Nurse shall complete the steps in Section (c)(3)(A)5-7.
-
Release from custody
-
Custody staff shall notify health care staff of pending transfers via the Parole/Transportation List.
-
Patients in a community health care facility – Custody staff shall notify institution UM in these circumstances and if the patient requires placement in a community health care facility, institution UM shall notify Headquarters UM who shall assist with obtaining an appropriate community placement and the transfer of health records, as needed to ensure continuity of care.
-
Patients housed in a CDCR facility with ongoing health care needs (i.e., pending surgery, on TB treatments) – Custody staff shall notify institutional UM staff who shall:
-
Coordinate with HIM to obtain the required medical Release of Information.
-
Coordinate with community health care providers, DAPO, and PRCS to ensure continuity of care.
-
Coordinate with the PCT to ensure that the patient is provided educational materials and up to a 60-day supply of authorized medications pursuant to the HCDOM, Section 3.5.20, Medication Continuity with Patient Movement: Transfer/Parole/Discharge/Re-entry Program. Medication orders shall be compliant with current pharmacy policy and regulatory limitations.
-
Coordinate with the PCT to ensure that the patient is provided 30 days of prescribed medical supplies pursuant to the HCDOM, Section 3.6.1, Durable Medical Equipment and Medical Supply.
-
-
Patients housed in a CDCR facility who have ongoing acute mental health needs (e.g., housed in a MHCB or higher LOC facility, on Clozapine) – the patient’s mental health clinician shall coordinate with the appropriate CDCR, DAPO, and PRCS staff to ensure continuity of care upon release from CDCR custody (refer to the Statewide Mental Health Program Pre-Release Program Policy and Procedure).
-
-
-
Out-to-Court
-
C&PR staff shall notify health care staff in advance of scheduled court dates as outlined in the institution’s transfer LOP.
-
The R&R Nurse shall screen the health record for contraindications to transfer (e.g., medical holds, potential medical holds, dental holds, specialty appointments) and contact the PCT to resolve. If issues are identified, the following shall be completed, as applicable:
-
The provider or care team shall contact the institution’s C&PR for assistance in contacting the gaining jurisdiction’s PCP.
-
If the PCT determines a patient is too ill, unstable, or unable to participate in the court proceedings effectively, institutional clinical leadership (e.g., Chief Medical Executive, Chief Nurse Executive, CEO, Warden) shall contact the CCHCS Office of Legal Affairs to coordinate and determine options for the patient.
-
For mental health patients at the MHCB, ICF, or Acute Psychiatric Program, procedures established by the statewide mental health program shall be followed.
-
The provider or care team shall coordinate with the Pharmacist-in-Charge and the out-to-court provider for transfer of unusual medications, (e.g., Factor IX®, transplant medications) to ensure continuity of care.
-
-
Out-to-Court Returns – seven or more calendar days
-
The R&R Nurse shall:
-
Process patients who are out-to-court for seven or more calendar days as an inter-facility transfer upon return.
-
Complete an Initial Health Screening and registration.
-
Ensure High-Risk patients have a PCP encounter within seven calendar days.
-
Ensure Medium or Low Risk patients have a PCP or PCRN encounter within 30 calendar days or as ordered by the provider.
-
Initiate a Mental Health PowerPlan previously ordered in a planned state for mental health LOC patients. If a Mental Health PowerPlan has not been ordered in a planned state, the patient shall be referred to mental health via the institution’s LOP.
-
-
Pending specialty orders and other information shall be communicated to the UM RN, Specialty RN, and PCP via the Cerner Specialty message pool and the designated care team’s message pool.
-
The PCP shall complete order reconciliation by the close of the next business day.
-
The UM RN shall process RFS orders that are in “pending”, “overdue”, or “in process” status in the UM MPTL per current guidelines.
-
The UM RN shall reconcile the completed non-formulary DME RFS.
-
If the UM RN or PCP identifies a high priority RFS is pending, they shall ensure a medical hold is placed, in accordance with the HCDOM, Section 3.1.11, Outpatient Specialty Services.
-
-
-
Out-to-Court Returns – less than seven calendar days
-
The R&R Nurse shall:
-
Complete an Initial Health Screening.
-
If the patient’s encounter has been closed, notify the PCP, and other service providers as appropriate, to ensure completion of order entry/reconciliation by the close of the next business day.
-
-
-
-
Layovers
-
The R&R Nurse shall:
-
Complete a face-to-face observation of patients who were added to the bus list less than seven days prior, which shall include vital signs, before the patient leaves the layover institution.
-
Document the patient’s status and vital signs in the health record.
-
Register the patient and complete an Initial Health Screening.
-
Notify the PCT/MHPC to ensure that order reconciliation is completed by the close of the next business day.
-
-
Intra-facility Transfer (Yard-to-Yard)
-
Custody staff shall notify the sending care team’s nursing staff (medication point of service) via the Pending Bed Assignments Report or other approved notice.
-
The sending facility care team shall review the Patient Summary Sheet and Medication Administration Record (MAR).
-
The sending medication nurse shall:
-
Review the patient’s MAR for NA/DOT medications.
-
Note the number of KOP medications the patient shall have in their possession and communicate that number to the escorting custody staff.
-
Place the Patient Summary Sheet and NA/DOT medications in a labeled, sealed envelope and provide it to the escorting custody staff.
-
-
The sending care team shall communicate the following alerts or other significant health care information to the receiving care team and other necessary care providers (e.g., TTA RN, Specialty Clinic, Mental Health provider):
-
Unusual medications (e.g., Factor IX®, transplant medications), unusual treatments, and missing medications, as some medications may be located or administered in locations other than the patient’s usual medication administration location.
-
Pending appointments.
-
-
After the sending care team has reviewed the patient’s record, custody staff shall ensure that the following occurs:
-
The patient has all of their KOP medications in possession by verifying against the count provided by the care team.
-
The patient is in possession of required DME and medical supplies.
-
The care team has provided a sealed envelope containing the Patient’s Summary Sheet and NA/DOT medication, if applicable.
-
-
Upon the patient’s arrival, the receiving care team shall complete the following:
-
Review the Patient Summary Sheet.
-
Verify pending appointments are transferred to the new care team schedule.
-
Reconcile all medications with the patient’s MAR and obtain missing medications to prevent interruption in administration.
-
Verify that the patient is in possession of all required DME and medical supplies. Obtain any missing items to ensure care is continued without interruption.
-
-
-
LOC Changes – To or From Higher Levels of Care (HLOC), Non-Mental Health
-
Admission criteria are primarily based on nursing and therapy needs and not based on medical diagnosis alone. Hemodialysis, continuous chronic oxygen therapy, or diet alone may not qualify for a medical bed.
-
A patient handoff shall be completed for each LOC change.
-
The PCT or designated health care staff shall:
-
Screen the health record.
-
Notify custody staff that the patient is being transferred and provide the required method of transfer based on the patient’s clinical condition (e.g., State car, Americans with Disabilities Act van, bus, ambulance).
-
Contact the PCP, or designee, to obtain orders for medication, therapies, and diagnostics, as indicated, to ensure continuity of care.
-
Communicate pertinent health care information to the receiving health care facility.
-
-
Designated health care staff shall:
-
Prepare a transfer packet for the patient as stated in Section (c)(2)(B).
-
Provide rescue medications to the patient for use during the transfer as clinically indicated (i.e., KOP or with an escort).
-
-
Patients transferring to or from a HLOC including, but not limited to, CTC, SNF, Psychiatric Inpatient Program (PIP), MHCB, community hospital or other community-based licensed inpatient facility, or OHU who require ongoing care in a Specialized Medical Bed (SMB), shall go through the TTA.
-
If the patient requires immediate transfer to a HLOC and an appropriate SMB is not available, the patient shall be managed at the institution consistent with their clinical needs, which may include keeping the patient in the clinic or sending the patient to the TTA until SMB placement can be made.
-
If appropriate care cannot be provided in the clinic or TTA, the patient shall be sent to a community hospital until SMB placement can be made.
-
-
The UM RN, or other nursing designee, shall notify HCPOP of the required bed type.
-
The UM RN shall submit a HCPOP bed request packet for all patients requiring SMBs. The packet shall include current iterations of the information below:
-
LOC Assessment
-
History & Physical (H&P)
-
Clinical notes pertinent to the care given at the time of transfer
-
Medication record
-
Laboratory and diagnostic test results
-
-
A written HCPOP endorsement is required for any OHU placement and CTC, SNF, and hospice admissions.
-
The UM RN shall notify HCPOP immediately if a SMB becomes available.
-
-
Patients requiring a HLOC at their endorsed institution shall go directly to the HLOC, based on bed availability. If there is no bed available, the patient shall go through the TTA.
-
The PCP shall update the Medical Classification Chrono.
-
The TTA or R&R Nurse shall contact the PCP, or designee, to obtain orders for medication, therapies, and diagnostics, as indicated, to ensure continuity of care.
-
Patients discharged to an outpatient setting from a community hospital, ED, or any non-mental health CDCR health care bed shall be seen by their PCP within five calendar days of discharge.
-
Patients discharged from the TTA who are classified as high-risk or experienced a drug overdose that responded to naloxone or a skin or soft tissue infection shall be seen by their PCP within five calendar days of discharge. All other patient events shall be reviewed by the PCT in the following day’s huddle to determine appropriate follow up as indicated in the HCDOM, Section 3.1.3, Care Teams and Patient Panels.
-
-
LOC Changes – To or From HLOC, Mental Health
-
A patient handoff shall be completed for each LOC change.
-
The PCT, or designated health care staff, shall screen the health record; an H&P is not required prior to any transfer for mental health care and treatment.
-
LOC assessment will be conducted for all transfers to or from HLOC.
-
Patients on a medical hold shall remain at the institution due to medical necessity until the PCT can assess and collaborate with mental health to determine the patients’ most appropriate location and transfer. Patients not on a medical hold shall be considered medically cleared for transfer.
-
Upon return to the patient’s prior LOC (e.g., return to EOP from a MHCB), the mental health clinician shall coordinate daily follow-ups with licensed nursing staff and custody.
-
Mental health patients shall be seen by their clinician as specified in the Mental Health Services Delivery System Program Guide.
-
Mental health clinicians may order additional follow up care as part of the discharge planning process. This is particularly significant after extended stays at HLOC (e.g., ACF, ICF, PIP).
-
-
Patients transferring to or from a HLOC including, but not limited to, CTC, SNF, PIP, MHCB, community hospital or other community-based licensed inpatient facility, or OHU shall go through the TTA.
-
The TTA or R&R Nurse shall contact the PCP, or designee, to obtain orders for medication, therapies, and diagnostics, as indicated, to ensure continuity of care.
-
Patients discharged to an EOP LOC from a MHCB or PIP bed shall be seen by the mental health RN Care Manager within 3 calendar days of discharge and by a psychiatrist within 14 calendar days of discharge.
-
Patients discharged from a MHCB or PIP bed to a CCCMS LOC, and on psychiatric medications at present or in the last 6 months shall be seen by a psychiatrist within 14 calendar days of discharge.
-
If the MHCB or hospital psychiatrist asks that a patient be seen sooner than 14 calendar days after discharge, the psychiatrist’s order for when the patient should be seen shall be followed.
-
-
-
-
References
-
California Code of Regulations, Title 22, Division 7, Chapter 12, Article 3, Section 97520.13, Patient Transfer
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.14, Medical Classification System
-
Health Care Department Operations Manual, Chapter 2, Article 2, Section 2.2.1, General Use and Disclosure of Protected Health Information
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.1, Complete Care Model
-
Health Care Department Operations Manual, Chapter 3, Article 3, Dental Care
-
Health Care Department Operations Manual, Chapter 3, Article 5, Section 3.5.20, Medication Continuity with Patient Movement: Transfer/Parole/Discharge/Re-entry Program
-
Health Care Department Operations Manual, Chapter 3, Article 6, Section 3.6.1, Durable Medical Equipment and Medical Supply
-
Health Care Department Operations Manual, Chapter 3, Article 7, Section 3.7.1, Emergency Medical Response System (Pre-EMRP Go Live Institutions)
-
Health Care Department Operations Manual, Chapter 3, Article 7, Section 3.7.1-1, Emergency Medical Response System (EMRP Go Live Institutions)
-
Health Care Department Operations Manual, Chapter 3, Article 8, Section 3.8.6, Tuberculosis Program
-
California Department of Corrections and Rehabilitation, Department Operations Manual, Chapter 6, Article 12, Section 62080.15.2
-
California Department of Corrections and Rehabilitation, Mental Health Services Delivery System Program Guide
-
California Department of Corrections and Rehabilitation, Electronic Health Record System (EHRS) Approved Workflows
-
-
Revision History
-
Effective: 01/2010
-
Revised: 08/11/2025
-
3.1.10 Specialized Health Care Housing
-
Procedure Overview
-
California Department of Corrections and Rehabilitation (CDCR) shall ensure appropriate specialized health care housing is available to meet the level of care needed for each patient. These services shall include access to both community and institution-based specialized health care housing. CDCR shall ensure that the continuum of services is available statewide, with licensed services provided at a subset of institutions.
-
CDCR shall ensure the coordination of planned health care to patients needing services for preventive care to prevent illness and injury, interventions for acute illness and injury, supportive care for patients able to attend to their own activities of daily living, rehabilitation services, short and long-term nursing care, and palliative and end-of-life care under the direction of an interdisciplinary health care team. The scope of services provided to patients shall be interdisciplinary and include at a minimum: medical, mental health, dental, nursing, pharmacy, diagnostic, rehabilitative, and assistive services appropriate to maximize the quality of life and functional status and to reduce morbidity and mortality.
-
-
Responsibility
-
Statewide
-
CDCR and California Correctional Health Care Services (CCHCS) departmental leadership at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial, and clinical systems are in place, and appropriate tools, training, technical assistance, and levels of resources are available to ensure this procedure is successfully implemented and maintained.
-
The Undersecretary, Health Care Services, CDCR, and the Directors, Health Care Operations and Health Care Policy and Administration, CCHCS, shall designate a statewide committee with responsibility for the management of specialized health care housing facilities and beds statewide. Standing members of the committee shall include at a minimum, the Deputy Directors of Medical, Nursing, Mental Health, and Dental Services, Health Care Placement Oversight Program (HCPOP), Utilization Management (UM), Pharmacy, Ancillary and Allied Health Services, and Regional Health Care Executives (RHCEs). The committee shall be responsible for ensuring appropriate services are available for patients statewide, coordination of care, access to the appropriate level of care, and reducing the risks associated with handoff and transfer of patients between health care teams and specialized health care housing units.
-
Statewide health care leadership from all disciplines shall be responsible for developing and distributing tools to assist institutions in the development of Local Operating Procedures (LOPs) for their specialized health care housing units.
-
HCPOP in coordination with the Specialized Health Care Housing Standing Committee is responsible for the endorsement of patients to specialized health care housing units in the event that the institution does not have any appropriate, non-contract inpatient level of care beds available.
-
UM has the primary responsibility for establishing and maintaining a standardized, auditable system for managing health care resources within CDCR.
-
-
Regional
-
RHCEs are responsible for implementation of this procedure at the subset of institutions within an assigned region. RHCEs shall, at a minimum, monitor timeliness, access, and admission and discharge rates to ensure that patients’ level of care within the specialized health care housing unit is appropriate for their health care needs.
-
-
Institution
-
The Chief Executive Officer (CEO) has overall responsibility for implementation and ongoing oversight of a system to provide management of the scope of specialized health care housing within their institution. The CEO delegates decision making authority to designated health care executives for the daily operations of the specialized health care housing units and ensures adequate resources are deployed to support the system.
-
The CEO and all members of the institutional leadership team are responsible for ensuring:
-
All necessary resources are in place to support the successful implementation of this procedure at all levels including, but not limited to:
-
Institution level
-
Patient panel level
-
Patient level
-
-
Access to and utilization of equipment, supplies, health information systems, patient registries and summaries, and evidence-based guidelines.
-
Implementation of institution-specific LOPs that fully support and implement this procedure (refer to Section (e), Local Operating Procedure Requirements).
-
-
Institutional leadership shall review the operation of each element of the specialized health care housing unit ensuring that the patient is at the appropriate level of care; staff resources are available and distributed in order to provide the required patient care services; and that staff have access to the required resources, services, procedures, templates, equipment, supplies, and any other resources necessary to meet patient health care needs.
-
Each CDCR licensed facility (e.g., Correctional Treatment Center (CTC), Mental Health Crisis Bed (MHCB), Psychiatric Inpatient Program (PIP), Hospice) shall designate a Governing Body in accordance with the sections of the California Code of Regulations applicable to their licensure. An institution with multiple licensed facilities shall designate one Governing Body with responsibility for oversight of all licensed care within the institution. The Governing Body shall also communicate with the institution Quality Management Committee (QMC) at a periodic frequency to meet its oversight obligations but not less than quarterly.
-
Each institution shall designate a subcommittee in writing that has responsibility for the administration of the specialized health care housing provided at their institution. The designated subcommittee shall report to the Institution QMC. This subcommittee shall address at a minimum the following operational elements within the specialized health care housing units:
-
Emergency Management
-
Infection Control and Prevention
-
Human Resources
-
Environment of Care
-
National Patient Safety Goals
-
Health Information Management (Electronic Health Record System)
-
Leadership
-
Life Safety
-
Medication Management
-
Performance Improvement
-
Provision of Care, Treatment, and Services
-
Clinical Laboratory Improvement Amendments/Waived Testing
-
-
The designated subcommittee shall be responsible for taking corrective action to resolve and/or elevate concerns identified in the review. The review and action taken shall be documented and forwarded to the Institution QMC.
-
Each institution that provides a licensed service within their specialized health care housing units shall ensure that they comply with all applicable laws, rules, regulations, and policies regarding facility organization, standing committees, and policy and procedure development and implementation.
-
-
-
Specialized Health Care Housing Overview
-
Nursing Services
-
Nursing Care shall be available 24 hours per day, 7 days per week under the direction of a Supervising Registered Nurse (RN).
-
Nursing care services are designed to ensure an appropriate level of care is delivered to each patient through timely access to health care services, initial and ongoing assessment, planning, intervention and evaluation in a system designed to promote health maintenance, reduce risk of debilitative injury, improve function, and maximize the patient’s quality of life through application of evidence-based nursing practice.
-
Specialized health care housing units that do not have 24/7 RNs may have the CDCR 7362, Health Care Services Request Form, available for patients who request them and shall utilize the process described in the Health Care Department Operations Manual (HCDOM), Section 3.1.5, Scheduling and Access to Care. The institution LOP shall outline processes specific to each unit’s management of the CDCR 7362s.
-
For specialized health care housing units that do not utilize CDCR 7362s, daily nursing documentation shall include:
-
Patient’s requests for care
-
Patient’s concerns or complaints
-
Nursing interventions provided
-
-
Nursing care management services shall be provided to patients within the specialized health care housing units. The extent of care management services varies according to the complexity of the patient. Nursing care management services begin at the time of admission and continue through discharge. Nursing care management services shall be provided through a collaborative process of patient evaluation, advocacy, care planning, facilitation, and interdisciplinary coordination.
-
Nursing care management services shall be coordinated and continued across all levels of care (ambulatory, acute, and inpatient), in all physical locations where patients receive care within CDCR, and include the patient’s transition to community-based services upon parole and/or discharge.
-
-
Medical Services
-
Medical services shall be available 24 hours per day, 7 days per week under the direction of the Chief Medical Executive (CME).
-
Services shall be provided in person, telephonically, or through an approved telehealth solution.
-
The institution CME, or designee, shall ensure that a roster is available at all times to specialized health care housing unit staff designating the attending health care provider who is responsible for patient care.
-
-
The attending health care provider is responsible for initiating admission, determining the anticipated length of stay, identifying treatment goals and discharge planning for patients being admitted to, discharged from, or changing levels of care within the specialized health care housing units.
-
Medical services shall be provided to patients within the specialized health care housing units in accordance with the HCDOM, Chapter 3, Health Care Operations, and any other applicable laws, rules, regulations, and court orders.
-
-
Mental Health Services
-
Mental Health services shall be available 24 hours per day, 7 days per week under the direction of the Chief of Mental Health.
-
Services shall be provided in person, telephonically, or through an approved telehealth solution.
-
The Chief of Mental Health shall ensure that a roster is available at all times to specialized health care housing unit staff designating the mental health care clinician who is responsible for supporting the specialized health care housing units.
-
-
Mental Health services shall be provided to patients within the specialized health care housing units in accordance with the Mental Health Services Delivery System (MHSDS) Program Guide and associated policies, as well as any other applicable laws, rules, regulations, and court orders.
-
The designated mental health clinician, within his or her scope of practice, is responsible for initiating admission, determining the anticipated length of stay, identifying treatment goals and discharge planning for the mental health care of patients being admitted to, discharged from, or changing levels of care within the specialized health care housing units.
-
-
Dental Services
-
Under the direction of the Health Program Manager III (HPM III) and the Supervising Dentist (SD), Dental Services shall be available at least 8 hours per day, Monday through Friday, excluding holidays. Emergency Dental Services shall be available 24 hours per day, 7 days per week.
-
Services shall be provided in person or telephonically.
-
The HPM III and the SD shall ensure that a roster is available at all times to specialized health care housing unit staff designating a dental provider who is responsible for supporting the specialized health care housing units.
-
-
Dental services shall be provided to patients within the specialized health care housing units in accordance with the HCDOM, Chapter 3, Article 3, Dental Care and all other applicable laws, rules, and regulations.
-
The designated dental provider is responsible for coordinating patient admission with the attending health care provider as well as determining the anticipated length of stay; identifying treatment goals; and discharge planning for the dental care of patients being admitted to, discharged from, or changing levels of care within the specialized health care housing units.
-
-
Pharmacy Services
-
Pharmacy Services shall be available to ensure timely availability of medication 24 hours per day, 7 days per week under the direction of the institution Pharmacist-in-Charge and the Statewide Chief of Pharmacy Services.
-
The licensed Correctional Pharmacy or Central Fill Pharmacy shall ensure furnishing or dispensing of medications to specialized health care housing unit staff for the treatment of patients.
-
Drugs shall be available through the appropriate use of automated drug delivery systems and approved clinic stock in licensed correctional clinics.
-
Appropriate after-hours services shall be made available through the use of centralized remote pharmacist verification services.
-
Centralized remote after-hours pharmacy services shall ensure offsite pharmacist verification of new medication orders will be utilized when the drug is available in an automated drug delivery system or licensed correctional clinic stock.
-
The use of voluntary call back pharmacist services after hours may be utilized if medications are not available in any medication storage area outside the pharmacy area or during hours when the centralized after hours pharmacy services are unavailable.
-
-
Pharmacy services shall be provided to patients within the specialized health care housing units in accordance with the HCDOM, Chapter 3, Article 5, Pharmacy, and all other applicable laws, rules, regulations, and court orders.
-
-
Ancillary and Allied Health Services
-
Ancillary and Allied Health Services shall be available to patients within the specialized health care housing units under the administrative direction of the Chief Support Executive. Responsibility for the clinical supervision of ancillary and allied health care staff remains with the CME, or designee.
-
Ancillary and Allied Health Services include, but are not limited to:
-
Diagnostic Services (e.g., Laboratory, Radiology)
-
Nutritional (Dietary) Services
-
Medical Supply
-
Biomedical Maintenance
-
Durable Medical Equipment (DME)
-
Physical Therapy
-
Respiratory Therapy
-
Rehabilitation Services
-
Adaptive and Assistive Services
-
-
The institutional medical leadership shall ensure that specialized health care housing unit patients have access to the Ancillary and Allied Health Services necessary to comply with their interdisciplinary treatment plan.
-
Services shall be provided, as appropriate for the patient and as determined by the ordering health care provider, through the following methods:
-
In person
-
Telephonically
-
Through an approved telehealth solution
-
Through a contracted provider or facility
-
-
Services provided to patients within the specialized health care housing units shall be performed in accordance with the HCDOM and any other applicable laws, rules, regulations, and court orders.
-
-
-
Procedure
-
Admission
-
Patients shall be admitted to a specialized health care housing unit only upon the order of a health care provider granted privileges for the admitting facility.
-
The admitting health care provider shall be responsible for ensuring that all admission orders are written within 24 hours and documented in the health record.
-
Verbal admission orders shall not be given; however, in exigent circumstances, telephonic admission orders may be provided to the RN. The admitting provider shall counter-sign the telephonic orders and document in the health record.
-
The admitting health care provider is responsible for coordinating with other disciplines to ensure that health care actions specific to that discipline are completed (e.g., an admitting mental health clinician is responsible for coordinating the completion of the admitting history and physical [H&P] with the attending medical health care provider).
-
The admitting health care provider shall identify plans including, but not limited to, PCP rounding and follow-up, and anticipated length of stay if known.
-
-
Patients admitted to a specialized health care housing unit shall have an admitting H&P performed by a medical provider within 24 hours of admission.
-
Patients admitted to a specialized health care housing unit shall have an initial assessment at the time of admission performed by an RN.
-
Other members of the nursing Care Team may collect data and assist in the development of the patient’s interdisciplinary care plan consistent with their licensure and scope of practice.
-
The RN retains responsibility for the finalization and documentation of the admission assessment and the interdisciplinary care plan. The interdisciplinary care plan shall be completed within 72 hours of the patient’s admission and updated as the patient’s condition changes, treatments change and interventions change.
-
The RN shall provide and document patient education that includes, at a minimum:
-
Frequency of provider visits
-
Frequency of nursing rounds
-
How to use the call light
-
How to request care
-
-
Patients being admitted to a specialized health care housing unit for urgent mental health treatment shall be evaluated by a mental health clinician within the timeframes specified in the MHSDS Program Guide and associated policies and directives. This evaluation is in addition to any examination or H&P completed by the medical health care provider.
-
Patients being admitted to a specialized health care housing unit for dental treatment shall be examined by a dental provider within the timeframes specified in the HCDOM, Section 3.3.5.4, Dental Priority Classifications. This examination is in addition to any examination or H&P completed by the medical health care provider.
-
Each specialized health care housing patient shall be informed of his/her rights and responsibilities during the admission process as specified in the LOP.
-
-
-
Patient Stay
-
The Patient Stay process includes minimum expectations for rounding (type, frequency and composition), communication, and documentation requirements.
-
The frequency of rounds and the composition of the team conducting the rounds are determined by the patient’s condition and the patient’s care setting.
-
Rounds shall include, but are not limited to, nursing rounds, grand rounds, team rounds, safety checks, supervisory rounds, and environment of care rounds.
-
Patients admitted to a specialized health care housing unit shall be assessed by a member of his/her Care Team through rounds at least daily. The purpose of the assessment is to:
-
Determine the patient’s ongoing health care needs.
-
Determine the appropriate level of care.
-
Determine the response to treatment.
-
Analyze progress towards identified goals.
-
Identify unmet health care needs.
-
Adjust the interdisciplinary plan of care to meet the current and anticipated health care requirements.
-
-
Care Team members shall document their patient care interactions in the health record.
-
Each member of the Care Team is responsible for communicating with other Care Team members regarding any change in the patient’s condition or any abnormal findings.
-
Communication shall be in a manner appropriate for the abnormal finding or change in condition consistent with the Care Team member’s level of licensure and scope of practice.
-
Communication shall be documented in the health record.
-
-
-
Transfers
-
Patients shall only be transferred between specialized health care housing units upon the written order of a privileged health care provider.
-
The health care provider initiating the transfer shall be responsible for ensuring that all transfer orders are completed prior to the patient’s transfer.
-
Transfer orders shall be documented in the health record.
-
Verbal transfer orders shall not be given except in emergencies as defined in the HCDOM, Section 3.7.1, Emergency Medical Response System.
-
In non-emergent exigent circumstances, telephonic transfer orders may be provided to the RN. The provider initiating the transfer shall counter-sign the telephonic orders and document in the health record.
-
-
The health care provider initiating the transfer is responsible for coordinating the patient’s level of care change with the receiving specialized health care housing unit’s admitting health care provider.
-
Transfers shall be completed in accordance with the requirements of the HCDOM, Section 3.1.9, Health Care Transfer; MHSDS Program Guide; HCDOM, Chapter 3, Article 3, Dental Care; and/or applicable court orders.
-
-
Discharge
-
Discharge planning shall begin upon admission to the specialized health care housing unit.
-
The goal of the discharge planning process is to maximize the patient’s level of self-care, maximize and preserve functioning, improve quality of life, and to determine the appropriate level of housing post discharge.
-
The patient’s discharge plan shall be interdisciplinary.
-
Discharge planning shall include post-parole/post-CDCR release self-care needs when appropriate for the patient.
-
-
Patients shall only be discharged from a specialized health care housing unit upon the written order of a privileged health care provider.
-
The health care provider initiating the discharge shall be responsible for ensuring that all discharge orders are completed prior to the patient’s discharge.
-
Discharge orders shall be documented in the health record.
-
Verbal discharge orders shall not be given.
-
In non-emergent exigent circumstances, telephonic discharge orders may be provided to the RN. The provider initiating the discharge shall counter-sign the telephonic orders and document in the health record.
-
-
The health care provider initiating the discharge is responsible for coordinating the patient’s discharge with the receiving Primary Care Team (PCT). While verbal provider-to-provider communication is the preferred method of transfer handoff communication, the discharge summary shall include, at a minimum:
-
Reason for admission.
-
Current diagnoses in the active problem list.
-
Description of major events during the stay and treatments rendered.
-
Pertinent diagnostic studies.
-
Current medications.
-
Future appointments, diagnostic studies, and treatments.
-
Supplies, assistive devices, and DME including items that are to be in the possession of the patient upon discharge.
-
Assistance with one or more activities of daily living.
-
Disposition.
-
-
The Patient Summary Sheet may be utilized as a supplement to the primary discharge summary documents. However, the specialized health care housing unit staff are responsible for ensuring compliance with discharge documentation requirements.
-
-
Release from CDCR Custody
-
For patients being released from a specialized health care housing unit:
-
The institution UM nurse, Classification & Parole Representative, and clinical team shall coordinate with headquarters UM, Division of Adult Parole Operations (DAPO), Post Release Community Supervision and community based health care providers to identify appropriate placement for the patient.
-
The specialized health care housing unit clinical team, the institution UM RN, and the Receiving and Release (R&R) nurse shall ensure documentation about the patient’s health history, current status, medications, treatments and pending follow-up is obtained, along with required releases of information, and provided to the community based providers.
-
Whenever possible, conferencing with the receiving clinical team is recommended to ensure timely and seamless transition of care.
-
-
Coordination of Services
-
Each specialized health care housing unit shall ensure that their LOP includes instructions on the coordination of services between each level of care within the specialized health care housing units including:
-
The current institution and between its specialized health care housing units.
-
Other CDCR specialized health care housing units.
-
CDCR institutions.
-
Contract facilities.
-
DAPO managed facilities.
-
Other community resources as appropriate to the patient’s health care needs and CDCR status.
-
-
The patient’s PCT shall ensure the patient’s health care needs are coordinated between all disciplines involved in the patient’s current and future care. The Care Team may be assisted by UM and HCPOP:
-
The Institution’s UM representative serves as the primary resource for the patient’s PCT in determining the appropriate level of care, coordinating services between CDCR facilities, contract facilities, and community resources.
-
The specialized health care housing unit LOP shall refer to the current HCDOM, Section 1.2.15, Utilization Management Program, for guidance on the utilization of health care resources at all levels of care.
-
-
HCPOP serves as the specialized health care housing unit’s primary resource for endorsing patients to other levels of care when it is determined that the patient’s current location does not meet their health care needs.
-
HCPOP serves as the primary liaison between the CDCR Division of Adult Institutions, DAPO, and Division of Juvenile Justice, ensuring that all custodial requirements for patient movement have been met.
-
The specialized health care housing unit’s LOP shall refer to the current HCDOM, Section 5.1.3, Medical Bed Management, for guidance on the utilization of HCPOP services at all levels of care.
-
Whenever possible, the institution’s Primary Care Provider or UM nurse shall collaborate with HCPOP to initiate a bed hold for patients who are anticipated to be released from a community hospital within three days.
-
-
-
-
-
Local Operating Procedure Requirements
-
Each CDCR institution shall develop an LOP that outlines the policies and procedures for each level of care provided by each specialized health care housing unit within their institution to fully implement the requirements in this procedure.
-
LOPs for areas licensed and/or accredited by a local, county, federal, or state licensing agency and/or accrediting body shall comply with all laws, rules, regulations, and requirements pertaining to the license and/or accreditation held by the licensed/accredited service.
-
Admissions process.
-
Patient rounding regarding type, frequency, and composition.
-
Patient stay process.
-
Availability of CDCR 7362s.
-
Transfer process.
-
Discharge planning process.
-
Coordination of services process.
-
Patient release or parole process.
-
Transfer to county facilities or other outside facilities process.
-
Health record documentation frequency requirements for all patient care interactions.
-
Provisions for the implementation, structure, and operation of a quality assurance process.
-
Patient rights and responsibilities
-
-
References
-
California Code of Regulations, Title 22, Division 5, Chapter 12, Article 5, Correctional Treatment Centers
-
Health Care Department Operations Manual, Chapter 1, Health Care Governance and Administration
-
Health Care Department Operations Manual, Chapter 2, Article 2, Confidentiality and Privacy
-
Health Care Department Operations Manual, Chapter 2, Article 3, Section 2.3.1, Health Information Management Overview
-
Health Care Department Operations Manual, Chapter 3, Article 1, Complete Care Model
-
Health Care Department Operations Manual, Chapter 3, Article 2, Medication Management
-
Health Care Department Operations Manual, Chapter 3, Article 3, Dental Care
-
Health Care Department Operations Manual, Chapter 3, Article 5, Pharmacy
-
Health Care Department Operations Manual, Chapter 3, Article 6, Durable Medical Equipment/Supplies and Accommodations
-
Health Care Department Operations Manual, Chapter 3, Article 7, Emergency Medical Response
-
Health Care Department Operations Manual, Chapter 3, Article 8, Public Health
-
Health Care Department Operations Manual, Chapter 4, Special Circumstances
-
Health Care Department Operations Manual, Chapter 5, Article 1, Section 5.1.3, Medical Bed Management
-
California Department of Corrections and Rehabilitation, Mental Health Services Delivery System Program Guide, 2009 Revision, and associated policies and directives
-
-
Revision History
-
Effective: 04/2019
-
3.1.11 Outpatient Specialty Services
-
Procedure Overview
-
This procedure describes the structures, processes and resources that California Correctional Health Care Services (CCHCS) and California Department of Corrections and Rehabilitation (CDCR) staff shall utilize to ensure patients have timely access to safe and cost-effective specialty services that are medically necessary in order to establish diagnoses, make recommendations for diagnostic work-up, provide therapy, and establish treatment plans that include frequency of follow-up appointments with the specialist or the Primary Care Provider (PCP).
-
-
Responsibility
-
Statewide
-
CCHCS and CDCR departmental leadership at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial, and clinical systems are in place and appropriate tools, training, technical assistance, and levels of resources are available to ensure patients have timely access to safe and cost-effective specialty services that are medically necessary.
-
-
Regional
-
Regional Health Care Executives are responsible for implementation of this procedure at the subset of institutions within an assigned region.
-
-
Institutional
-
The Chief Executive Officer (CEO) has overall responsibility for implementation and ongoing oversight of the system at the institution. The CEO and all members of the institution leadership team are responsible for establishing an organizational culture that promotes interdisciplinary teamwork and continuous process improvement. The CEO delegates decision-making authority to the Chief Medical Executive (CME) and Chief Nurse Executive (CNE) for daily operations of specialty services to ensure that resources are deployed to support the system including, but not limited to, the following:
-
Ensuring access to equipment, supplies, health information systems, Patient Registries, Patient Summaries, and evidence-based guidelines.
-
Adequately preparing new Care Team members to assume team roles and responsibilities, including onboarding.
-
Providing Care Team members with adequate resources, staffing, physical plant, information technology, and equipment and supplies to accomplish daily tasks.
-
Requiring that Care Team members review pertinent patient information related to access to specialty services.
-
Requiring that each Care Team conduct Population Management Working Sessions, pursuant to the Health Care Department Operations Manual (HCDOM), Section 3.1.6, Population and Care Management Services, utilizing tools such as Dashboards, Patient Registries, and Patient Summaries to address concerns related to potential gaps in specialty services.
-
Providing ongoing training and assessing competence of Care Team members.
-
Reviewing and comparing institution Care Team performance including the overall quality of services, health outcomes, assignment of consistent and adequate resources; utilization of Dashboards, Patient Registries, Patient Summaries, and decision support tools; and addressing issues as necessary.
-
Updating procedures, roles and responsibilities, and training as new tools and technology become available.
-
Collaborating with the Warden to ensure that custody staff are available to provide timely, safe, and efficient escort and transportation of patients to specialty appointments.
-
Requiring institution leadership to establish a consistent and dedicated back-up system to ensure that specialty services’ scheduling is managed when staff are on leave or otherwise unable to meet daily demands to avoid specialty care delays.
-
-
The CME is responsible for the overall medical management of patients and ensures resources are available to meet the needs of the population.
-
The CNE is responsible for:
-
Ensuring that the institution has a designated Supervising Registered Nurse (SRN) to monitor specialty scheduling processes on a daily basis and identify and address or elevate barriers to access.
-
Managing and overseeing daily operations of the specialty scheduling system to include telemedicine and onsite and offsite scheduling processes.
-
Coordinating the delivery of health care services which includes familiarizing team members with the use of contracted services via access to and navigation of the provider directory.
-
Ensuring the most effective delivery of health care services to reduce cost and patient refusals by minimizing the need for long distance travel through effective use of the provider directory, local specialists, telemedicine services, and elevating scheduling and access to care issues to Direct Care Contracts’ Specialty Network Administration Program, and Headquarters (HQ) Utilization Management (UM) as necessary.
-
-
The Chief Physician and Surgeon (CP&S), SRN, and appropriate specialty services staff shall meet on an ongoing and weekly basis to ensure that patients with specialty referrals have timely access to these services.
-
The Institution Utilization Management Committee shall meet pursuant to the HCDOM, Section 1.2.15, Utilization Management Program, to review trends in specialty services including, but not limited to, timeliness of services and unexplained or significant outlier patterns of specialty services in order to reduce avoidable and unnecessary utilization and costs.
-
-
-
Procedure
-
General Requirements
-
An eConsult shall be initiated for appropriate specialties and conditions prior to generating a Request for Service (RFS).
-
Specialty services requests shall only be placed by PCPs or dentists who practice within CDCR. The ordering PCP shall complete the request in Cerner and shall indicate the timeframe in which the service is necessary (e.g., routine, medium, or high priority health care requests). Routine priority health care requests shall be the default priority for any health care request. Specialty service requests by a dentist shall follow Electronic Dental Record System Workflow #1-7.1 and Electronic Health Record System (EHRS) Workflow 100-71.
-
The PCP shall inform the patient of the plan for specialty referral including a general timeframe of expected service.
-
If a specialty service is scheduled outside of compliance timeframes, the Primary Care Team (PCT) shall evaluate and inform the patient that the requested service has been scheduled.
-
The information provided to the patient shall be documented in the health record.
-
The specific date, time, and location of the offsite appointment shall not be shared with the patient.
-
-
Patients with pending high priority specialty services shall be placed on a medical hold to prevent transfer and discontinuity of care pursuant to the HCDOM, Section 1.2.14, Medical Classification System.
-
If a patient is approved for a medium priority or routine priority specialty service and is subsequently transferred to another institution before the service occurs, the receiving institution shall continue with the original RFS and place an order maintaining the original compliance date unless the PCP at the receiving institution examines the patient and determines that it is no longer medically necessary or can be rescheduled to a later date. The PCP shall document their findings in the health record at the time the specialty service is cancelled or rescheduled.
-
The PCP or dentist shall continue to monitor the patient as clinically indicated, until the initial specialty service has occurred. The PCP or dentist shall document the patient encounters in the health record.
-
-
Pre-authorization Process
-
Emergent health care requests are exempt from the pre-authorization process.
-
The PCP shall submit the RFS order for electronic routing to the UM nurse. The UM nurse shall complete the first level review to determine if the RFS order meets evidence-based clinical decision support criteria.
-
Upon completion of the UM nurse review, the RFS order shall be electronically routed to the CME or CP&S for second level review.
-
At their discretion, the CME or CP&S may obtain input from other medical providers at the regularly scheduled provider meetings in order to determine medical necessity. The decision-making authority to approve or deny the RFS order at the second level remains with the CME or CP&S.
-
Requests for high or medium priority specialty services shall be processed in a manner that allows for both the first and second level of review to be completed within five calendar days from the date of the RFS order.
-
Requests for routine priority specialty services shall be processed in a manner that allows for both the first and second level of review to be completed within seven calendar days from the date of the RFS order.
-
-
The Statewide Medical Authorization Review Team (SMART) is the third level of review and shall review those services which are determined to require HQ approval within 30 calendar days of receipt of a routine priority health care RFS, 15 calendar days of receipt of a medium priority health care RFS, and seven calendar days of receipt of a high priority health care RFS.
-
Notwithstanding the above, requests for gender affirming surgery (GAS) or revisions to GAS shall be processed in their entirety pursuant to the HCDOM, Section 1.2.16, Gender Affirming Surgery Review Committee. All GAS requests require an HQ level of review.
-
-
If the RFS order is denied, the reason for the denial shall be documented in the health record, and the PCP shall be notified via the health record. The PCP shall review the decision, and, if determined appropriate, resubmit the RFS with additional information, documenting any such action in the health record. The PCP shall discuss the decision and if necessary provide the patient with alternate treatment strategies during the next encounter which shall be within 30 calendar days of the denial of the specialty service.
-
If the RFS order is approved, it shall be valid for 12 months even if the patient declines an appointment. Ongoing treatments such as for cancer, pacemaker or Automated Implantable Cardioverter Defibrillator interrogation, and hemodialysis require only an initial RFS and do not expire after 12 months.
-
The UM nurse or other designated specialty clinic staff shall determine if the services can be provided via telemedicine, onsite, or require an offsite appointment and schedule as appropriate. If a change in location is necessary at any point after the initial determination is made, a new RFS is not required unless the RFS has expired.
-
If after the patient has been scheduled for the specialty appointment and there is a need to schedule the patient with a different specialty provider, a new RFS is not required unless it has expired or the new specialty provider requests a new RFS.
-
If at any point the priority of the specialty services changes (e.g., from medium to high priority), a new RFS shall be submitted by the PCP.
-
Requests for specialty services from a dentist shall be approved by a Supervising Dentist (SD) or Regional Dental Director (RDD).
-
-
Patient Declined Appointments
-
The procedures set forth in HCDOM, Section 3.1.5, Scheduling and Access to Care, shall be followed for patients who decline to comply with specialty appointment ducats.
-
The declined appointment shall not be automatically rescheduled. Licensed health care staff shall:
-
Provide education to the patient using effective communication to ensure the patient is fully informed that they are declining a medically necessary service if the patient communicates their intention to decline.
-
Obtain a signed CDCR 7225, Refusal of Examination and/or Treatment, through an informed refusal process and subsequently cancel the order in the EHRS.
-
If the patient refuses to sign the CDCR 7225, two licensed health care staff shall sign. In restricted housing units and specialized health care housing, the CDCR 7225 may be signed by two staff members, one of whom shall be a licensed health care staff.
-
-
Document the reason for declining the appointment in the EHRS and that effective communication was reached.
-
Message the Specialty RN upon conclusion of the encounter, confirming a signed CDCR 7225 was completed and is in the EHRS, so the Specialty RN can proceed with cancellation.
-
The Specialty RN shall message the licensed health care staff to ensure the patient is ducated to the clinic in order to obtain an informed refusal pursuant to the HCDOM, Section 3.1.5(c)(3)(C)3 if they are unable to confirm there is a signed CDCR 7225 in the EHRS for the respective specialty services appointment.
-
The licensed health care staff shall message the Specialty RN upon conclusion of the encounter, confirming a signed CDCR 7225 has been completed and is in the EHRS so that the Specialty RN can proceed with cancellation.
-
-
-
If the patient changes their mind regarding a specialty appointment refusal and wishes to see the specialist, the following actions shall be taken:
-
The PCP shall determine if the specialty service is still clinically indicated and, if so, message the Specialty RN.
-
If the initial RFS has expired, the Specialty RN shall request that the PCP place a new RFS order.
-
For initial specialty consults, the Specialty RN shall place a “referral to” order.
-
For follow-up specialty services, the PCP shall place a follow-up specialty order.
-
If the specialty service is no longer clinically indicated, the PCP shall discuss the treatment plan with the patient and document in the EHRS.
-
-
If the patient communicates their intention to be rescheduled for the specialty appointment via the CDCR 7362, Health Care Services Request Form, process or during a nursing encounter, the licensed health care staff shall message the PCP to determine if a primary care appointment is needed prior to proceeding with a new specialty service appointment.
-
If the PCP determines a primary care appointment is necessary, the patient shall be scheduled as appropriate.
-
If the PCP determines that the specialty service is still clinically indicated, they shall message the Specialty RN.
-
If the initial RFS has expired, the Specialty RN shall request that the PCP place a new RFS order.
-
For initial specialty consults, the Specialty RN shall place a “referral to” order.
-
For follow-up specialty services, the PCP shall place a follow-up order.
-
-
-
-
Specialty Appointments Occurring Outside the Institution
-
The designated health care staff shall complete the clinical portion of the CDC 7252, Request for Authorization of Temporary Removal for Medical Treatment, for health care services that are provided offsite.
-
The designated health care staff shall include relevant information for transportation staff regarding infectious precautions and disabilities requiring accommodation as well as any medical transportation needs in the “Remarks” section of the CDC 7252.
-
The designated health care staff shall sign the completed CDC 7252 and forward it to the designated custody staff. Custody staff shall prepare the “Custodial status” of the CDC 7252 and shall ensure all necessary signatures are obtained.
-
Custody staff shall contact the institutional transportation team that provides transportation for the patient to the scheduled appointment.
-
The CME or CP&S shall prioritize the scheduled appointments when transportation needs exceed custody availability. Appointments shall be rescheduled and should not exceed the initial timeframe based on clinical needs.
-
The designated health care staff shall place a copy of the RFS order and any other pertinent clinical information in an envelope and provide it to custody staff for delivery to the specialty provider. Pertinent clinical information includes, but is not limited to:
-
For initial encounters:
-
CDC 7243, Health Care Services Physician Request for Services.
-
Health care provider’s progress notes.
-
Relevant laboratory studies, imaging studies, and diagnostic results.
-
Current medication profile and allergies.
-
Any additional pertinent information.
-
Effective communication accommodations shall be provided in accordance with the Armstrong Remedial Plan and related Court Orders.
-
-
For follow-up encounters:
-
Any subsequent consults, test results, diagnostic results, workups, and physician’s orders or recommendations requested as a result of previous encounter(s).
-
The status of the patient’s effective communication accommodations information shall be reviewed, and effective communication shall be provided in accordance with the Armstrong Remedial Plan and related court orders.
-
-
-
Custody staff shall obtain the clinical documentation including, but not limited to, the specialty consultation report, prescriptions, clinical notes, discharge summaries, and brief operative notes, from the specialty provider and return the clinical documentation to the Triage and Treatment Area (TTA) upon return of the patient to the institution.
-
All patients who receive specialty services outside the institution shall be processed in the TTA (Standby Emergency Medical Services at California Health Care Facility) upon return to the institution.
-
The TTA RN shall assess the patient, review the findings and recommendations made by the specialist, and document their findings in the health record.
-
The TTA RN shall notify the PCP or on-call provider of any immediate medication or follow-up requirements. If the specialty appointment included treatment of fractures to the maxilla, mandible, dental related infection, or resulted in maxillo-mandibular fixation, the TTA RN shall also notify the SD or dentist on-call. If the specialty appointment included pathology specimen collection, the procedures listed in the HCDOM, Section 3.1.14, Laboratory Services, Appendix 3, Offsite Pathology Orders, shall be followed.
-
The TTA RN shall enter and implement all telephone orders given by the PCP or on-call provider including but not limited to, housing, Durable Medical Equipment (DME), treatments, and scheduling. For a follow-up appointment with the PCT, the provider shall remain on the line until the order has been read back and verified.
-
The TTA RN shall submit the clinical documentation to Health Information Management (HIM) staff for scanning into the health record.
-
-
If a patient returns without the clinical documentation, the TTA RN shall call the specialty provider to obtain a copy of the clinical documentation.
-
The telephone contact shall be documented by the TTA RN in the health record.
-
If the specialty provider is unavailable, the TTA RN shall contact the PCP or on-call provider for direction.
-
If unable to obtain the clinical documentation, the TTA RN shall inform HIM staff to obtain it.
-
-
Clinical documentation is required to be submitted by the specialty provider within 48 hours of the encounter with the exception of studies that require longer than 48 hours to complete. Exceptions include, but are not limited to, cultures, biopsies, pathology, cytology, cardiac lab studies, sleep lab studies, pulmonary studies, neurologic studies, and other specialized labs.
-
-
Specialty Clinic Appointments Occurring Within the Institution
-
If trained and provisioned access, the onsite specialty provider shall document their recommendations and findings in the health record or provide written documentation to the designated nursing staff on the day of the encounter.
-
If the onsite specialty provider is not trained and provisioned access to the health record, pertinent clinical information shall be provided including, but not limited to:
-
For initial encounters:
-
CDC 7243.
-
Health care provider’s progress notes.
-
Relevant laboratory studies, imaging studies, and diagnostic results.
-
Current medication profile and allergies.
-
Any additional pertinent information.
-
Effective communication accommodations shall be provided in accordance with the Armstrong Remedial Plan and related Court Orders.
-
-
For follow-up encounters:
-
Any subsequent consults, test results, diagnostic results, workups, and physician’s orders or recommendations requested as a result of previous encounter(s).
-
The status of the patient’s effective communication accommodations information shall be reviewed, and effective communication shall be provided in accordance with the Armstrong Remedial Plan and related court orders.
-
-
-
The designated nursing staff shall:
-
Review the findings and recommendations made by the specialty provider.
-
Notify the PCP or on-call provider of any immediate medication or follow-up requirements.
-
Implement all telephone orders given by the PCP or on-call provider including, but not limited to, housing, DME, treatments, and scheduling. For a follow-up appointment with the PCT, the provider shall remain on the line until the order has been read back and verified.
-
Forward all written documentation to HIM staff for scanning into the EHRS.
-
Forward documentation directly entered into the EHRS to the PCP for review.
-
-
-
Contracted Specialty Clinic Appointments Occurring via Telemedicine
-
Use of Clinical Presenters
-
Consistent with HCDOM, Section 3.4.1, Telemedicine Specialty Services and Primary Care, the Clinical Presenter or Telemedicine Coordinator presents the patient from the originating site to the hub site telemedicine services provider and is responsible for clinical support at the institution’s site during the telemedicine encounter.
-
The presenter shall be an RN trained to support the telemedicine clinic, who is available at the originating site to present the patient, manage the telemedicine peripheral examination instruments and assist in performing any hands-on exams to complete the encounter successfully.
-
-
Clinical Presenter Chart Review Prior to Clinic (Pre-Clinic)
-
It is the responsibility of the originating institution’s Telemedicine Coordinator to review the health record prior to the telemedicine encounter to ensure that all required testing and diagnostics have been conducted and that the results and reports have been uploaded to the web-based medical documents transfer system.
-
The clinical information required for the telemedicine encounter shall be uploaded to the Health Insurance Portability and Accountability Act (HIPAA)-compliant, web-based medical documents transfer system a minimum of three business days prior to the encounter. Any applicable, additional clinical information obtained between the date sent and the encounter shall be sent to the hub site provider or designee immediately. Patients without the required work-up may not be seen until the necessary pre-work-up has been completed.
-
For initial encounters, medical information shall be obtained per the telemedicine encounter checklist including, but not limited to:
-
CDC 7243.
-
Health care provider’s progress notes.
-
Relevant laboratory studies, imaging studies, and diagnostic results.
-
Current medication profile and allergies.
-
Any additional pertinent information.
-
Effective communication accommodations shall be provided in accordance with the Armstrong Remedial Plan and related Court Orders.
-
-
For follow-up encounters, medical information shall be obtained per the telemedicine encounter checklist, including:
-
Any subsequent consults, test results, diagnostic results, workups, and physician’s orders or recommendations requested as a result of previous encounter(s).
-
The status of the patient’s effective communication accommodations information shall be reviewed, and effective communication shall be provided in accordance with the Armstrong Remedial Plan and related court orders.
-
-
-
-
Use of the Health Record
-
The originating institution shall have the health record available at the time of the patient’s telemedicine encounter. The Clinical Presenter at the originating institution shall review the health record prior to the encounter and when necessary, or at the hub provider’s request, shall provide additional information from the health record.
-
-
Clinic Service Follow-up (Post-Clinic)
-
After all telemedicine encounters the Clinical Presenter shall complete the CDCR-approved effective communication documentation and shall document in the progress notes the hub provider’s name, specialty, date of the encounter, and note that the session was conducted via telemedicine.
-
The contracted, non-CCHCS hub provider shall dictate and sign a final consultation and recommendation and submit the documentation to the institution’s Telemedicine Coordinator at the originating site via the HIPAA-compliant, web-based medical documents transfer system within three business days from the encounter. These are considered to be the original records and are routed per the institution’s process for placement into the health record.
-
-
-
Follow-up with the Primary Care Team after Specialty Services
-
The PCP or dentist shall endorse the specialty consultation report within five calendar days of receipt and document in the health record that the review has been completed and whether the PCP or dentist agrees or disagrees with the specialist’s recommended actions. If there is disagreement with the recommended actions, the PCP or dentist shall document the reason for disagreement. If the PCP or dentist agrees with the specialist’s recommendation, the appropriate order(s) shall be placed.
-
Following a high priority specialty services appointment, the patient is required to be seen by the PCP or dentist within five calendar days.
-
Following a medium or routine priority specialty services appointment, the PCP shall review the clinical documentation and determine whether a primary care follow-up appointment is needed. If a PCP follow-up appointment is not needed, the PCP shall send a letter to the patient within five calendar days of receipt of the specialty report notifying them of relevant diagnostic study results, recommended specialty treatments that will be ordered, and the timeframe in which these treatments will be ordered.
-
After an initial appointment with a specialist, subsequent appointments with that specialist or recurrent treatments do not require a follow-up appointment with the PCP nor written patient notification unless there are significant changes in the treatment plan as determined by the PCP or there are other reasons the PCP determines follow-up or notification is necessary.
-
At the follow-up appointment, the PCP or dentist shall discuss the specialty provider’s findings and recommendations with the patient, as clinically appropriate, and document the discussion in the health record.
-
Ongoing treatments such as dialysis, chemotherapy, radiation therapy, pacemaker interrogations, and related follow-ups require only an initial approval to initiate the series of treatments and consultations.
-
If the specialty provider recommends a new procedure, surgery, or specialist consultation, and the PCP or dentist agrees with the specialty provider’s recommendations, a new RFS shall be submitted.
-
Follow-up with the specialty provider after a procedure or surgery does not require another RFS order if completed within the global surgery schedule timeframes.
-
All other specialty follow-up services occurring 12 months after the date of the original RFS order require a new RFS order.
-
-
Specialty providers may not directly order follow-up consultations, diagnostic studies or treatments. The specialty provider shall make recommendations and the PCP or dentist shall review these recommendations to determine the need based on clinical guidelines, if applicable, and medical necessity.
-
If there are questions regarding medical necessity, the PCP shall discuss the case with the CME or designee including possible referral to the SMART.
-
If it is determined that the follow-up consultations, diagnostic studies or treatments recommended by the specialty provider do not meet clinical guidelines and are not medically necessary, the PCP shall document the reason in the health record.
-
If applicable, an eConsult shall be utilized.
-
Recommendations regarding dental treatments shall be approved by the SD or RDD.
-
-
-
Statewide Medical Authorization Review Team
-
The SMART is the third level of review and shall review cases appealed by the PCP or that meet criteria for a higher level of review to determine if the specialty service is medically necessary.
-
Membership
-
The SMART Chairperson shall be designated by the Deputy Director, Medical Services.
-
The SMART membership shall consist of Regional Deputy Medical Executives, at least two other headquarters-based physician managers, and two physician managers from the field.
-
-
Meetings
-
The SMART shall meet as often as is necessary to conduct its business within established timeframes, but not less frequently than monthly.
-
A quorum is met when a minimum of 50 percent of the members are in attendance. A quorum must be present to take action on any agenda item.
-
-
Committee Proceedings Documentation
-
Records of committee proceedings shall be kept at a secure, accessible medical program site for a period of three years. At minimum, the record shall describe all committee actions and recommendations.
-
The proceedings and records of the SMART shall be confidential and protected from discovery to the extent permitted by law.
-
Patients shall be provided a letter stating the outcome of the SMART review.
-
-
-
-
-
References
-
California Civil Code, Division 1, Part 2.6, Section 56, et seq.
-
California Evidence Code, Division 9, Chapter 3, Section 1157
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.14, Medication Classification System
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.15, Utilization Management Program
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.16, Gender Affirming Surgery Review Committee
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.6, Population and Care Management Services
-
California Department of Corrections and Rehabilitation, Department Operations Manual, Section 62070.9.3
-
Centers for Medicare and Medicaid Services Global Surgery Booklet
-
-
Revision History
-
Effective: 04/2019
Revised: 02/18/2026
-
3.1.13 Medical Imaging Services
-
Policy
-
California Correctional Health Care Services (CCHCS) staff shall ensure patients have timely access to safe and cost-effective medical imaging services that are medically necessary to establish diagnoses, make recommendations for additional diagnostic work-up, and establish treatment plans. The Medical Imaging Services (MIS) program shall perform, process and interpret results of medical imaging examinations both within the institutional MIS departments and through contracted onsite services. The MIS program shall maintain accurate records of both onsite and offsite medical imaging in a retrievable manner for a minimum of seven years, adhering to all applicable retention, privacy and security, and safety guidelines as required by federal and state laws.
-
-
Responsibility
-
Statewide
-
California Department of Corrections and Rehabilitation and CCHCS departmental leadership at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial, and clinical systems are in place and appropriate tools, training, technical assistance, and resources are available to ensure patients have timely access to safe and cost-effective medical imaging services that are medically necessary.
-
The Statewide Chief, MIS is responsible for the implementation and maintenance of a safe and effective MIS program to include:
-
Develop statewide standard operating procedures for use at the local level.
-
Monitor annual institution Radiology Supervisor & Operator (RS&O) and quarterly mammography inspections, medical imaging equipment registration and calibration, and radiation safety procedures.
-
Provide oversight and initiating statewide MIS contracts, procurements, policies and procedures, workflows, and forms. Monitor the execution of statewide contracts and procurement agreements.
-
Provide oversight of all medical imaging examination preparations and protocols and quality assurance of all examinations performed onsite, in conjunction with the CCHCS contracted radiology group.
-
Provide consultation and advice to health care providers and institution staff regarding their institution MIS departments.
-
-
-
Regional
-
Regional Health Care Executives are responsible for implementation of this procedure at the subset of institutions within an assigned region.
-
The Regional Health Care Executive is responsible for the oversight of the assigned regional Senior Radiologic Technologist (SRT) staff.
-
The Statewide MIS Chief shall provide functional direction to the SRT staff.
-
-
The SRT shall:
-
Perform administrative duties at the regional level.
-
Monitor and assist MIS operations at multiple institutions within their assigned region.
-
Perform institution-based medical imaging duties as needed and when appropriate.
-
Monitor institution-based MIS staff performance within their assigned region through regular onsite compliance auditing, training, competency verification, and any subsequent remediation.
-
Coordinate with the Statewide MIS Chief, or designee, and the appropriate hiring authority, or their designee, on all competency verification related processes, findings, and remediation.
-
-
-
Institutional
-
The Chief Executive Officer (CEO), or designee, is responsible for implementation of this policy at the institution level.
-
The Chief Support Executive (CSE) or Correctional Health Services Administrator (CHSA) shall:
-
Hire institution MIS staff members, ensuring appropriate training is provided and completed, and monitoring of staff performance. Institution medical leadership may consult with MIS headquarters regarding staff member duties and quality of staff performance.
-
Determine institution MIS departments’ operating hours based on institutional needs.
-
Monitor performance of contractual and clinical onsite mobile service providers in collaboration with MIS headquarters.
-
Monitor performance of onsite medical imaging providers in collaboration with MIS headquarters.
-
-
Institutional radiology departments shall:
-
Ensure all patient medical imaging orders are entered into the Electronic Health Record System (EHRS) including orders for examinations to be performed onsite, verifying the orders in the CCHCS Radiology Information System (RIS), and ensuring all examination images are received by the CCHCS Picture Archiving and Communication System (PACS).
-
Perform all onsite X-Ray examinations.
-
Perform all onsite mammography ordered for screening and diagnostic purposes at women’s institutions, where available.
-
Ensure accuracy in all health records as they pertain to MIS.
-
Ensure appropriate maintenance of radiology department equipment.
-
Monitor mobile imaging technologist timeliness and adherence to State requirements.
-
Ensure mobile imaging technologists complete all steps in the examination process.
-
Report institution and mobile medical imaging equipment issues immediately to the CHSA, CEO and MIS headquarters.
-
Ensure the radiology department is current on X-Ray, mammography registration, inspections, and maintaining records of all required inspections, licenses, and permits.
-
Monitor the onsite schedule, obtaining results and examination images in collaboration with the Imaging Records Center.
-
-
The CCHCS contracted radiology group shall:
-
Interpret all onsite and mobile examination images loaded into PACS and contacting health care providers by telephone as clinically indicated.
-
Ensure availability to interpret images and to be contacted by the institutions Monday through Friday, between the hours of 7:00 a.m. and 7:00 p.m.
-
Perform annual RS&O and quarterly mammography inspections with a report of their findings to the inspected institution and Chief, MIS.
-
-
-
-
Procedure
-
General Ordering Procedures
-
When a health care provider determines medical imaging is necessary, the health care provider shall submit an order for imaging in the EHRS and include the priority timeframe to complete the service.
-
For STAT X-Ray orders, the patient is immediately sent to the radiology department for the ordered service. For all other STAT imaging orders, the patient is immediately sent to an outside hospital.
-
All initial X-Rays are ordered as high priority and are provided as ordered or within 14 calendar days from the date of the order if a timeframe is not specified.
-
Medium priority medical imaging services shall be provided as ordered or within 15-45 calendar days from the date of the order if a timeframe is not specified.
-
Routine priority medical imaging services shall be provided as ordered or within 46-90 calendar days from the date of the order if a timeframe is not specified.
-
-
The ordering health care provider shall inform the patient of the plan including a general timeframe of expected service.
-
If a service is scheduled or rescheduled outside of compliance timeframes, the primary care team shall evaluate and inform the patient.
-
If the patient’s condition has declined and cannot wait for the scheduled appointment appropriate action shall be taken.
-
The information provided to the patient shall be documented in the EHRS.
-
The specific date, time, and location of an offsite appointment shall not be shared with the patient.
-
-
Orders for X-Ray examinations, abdominal ultrasounds, fibroscans, and mammograms do not require pre-authorization and will be automatically routed in the EHRS to the imaging scheduler.
-
Orders, other than X-Ray examinations, abdominal ultrasounds, fibroscans, and mammograms, will generate an electronic Request for Service (RFS) that requires Utilization Management (UM) pre-authorization as outlined in Section (c)(2) of this procedure.
-
Approved orders will be automatically routed in the EHRS to the imaging scheduler. The imaging scheduler shall determine if the examination will be performed onsite or offsite.
-
Onsite studies include Computerized Tomography, Magnetic Resonance Imaging, Ultrasound, mammography, and general X-Ray. Refer to the onsite scheduling process outlined in Section (c)(3) of this procedure.
-
Offsite studies include, but are not limited to, nuclear medicine, biopsy, and fluoroscopy examinations. Refer to the offsite scheduling process outlined in Section (c)(4) of this procedure.
-
-
-
Pre-Authorization Process for Imaging Studies Ordered by CCHCS Providers
-
STAT orders are exempt from the pre-authorization process.
-
Orders, other than for plain film X-Ray examinations, abdominal ultrasounds, fibroscans, and mammography, are electronically routed to the UM nurse, or designee, for the first level review to determine if the RFS meets evidence-based clinical decision support criteria.
-
Upon completion of the first level review, the RFS will be electronically routed to the Chief Medical Executive (CME), Chief Physician and Surgeon (CP&S), or Supervising Dentist (SD) for second level review.
-
At their discretion, the CME, CP&S, or SD may obtain input from other health care providers at the regularly scheduled provider meetings to determine medical necessity. The decision-making authority to approve or deny the RFS at the second level remains with the CME, CP&S, or SD.
-
High and medium priority services shall be processed in a manner that allows for both the first and second level of review to be completed within five calendar days from the date of the order.
-
Routine priority services shall be processed in a manner that allows for both the first and second level of review to be completed within seven calendar days from the date of the order.
-
-
The Statewide Medical Authorization Review Team (SMART) is the third level of review and shall review cases appealed by the provider within 14 calendar days of the date of the order.
-
If the RFS meets clinical criteria and is approved, it will be automatically routed in the EHRS to the individual scheduling the appointment.
-
If the RFS is denied, the provider shall document the decision and provide the patient with alternate treatment strategies during the next encounter, which shall be within 30 calendar days of the denial of the medical imaging study.
-
Orders designated (DENTAL) shall only be utilized by dental providers and require prior approval by the SD or Dental Authorization Review Committee. These orders are not routed to the CME, CP&S, or Statewide Medical Authorization Review Team.
-
-
Imaging Studies Completed Onsite or via Onsite Mobile Services
-
The designated staff shall schedule the appointment in the EHRS which interfaces with RIS and the Strategic Oversight Management System.
-
For STAT X-Ray orders, the patient is immediately sent to the radiology department for the ordered service, which shall be performed upon arrival of the patient in the radiology department.
-
Orders for all other medical imaging services shall be performed within the priority timeframes specified in Section (c)(1) of this procedure.
-
When a radiology scheduled procedure conflicts with another appointment, the individual scheduling the appointment shall communicate with the appropriate department prior to overriding the previously scheduled appointment.
-
-
The Radiologic Technologist (RT) shall check the order against imaging protocols to verify the proper examination was ordered. If a change is required, the technologist may change the order with a co-signature required from the ordering health care provider.
-
The RT shall communicate the necessary examination preparation to designated nursing staff per the institutional Local Operating Procedure (LOP). The designated nursing staff shall perform necessary patient examination preparation.
-
At the appointment, the RT shall:
-
Use at least two patient identifiers (e.g., patient name or ID card, California Department of Rehabilitation and Corrections number, date of birth) to positively identify the correct patient.
-
Arrive the patient in RIS.
-
Perform the requested service using established protocols.
-
Perform quality check of acquired images.
-
Submit the images to PACS for interpretation. Images are stored in the PACS.
-
End the appointment with clinically appropriate instructions to the patient.
-
-
The Radiologist shall read, interpret, and document results. The signed report is automatically transmitted to the EHRS and RIS.
-
All critical results shall be immediately communicated to the ordering health care provider via telephone. If the results are received after hours or the ordering health care provider is unavailable, the report shall be communicated to the Triage and Treatment Area (TTA) staff via telephone for appropriate notification to the on-call provider. (See Appendix 1, Communication Urgency Level for Radiologic Findings.)
-
STAT examination reports shall be read and finalized within two hours from the time the examination is available for interpretation.
-
All other examination reports shall be read and finalized within four hours from the time the examination is available for interpretation.
-
Addendum requests shall be completed within three calendar days of the request being placed.
-
-
-
Imaging Studies Ordered Onsite and Completed Offsite
-
The designated staff responsible for scheduling shall:
-
Contact the offsite facility to schedule the appointment.
-
Request that upon completion of the exam, the offsite facility:
-
Immediately communicate all critical results to the TTA for appropriate action. (See Appendix 1, Communication Urgency Level for Radiologic Findings.)
-
Submit the interpretive report to the requesting institution within two business days of approval by the Radiologist.
-
Submit the images to the CCHCS Health Information Management (HIM) department within three business days.
-
-
Schedule the order in the EHRS.
-
Submit any related examinations or information to the offsite facility.
-
-
When a scheduled radiology procedure conflicts with another appointment, the individual scheduling the appointment shall communicate with the appropriate department prior to overriding the previously scheduled appointment.
-
The RT shall communicate the necessary examination preparation to Nursing (or designated) staff per the institution LOP. Nursing (or designated) staff shall perform and ensure patient preparation is completed including the communication of any related instructions the patient needs to receive.
-
The designated specialty clinic staff shall ensure all necessary arrangements are made for patient transportation to the offsite facility pursuant to the Health Care Department Operations Manual, Section 3.1.11, Outpatient Specialty Services.
-
Staff shall follow offsite specialty services workflow within the EHRS for patients undergoing offsite specialty radiology examinations.
-
-
The offsite report is received by the HIM department at the institution which shall be transmitted to the ordering provider for review and endorsement in the EHRS.
-
The HIM department moves the offsite report (PDF file) to the designated local “not completed” radiology folder. Institution radiology staff shall:
-
Upload or scan the report into the RIS.
-
Assign (create non-medical addendum for off-site) a RIS task for the Imaging Records Center.
-
Complete the exam.
-
-
The Imaging Records Center shall:
-
Request the images from the outside facility if they have not been received by HIM.
-
Upload the images into PACS and finalize examination.
-
-
-
Imaging Studies Completed While Patient is Offsite at a Hospital
-
The corresponding offsite imaging report will be received by the HIM department at the institution within three days of discharge and made available to the provider and the radiology department.
-
The designated radiology staff shall:
-
Place an order and schedule it in the EHRS with the actual date of service,
-
Upload or scan the report into RIS,
-
Assign a task (create non-medical addendum for off-site) in RIS, and
-
Complete the exam for the Imaging Records Center.
-
-
The Imaging Records Center shall:
-
Request the images from the outside facility if they have not been received by HIM.
-
Upload the images into PACS and finalize examination.
-
-
-
Provider Review of Imaging Studies Results and Patient Notification and Follow-up
-
Following the finalization of all imaging studies as described in Sections (c)(3) through (5) above, the health care provider shall:
-
Review and endorse the report within five calendar days of receiving an examination report notification into the EHRS.
-
Create a patient notification letter in the EHRS at the time of the provider’s review of the examination results. The patient notification letters shall include the following:
-
Date of the examination results.
-
Name of the health care provider who reviewed and endorsed the medical imaging result.
-
The clinical significance or meaning of the medical imaging results such as, but not limited to, whether the results are unchanged, or within normal limits, or as expected, or whether additional testing is required.
-
Whether a follow-up appointment with the provider is required and that it will be scheduled.
-
-
-
Patient notification letters shall be printed for collection by the designated staff member to be distributed to the patients.
-
Patients may request to view their detailed medical imaging records free of charge at their institution’s HIM office.
-
The Primary Care Team (PCT) shall schedule the patient for a follow-up appointment as clinically indicated. At the follow-up appointment, the designated PCP shall discuss the findings and recommendations with the patient and document the discussion in the EHRS.
-
-
Imaging Study Cancel and Place a New Order
-
An imaging study may be canceled for reasons including, but not limited to, patient refusal, the study is no longer clinically indicated, conflicting appointments, or the incorrect study was ordered.
-
The designated health care staff shall cancel and place a new order in the EHRS with a co-sign, note the specific reason for cancellation, and notify the ordering health care provider via telephone and a message using the EHRS message center regarding the cancelled order.
-
The ordering health care provider shall sign-off on order changes or cancellation in the EHRS.
-
-
Quality Assurance and Quality Control
-
Testing Equipment and Supplies
-
On a quarterly basis, satisfactory operation of all X-Ray equipment shall be checked by examining the following:
-
Equipment condition
-
Each of the items listed in the quality control checklist below should be inspected by a RT on a quarterly basis or after service or maintenance on the X-Ray unit. Items not passing the visual check should be replaced or corrected as soon as possible.
-
Mechanical Integrity
-
Check for loose or absent screws, bolts, or other loose elements.
-
Functioning of meters, dial, and other indicators.
-
Collimator light brightness and cleanliness.
-
Operation lights on control panel are sufficiently lit (illuminated) to function in a darkened examination room.
-
Collimator beam limiting devices functioning correctly and verification of proper alignment to bucky devices.
-
-
Mechanical Stability
-
Locks and detents operable.
-
Over-head X-Ray tube boom smoothness of motion.
-
Table bucky devices and wall unit cassette holders are stable and move smoothly.
-
If the X-Ray table has angulation functions, check the smoothness and accuracy of the angulations cable.
-
Condition of cables termination rings are fastened, no insulation breaks, and they hang properly as to not interfere with the operations of the unit.
-
-
-
Inspection of the condition of computed radiology (CR) or digital radiology (DR) cassettes and imaging devices.
-
All lead or lead equivalent aprons and gloves, to include those in Dental, shall be checked annually by performing a radiologic image review to ensure no cracks or damage. All inspections shall be logged in the Annual Lead or Lead Equivalent Apron log in the Quality Control book kept in the radiology department.
-
CCHCS shall contract with a medical physicist to perform annual testing of all required radiographic equipment as needed and mammography equipment, if applicable.
-
-
Equipment compliance with state regulatory provisions as required under Title 17 of the California Code of Regulations (CCR) 30305 and 30307. Technique charts shall be made available for each room to maintain consistent exposure factors and image quality.
-
-
-
Repeat Analysis
-
The criteria associated with repeating an exposure is subjective. Institutional radiology departments should strive for a repeat rate no greater than five to seven percent monthly and submitted to MIS.
-
Determine the total number of repeated exposures and the total number of exposures. The overall repeat rate is the total of repeated exposures divided by the total number of exposures during the tested period.
-
Repeat analysis should be performed quarterly and requires an ongoing tracking of number of exposures.
-
The repeat analysis report shall include each RT, modality, and exam procedure performed.
-
-
The percentage of repeat exposures shall provide the institutional radiology departments with information that focuses attention on the proper corrective action needed to reduce that percentage.
-
-
Artifact Evaluation and Prevention
-
All DR or CR cassettes and imaging devices shall be identified with a number placed on the back of each cassette for inventory and quality assurance tracking.
-
Identify and correct artifacts that may obscure clinical findings on radiographs.
-
DR or CR cassettes and imaging devices shall be charged (if applicable) as needed and cleaned according to manufacturer specifications.
-
Documentation of when cassettes, imaging plates, and imaging devices are cleaned. This must be maintained to ensure compliance with Title 17 of the CCR 30305 and 30307.
-
-
-
-
-
Appendices
-
Appendix 1: Communication Urgency Level for Radiologic Findings
-
-
References
-
California Code of Regulations, Title 17, Division 1, Chapter 5, Subchapter 4, Group 3, Article 4, Section 30305 and Section 30307
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.11, Outpatient Specialty Services
-
California Department of Corrections and Rehabilitation, Department Operations Manual, Chapter 9, Article 6, Sections 91060.1-91060.17, Radiology Services
-
-
Revision History
-
Effective: 06/2012
Revised: 09/17/2025
-
-
Appendix 1: Communication Urgency Level for Radiologic Findings
Anatomical Region Category 1: Communicate Immediately (Call) Category 2: Communicate Within Hours
(Sign within 4 hours)Category 3: Communicate Within Days
(Sign within 4 hours)General ∙ Malpositioned line or tube of immediate clinical concern (e.g., ET tube or enteric tube in bronchus)
∙ Foreign body with potential immediate and/or severe consequences
∙ Any finding that the interpreting radiologist determines requires immediate physician notification∙ Clinically significant mass, tumor or infection
∙ Finding highly suggestive of malignancy
∙ Intravascular line in suboptimal location, moderate risk (e.g., Intended central line in jugular or azygous vein, right atrium)
∙ Retained surgical instruments, sponges, devices
∙ Misplaced or migrated surgical or other implanted devices (e.g., IVC filter, gastric band, pacemaker wires)
∙ Adverse event from diagnostic imaging or interventional procedure
∙ Significant congenital anomaly∙ Probable malignancy, any location, no acute danger to patient
∙ Significant nonmalignant diagnosis, any location, no acute danger to patient
∙ Incidental finding on imaging study requiring further workup or longer-term follow-upNeurologic/ Head and neck ∙ Intracranial or spinal hemorrhage (parenchymal, subarachnoid, subdural epidural)
∙ Intracranial mass with significant mass effect (midline shift/herniation/hydrocephalus)
∙ Brain herniation
∙ Symptomatic hydrocephalus (malfunctioning shunt or new diagnosis of any cause)
∙ Depressed skull fracture
∙ Posttraumatic pneumocephalus
∙ Arterial dissection
∙ Severe spinal cord compression of any cause
∙ Unstable spine fracture
∙ Cord hemorrhage or infarct
∙ Airway obstruction or impending obstruction (epiglottis, retropharyngeal abscess, tonsillitis, facial fracture, other)∙ Critical arterial stenosis or occlusion
∙ Non-ruptured intracranial aneurysm
∙ Intracranial mass without significant mass effect (no midline shift/herniation)
∙ Non-hemorrhagic stroke, not thrombolytic candidate
∙ Linear skull fracture
∙ Facial fracture, no airway compromise, likely to need surgical repair
∙ Stable spinal fracture without cord compression
∙ Spinal mass without cord compression
∙ Spinal cord edema
∙ Discitis
∙ Airway narrowing, not severely obstructive
∙ Abscess, any location
∙ Encephalitis∙ Small intracranial mass, likely benign, no mass effect
∙ Hemodynamically significant arterial stenosis (carotid or vertebral), not associated with acute symptoms or otherwise immediately threatening
∙ Suspected brain metastases, established cancer diagnosisGI ∙ Unexplained pneumoperitoneum
∙ Closed loop intestinal obstruction
∙ Intestinal ischemia and/or portal/mesenteric venous gas
∙ Pseudoaneurysm or active hemorrhage (post trauma, GI bleed, other)
∙ High grade intra-abdominal organ injury (liver, spleen, pancreas, other) and/or bowel injury post trauma, acute intervention likely∙ Abscess, any location
∙ Intestinal obstruction, no evidence of acute ischemia
∙ Intra-abdominal infection, likely surgical or interventional candidate (Appendicitis, cholecystitis, diverticulitis, abscess, other)
∙ Large volume ascites
∙ Low to moderate grade intraabdominal organ injury and/or bladder or bowel injury post trauma, observation likely
∙ Pneumatosis in bowel wall, no other signs of ischemia∙ Low volume ascites (any cause), portal hypertension, and/or cirrhosis GU/OB ∙ Testicular torsion
∙ Ovarian torsion
∙ Ectopic pregnancy (high likelihood)
∙ Placental abruption
∙ Uterine rupture
∙ High grade kidney injury and/or ureteral or bladder injury post trauma, acute intervention likely
∙ Absent perfusion postoperative kidney
∙ Oligohydramnios (less than fifth percentile for age)∙ Placenta previa or suspected placenta accreta, increta, percreta in third trimester
∙ Embryonic/fetal demise
∙ Incompetent cervix in pregnancy
∙ Abdominal umbilical cord Doppler or IUGR
∙ Urinary tract obstruction (stone, tumor, other)
∙ Pyonephrosis/renal abscess
∙ Abnormal appearing pregnancy for which short interval follow-up is recommended
∙ Indeterminate findings for ectopic versus normal pregnancy∙ Placenta previa or possible previa in second trimester
∙ Suspected placenta accrete, increta, percreta in second trimester
∙ Abnormal findings on routine obstetrical ultrasound (possible fetal abnormality, abnormal growth, abnormal fluid volume, other) not likely to need acute interventionBreast ∙ Biopsy recommended ∙ Follow-up imaging recommended MSK ∙Non-spinal fracture and/or dislocation with risk of vascular compromise
∙Necrotizing fasciitis∙ Bone lesion at risk for pathologic fracture (femur, other)
∙ Non-spinal fracture and/or dislocation without vascular compromise, likely to need intervention
∙ Large hematoma without or with fracture, especially with compression of adjacent structures
∙ Fracture follow-up imaging, significant change in alignment or concern of infection
∙ Infection (including septic arthritis and osteomyelitis)
∙ SCFE
∙ Hardware complicationChest ∙Tension pneumothorax
∙Pulmonary embolus (CT or high probability V/Q scan), hemodynamically unstable, central embolus, and/or extensive emboli
∙Lung lesion with high possibility of being active TB
∙Large pericardia effusion and/or suspected tamponade or any cause
∙Active posttraumatic hemorrhage
∙Tracheal obstruction or impeding obstruction∙ Superior vena cava occlusion (including SVC syndrome) ∙ Pneumothorax, no evidence of tension
∙ Lobar or lung collapse
∙ Pneumomediastinum, interstitial emphysema, extensive subcutaneous emphysema
∙ Pulmonary embolus, hemodynamically stable, limited extent peripheral emboli
∙ Moderate or large pleural effusion
∙ Significant superior vena cava compression or narrowing
∙ PneumoniaCardiac/
Vascular∙Ruptured/leaking arterial aneurysm (thoracic or abdominal aortic or other)
∙Limb-threatening arterial or venous occlusion or high-grade stenosis
∙Arterial dissection or intramural hematoma (aortic, other)
∙Acute myocardial infarction∙ Hemodynamically significant arterial stenosis or occlusion, associated with acute symptoms
∙ Occluded coronary or other bypass graft with associated symptoms
∙ Deep venous thrombosis
∙ Arterial pseudoaneurysm post vascular access
∙ Thoracic aortic aneurysm ³ 6 cm or Abdominal aortic aneurysm ³ 5 cm, no evidence of acute instability
∙ Previously unknown chronic arterial dissection or intramural hematoma∙ Nondisplaced minor fracture or questioned fracture low risk for worsening
∙ Routine fracture follow-up imaging, healing not progressing as expected or minor change in alignment
3.1.14 Laboratory Services
-
Procedure Overview
-
California Correctional Health Care Services (CCHCS) staff shall utilize available processes and resources to ensure patients are provided timely access to laboratory services that are clinically necessary.
-
-
Responsibility
-
Statewide
-
CCHCS and California Department of Corrections and Rehabilitation (CDCR) departmental leadership at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial, and clinical systems are in place and appropriate tools, training, technical assistance, and levels of resources are available to ensure patients have timely access to laboratory services that are clinically necessary.
-
A statewide contract shall be maintained with an outside agency (contract laboratory), or multiple agencies, to provide routine laboratory analysis or testing, including a limited 24-hour STAT laboratory testing menu.
-
The Statewide Chief of Laboratory Services is responsible for the implementation and maintenance of a safe and effective Laboratory Services program. Specifically, they are responsible for:
-
Developing standard operating procedures for institution adoption into Local Operating Procedures (LOPs).
-
Monitoring and assisting institutions with their maintenance of Clinical Laboratory Improvement Amendments (CLIA) certificate of waivers and Clinical and Public Health Laboratory Licenses, and compliance activities and regulatory agency inspections related to Laboratory Services.
-
Initiating and overseeing statewide Laboratory Services contracts, procurements, policies and procedures, workflows, and forms. This includes coordinating Laboratory Services-related Electronic Health Record System (EHRS) issues with Department technical staff to prioritize corrective measures and maintenance activities.
-
Overseeing and providing quality assurance of the Laboratory Services onsite delivery of services in conjunction with the CCHCS contracted outside agency, or agencies.
-
Providing consultation and advice to health care providers and institution staff regarding their institution Laboratory Services departments and the Department’s Laboratory Services test order menu.
-
Monitoring the performance of the contracted outside agency, or agencies, in collaboration with the medical leadership Laboratory Services staff at the institutions and headquarters.
-
Overseeing and coordinating the competency assessment and related remediation efforts of local, regional and headquarters Laboratory Services staff.
-
-
-
Regional
-
Regional Health Care Executives are responsible for implementation of this procedure at the subset of institutions within an assigned region.
-
The Regional Health Care Executive is responsible for the oversight of the assigned regional Senior Clinical Laboratory Technologist (SrCLT) staff.
-
The Statewide Chief of Laboratory Services shall provide functional direction to the SrCLT staff.
-
-
The SrCLT shall:
-
Perform administrative duties at the regional level.
-
Monitor and assist Laboratory Services operations at multiple institutions within their assigned region.
-
Perform institution-based laboratory duties as needed and when appropriate.
-
Monitor institution-based Laboratory Services staff performance within their assigned region through regular onsite compliance auditing, training, competency verification, and any subsequent remediation.
-
Coordinate with the statewide Chief of Laboratory Services, or designee, and the appropriate hiring authority, or their designee, on all competency verification related processes, findings, and remediation.
-
-
-
Institutional
-
The Chief Executive Officer (CEO), or designee, is responsible for implementation of this policy at the institution level.
-
The CSE, or designee, is responsible for ensuring the institution has designated specimen collection and processing locations (laboratories) and a clinic administrator responsible for properly functioning lab processing equipment, properly maintained and an adequate amount of lab-related supplies, an adequate number of trained staff, appropriate patient scheduling processes are in place for laboratory services, and established lab processing work flows and lab LOPs are adhered to.
-
A standardized LOP template shall be developed by the statewide Chief of Laboratory Services.
-
At a minimum, an institution shall adopt a standardized LOP template customized for their site containing the following elements https://cdcr.sharepoint.com/sites/cchcs_lifeline_labs:
-
General Lab Policy and Staff Roles and Responsibilities.
-
Laboratory Orders.
-
Specimen Collection and Processing.
-
Paternity Testing.
-
-
LOP adoption requires the signature of an institution’s Laboratory Director as designated on the CLIA Certificate of Waiver, and the Statewide Chief of Laboratory Services.
-
The LOPs shall be reviewed, updated if required, and resigned at a minimum of every two calendar years or more frequently as required.
-
Laboratory Services and non-Laboratory Services staff shall adhere to LOP requirements.
-
-
Health care providers are responsible for ordering medically necessary laboratory studies consistent with community standards and department policy and care guidelines.
-
-
-
Procedure
-
Routine Laboratory Orders Processing
-
Routine Laboratory Orders typically include the following collection priorities with the indicated collection time defaults within the EHRS:
-
AM Draw: Next morning.
-
ASAP: Next day.
-
Routine: 30 calendar days.
-
Timed Study: Collection defaults to ordering provider’s selected collection date.
-
-
Health care providers shall place orders for all laboratory services in the EHRS.
-
Scheduling and ducating procedures shall be followed for the specimen collection of the ordered lab test(s). The ducating and scheduling process may be adjusted to facilitate timely specimen collections on or before the requested collection date.
-
The laboratory specimen shall be obtained from the patient on or before the requested collection date. The specimen shall not be collected prior to the requested collection date if prohibited by policy or the ordering provider.
-
If there are questions regarding an order, staff shall call or message the ordering health care provider for clarification.
-
Laboratory orders shall be collected by Laboratory Services staff during laboratory’s normal business hours.
-
Staff may collect a routine lab test the next business day should the requested collection date fall on a weekend or holiday.
-
-
Staff are prohibited from canceling or discontinuing Laboratory Services orders unless staff are authorized by the ordering provider or as specified under Section (c)(5)(C) of this policy to perform the cancellation or discontinuation, and there is EHRS documentation of the reason for the cancellation or discontinuation. The ordering provider, or appropriate clinical staff, shall be consulted prior to canceling or discontinuing an order suspected of being a clinically unnecessary duplicate order, or associated with recurring or multiple future collection appointments such as most Timed Study laboratory orders.
-
Staff shall record the specimen collection date and time and person collecting the specimen in the EHRS and update the status of the specimen to “collected.”
-
Laboratory specimens shall be labeled in the presence of the patient and not labeled prior to collecting the specimen.
-
-
For specimens not collected by Laboratory Services staff, e.g., an inpatient collection, the designated staff shall arrange for pick-up or delivery, and final processing at the institution laboratory during laboratory’s normal business hours.
-
The specimen shall remain in a secure storage area with appropriate environmental conditions to maintain the specimen in an adequate state until retrieved by staff or dropped off at the laboratory.
-
Un-retrieved specimens, or specimens collected outside of Laboratory Services normal business hours, shall have a designated secure storage area with appropriate environmental conditions to maintain the specimen in an adequate state.
-
-
Storage of laboratory specimens awaiting collection by couriers shall be secured in a locked container. Institution health care leadership or a designated health care team member shall coordinate with custody staff to determine the type(s) of locked boxes to be used and their appropriate placement for couriers to readily access and retrieve specimens.
-
The specimen shall be picked up by the contracted outside agency each business day at the contractually agreed upon time.
-
All non-critical results shall be reported to the institution within 24 hours of completion of the contracted laboratory’s analysis or testing in accordance with the Laboratory Services contract or test schedule.
-
-
STAT Laboratory Orders Processing
-
Health care providers shall place orders for all STAT laboratory services in the EHRS. The order shall be collected as soon as possible on the same calendar day and the results from the reference lab shall be available within contractual timeframes.
-
Refer to the Health Care Department Operations Manual (HCDOM), Section 3.1.5, Scheduling and Access to Care, under Section (c)(3)(C)(4), if a laboratory appointment originated from the order of a psychiatrist.
-
STAT laboratory order processing requirements and timelines only apply to lab tests designated on the Approved CDCR-CCHCS STAT Testing Menu. Providers shall only place STAT orders from the approved Testing Menu.
-
Providers seeking STAT results for lab tests not designated on the Approved CDCR-CCHCS STAT Testing Menu or results faster than their institution’s expected STAT processing time have the option of referring for offsite laboratory services. Providers shall follow the LOPs and policies applicable to offsite care at their facility.
-
-
If there are questions regarding an order, staff shall call or message the ordering health care provider for clarification.
-
For STAT orders, the patient shall be sent to the appropriate location for collection of the ordered STAT lab test(s).
-
STAT laboratory specimens shall be collected by staff as soon as possible on the same calendar day.
-
Staff shall place the STAT pick-up notification call to the contracted laboratory immediately following the order’s change to an “in-transit” order status.
-
Upon notification of a STAT order for laboratory services, the specimen shall be picked up by the contracted laboratory within two hours for non-rural institutions and three hours for rural institutions, 24 hours per day, seven days per week.
-
Processing of the patient’s specimen shall begin immediately upon arrival at the contracted laboratory.
-
STAT results shall be provided by the contracted laboratory via telephone to the Triage and Treatment Area (TTA), or designated health care team member, within four hours of the telephone request for pick-up for non-rural institutions and five hours for rural institutions.
-
If the STAT results are received after hours or the ordering provider is unavailable, the TTA staff shall notify the on-call provider within 30 minutes and document the notification in the EHRS.
-
-
Specimens Requiring Special Handling or Processing
-
Staff collecting specimens that require special handling shall follow the specimen requirements provided by the outside agency contracted to provide laboratory analysis and testing.
-
CCHCS laboratory tests requiring special handling or processing include the following:
-
Clinical Urine Drug Screening for Substance Use Disorder treatment (refer to Appendix 1).
-
Reception Center Clinical Urine Drug Screening (refer to Appendix 2).
-
Offsite Pathology Orders (refer to Appendix 3).
-
-
-
Review and Notification of Laboratory Test Results
-
Laboratory test results shall be electronically interfaced from the contracted laboratory’s system to the EHRS.
-
Test results that cannot be electronically interfaced shall be faxed or printed and provided to the designated location within the institution and scanned into the EHRS by Health Information Management.
-
A notification will appear in the ordering health care provider’s Message Center informing them of the return of the results.
-
Results or reports that are routed incorrectly to a provider’s Message Center may be refused by the reviewing provider within the EHRS and shall be forwarded to the appropriate health care provider.
-
If the result of a laboratory test is “Test Not Performed,” the designated staff may submit a replicate order on behalf of the health care provider unless otherwise directed by the health care provider.
-
If there are questions regarding an order, the ordering health care provider shall be contacted for clarification.
-
-
The health care provider shall electronically review and endorse each laboratory result within five calendar days of the date of receipt.
-
The health care provider shall create a patient notification letter in the EHRS at the time of review and endorsement of each laboratory result. The patient letters shall include the following:
-
Date of the laboratory test/screening.
-
Name of the health care provider who reviewed and endorsed the lab result.
-
The clinical significance or meaning of the lab results such as, but not limited to, whether the results are unchanged, or within normal limits, or as expected, or whether additional testing is required.
-
Whether a follow-up appointment with a provider is required, and if so, that it will be scheduled.
-
-
Patient notification letters shall be printed for collection by the designated staff member to be distributed to the patients.
-
Patients may request to view their detailed laboratory test results by submitting a completed CDCR 7385, Authorization for Release of Protected Health Information, through Nursing Services. There is no fee for the patient to request and view their results.
-
Any critical laboratory values shall be immediately reported by the contracted laboratory via telephone to the TTA or designated health care team member and requires a read back to be communicated for clarification.
-
If critical laboratory values are received after hours or the ordering provider is unavailable, the TTA staff shall notify the on-call provider within 30 minutes and document the notification in the EHRS.
-
-
Patient Refusals or Failures to Report for Laboratory Services
-
If the patient does not arrive for a laboratory appointment, the designated staff member scheduled to collect the specimen shall notify custody staff of the patient’s failure to report to the clinical appointment.
-
If the patient arrives in the laboratory services area and permits specimen collection, the collection shall be performed.
-
The staff responsible for the specimen collection shall cancel the laboratory order if the patient refuses specimen collection at the laboratory services area. If the patient fails to report (“no-show”) not due to a stated refusal, the staff responsible for the specimen collection shall attempt to reschedule the patient one-time for a second specimen collection attempt. The laboratory order shall be canceled by staff if the patient refuses or fails to report for the second scheduled appointment.
-
The staff responsible for specimen collection shall document the reason for the order cancellation in the EHRS. The EHRS will automatically notify the ordering provider of the cancellation.
-
The staff responsible for the specimen collection shall not be responsible for leaving the designated laboratory services area to document a patient refusal or failure to report.
-
Staff shall require authorization from the ordering provider for the cancellation of laboratory orders not associated with a patient refusal, or second specimen collection attempt associated with a failure to report for laboratory service.
-
The cancellation and rescheduling process(es) outlined in Appendix 1 of the HCDOM, Section 3.1.14 for the special handling circumstances and cancellation process for Clinical Urine Drug Screening supersedes this section.
-
The documentation (CDCR 7225, Refusal of Examination and/or Treatment) and counseling procedure for the failure to report for a medical appointment outlined in the HCDOM, Section 3.1.5, Scheduling and Access to Care, does not apply in cases of patient refusals or failures to report related to laboratory orders.
-
-
The ordering provider and the patient’s care team shall inform the patient of the health care consequences, and the determination of the appropriate next clinical steps because of a patient refusal or failure to report for Laboratory Services. Appropriate next clinical steps could include, but are not limited to, ordering clinically necessary laboratory services.
-
-
-
Appendices
-
Appendix 1, Clinical Urine Drug Screen for Substance Use Disorder Treatment
-
Appendix 2, Reception Center Urine Drug Screening
-
Appendix 3, Offsite Pathology Orders
-
-
References
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.5, Scheduling and Access to Care
-
-
Revision History
-
Effective: 01/2006
Revised: 11/12/2025
-
-
Appendix 1: Clinical Urine Drug Screening
-
Urine Drug Screening (UDS) is a urine toxicology study used for therapeutic monitoring purposes, but may be ordered when a patient presents with an altered level of consciousness.
-
Clinical UDS results, a patient’s decision to decline a Clinical UDS, and/or a patient’s inability to provide a sample due to paruresis or other reason shall only be available to clinical staff on a need to know basis in order to manage a patient’s health care needs, and these results shall not be shared with custody.
-
CLINICAL UDS ORDERING
-
Health care providers shall place orders for the initial UTOX Screen lab test with an “ASAP” collection priority in the Electronic Health Record System (EHRS).
-
The requested collection date for the UTOX Screen lab test will default within the EHRS to one calendar day from the date of the order.
-
Staff may collect the initial UTOX Screen lab test on the next business day should the requested collection date fall on a weekend or holiday.
-
-
Health care providers shall place orders for follow-up UDS testing (UTOX Monitoring or UTOX Panel tests) as a “Timed Study” collection priority in the EHRS.
-
The UTOX Monitoring and UTOX Panel lab tests may be ordered with requested collection dates within one week of one another and should not be considered duplicate orders.
-
Health care providers should alert Laboratory Services staff of follow-up UDS orders with requested collection dates within one week of another by placing an order comment in the EHRS.
-
The specimen collection for the follow-up clinical UDS test shall occur within the timeframe defined by the laboratory order’s requested collection date. Staff may not collect the UDS specimen more than one calendar day prior to the requested collection date.
-
-
-
CLINICAL UDS COLLECTION & HANDLING
-
Clinical UDS requires special collection and handling to ensure the specimen is free from adulteration.
-
Taking into consideration facility physical limitation characteristics, arrangements shall be made to provide patient privacy during UDS specimen collection. This could consist of an enclosed stall in a multi-stall restroom; a single person restroom; a partitioned area allowing for individual privacy; a commode within a single-occupancy inpatient setting; or other acceptable location.
-
Staff shall review the following UDS collection instructions with the patient:
-
The patient should remove any unnecessary garments, such as a jacket with pockets, or personal property and leave the items outside the collection site.
-
Following the completion of collection, the patient should hand the closed container to the staff when instructed to do so.
-
The patient should not flush the commode unless instructed to do so by the staff.
-
The patient should not wash their hands until instructed to do so by the staff.
-
-
Staff do not directly observe the patient collecting the UDS sample, but shall actively proctor the process and perform the following actions subject to the limitations of custody, institution infrastructure, and physical layout:
-
Inspect the collection site before the patient enters the area to identify and remove potential specimen adulterants prior to patient entry.
-
Listen for unauthorized commode flushing or handwashing by the patient during the collection process.
-
Verify the patient provided the necessary sample volume as defined by the UDS requirements. If the sample volume is not adequate, discard the sample and reschedule the patient for a recollection within one calendar day.
-
Instruct the patient to wash their hands after the urine specimen collection is complete.
-
Inspect the commode bowl and collection site for unusual paraphernalia that could have been used to alter or substitute a specimen.
-
Flush the commode bowl or instruct the patient to do so following the completion of the specimen collection and inspection of the commode.
-
-
Staff shall observe the sample container for adulteration or substitution by observing the physical characteristics of the specimen such as:
-
Unusual color.
-
Temperature – the urine cup temperature strip should change from black to green signifying the sample is within the expected temperature range.
-
Presence of foreign objects or material
-
-
Staff shall document any observations in the EHRS indicating possible issues with specimen integrity.
-
Unless directed by the ordering provider or institution leadership, the corresponding UDS order shall be cancelled or discontinued by staff, and the ordering provider shall be notified of the circumstances leading to the cancellation or discontinuation.
-
-
Staff shall document the following circumstances in the EHRS indicating inability to collect adequate specimen:
-
The patient is unable to void, or an adequate sample volume was not collected by the patient, or both.
-
The patient fails to report (“no show”) for their scheduled UDS collection appointment, and the failure to report was not associated with a specimen collection refusal.
-
For laboratory orders exhibiting either of these two circumstances, staff shall attempt to reschedule the patient one-time within one subsequent calendar day for a second specimen collection attempt. Unless directed by the ordering provider or institution leadership, the corresponding laboratory order shall be cancelled or discontinued by staff if there was a failure to obtain an adequate specimen with no suspicion of adulteration within this one calendar day period. The ordering provider shall be notified of the circumstances leading to the cancellation or discontinuation of the order.
-
-
Specimens with an adequate volume and no reasonable suspicion of adulteration shall have a completed label. Ensure the collection cup is closed tightly and secure the lid with parafilm to prevent leakage in transit. If parafilm is used, lid tape is not required.
-
-
Appendix 2: Reception Center Clinical Urine Drug Screening
-
A Reception Center Clinical Urine Drug Screening (UDS) will be offered to all newly arriving individuals committed to CDCR custody. The UDS will be part of the Diagnostic Screening Tests and Assessments contained within the Reception Center Initial Health Screening and Triage as outlined in the Health Care Department Operations Manual, Section 3.1.8, Reception Center. The Reception Center Clinical UDS will be used for screening purposes only.
-
Reception Center Clinical UDS results, a patient’s decision to decline a Reception Center Clinical UDS, and/or a patient’s inability to provide a sample due to paruresis or other reason shall only be available to clinical staff on a need to know basis in order to manage a patient’s health care needs, and these results shall not be shared with custody.
-
RECEPTION CENTER CLINICAL UDS ORDERING
-
The order for Reception Center Clinical UDS testing will be available as part of the R&R New Arrival PowerPlan.
-
Reception Center Clinical UDS testing will consist of a CCHCS UTOX Screen test.
-
The Reception Center Clinical UDS testing will be ordered as a Routine collection priority.
-
RECEPTION CENTER CLINICAL UDS COLLECTION AND HANDLING
-
The Reception Center Clinical UDS screening does not require any special collection and handling requirements to safeguard the specimen from tampering due to the screening nature of the assessment.
-
Specimen can be collected following the process as other urine samples, and should follow the collection requirements particular to this test.
-
One UDS specimen container shall be given to the patient to collect the UDS and Chlamydia and Gonorrhea tests, if ordered.
-
Per the collection requirements, ensure the patient provided an adequate specimen volume to complete the Chlamydia and Gonorrhea and UDS testing.
-
If Chlamydia and Gonorrhea are ordered, transfer 2 mL of urine to the Aptima Transport tube, ensure urine volume is within the fill window on the Aptima tube. Label Aptima tube appropriately.
-
Process the UDS specimen per the Local Operating Procedure.
-
All urine specimen containers should be labeled appropriately. Staff shall record the specimen collection date and time and person collecting the specimen in the Electronic Health Record System and update the status of the specimen to “collected”.
-
Laboratory specimens shall not be labeled prior to collecting the specimen.
-
-
-
-
Appendix 3: Offsite Pathology Orders
-
A laboratory order for Offsite Pathology shall be placed in the electronic health record system (EHRS) when a patient has a pathology specimen collected from an offsite procedure.
-
This process does not apply to pathology specimens collected onsite at institutions and sent to a contracted laboratory for analysis.
-
OFFSITE PATHOLOGY NURSE AND PROVIDER MONITORING
-
The TTA, specialty, or designated nurse responsible for the intake of offsite specialty records shall receive the patient’s offsite records and forward them to their institution Health Information Management (HIM) unit for processing.
-
HIM shall follow-up with the offsite provider if no patient records accompany a patient returning from an offsite procedure.
-
-
HIM shall receive, prepare, scan, and upload the offsite records into the EHRS. An EHRS Message Center notification to the provider to review the records will be generated.
-
The provider shall review offsite procedure notes to verify the collection of a pathology specimen and its pending status from an offsite procedure.
-
-
OFFSITE PATHOLOGY WORKFLOW
-
The provider shall place an order for Offsite Pathology in EHRS if records indicate the collection of a pathology specimen during an offsite procedure.
-
Offsite Pathology shall be ordered as a Routine collection priority.
-
-
Institution laboratory staff shall monitor their Pending Log for any Offsite Pathology orders and notify HIM of any pending orders on a weekly basis. The Patient Care Team shall monitor the daily Huddle Report and EHRS Care Team Hub for pending Offsite Pathology orders and notify HIM of any pending orders on an as needed basis.
-
HIM shall scan and upload the report of the offsite pathology specimen results into EHRS once it is received.
-
HIM shall then forward the offsite pathology specimen report to the provider through a Message Center notification.
-
The provider shall review, endorse, and create the patient notification letter for the results within five calendar days of the date of receipt.
-
The patient notification letter created by the provider shall include the same elements listed in (c)(4)(E) of the procedure.
-
-
The regional or headquarters Senior Clinical Laboratory Technologist shall enter “See Scanned Report” in EHRS (PathNet) to close out the Offsite Pathology order.
-
3.1.15 Access to Contraceptive and Family Planning Services
-
Policy
-
California Correctional Health Care Services (CCHCS) shall provide female patients capable of becoming pregnant their choice of birth control methods and emergency contraception, approved by the United States Food and Drug Administration, unless medically contraindicated. CCHCS shall also provide nonprescription birth control methods, without the requirement of seeing a licensed health care provider.
-
CCHCS licensed health care providers trained on reproductive health and family planning services, based on their residency and board certification, shall provide nondirective, unbiased, and non-coercive prescription contraceptive services and contraceptive counseling.
-
-
Purpose
-
To ensure female patients capable of becoming pregnant have access to family planning services, contraceptive counseling, and the birth control method of their choice.
-
-
Responsibility
-
The Chief Executive Officer, or designee, is responsible for the implementation, monitoring, and evaluation of this policy.
-
-
Local Operating Procedure
-
Central California Women’s Facility, California Institution for Women, and Folsom Women’s Facility shall maintain a Local Operating Procedure (LOP) to implement the statewide policy. The elements of the LOP shall include, but not be limited to:
-
Availability of family planning services including contraceptive counseling services.
-
Availability of women’s health education.
-
Pre-parole counseling between 60-180 days prior to scheduled release.
-
Patient’s choice of birth control.
-
-
References
-
California Penal Code, Part 3, Title 2, Chapter 3, Section 3408
-
California Penal Code, Part 3, Title 2, Chapter 3, Section 3409
-
Revision History
-
Effective: 07/2017
Revised: 08/2021
-
3.1.16 Patient Care During Pregnancy and Childbirth
-
Policy
-
California Correctional Health Care Services (CCHCS) staff shall provide health care for the pregnant patient population. CCHCS provides comprehensive and coordinated health care services to female patients commensurate with each patient’s risk level and complexity, based on the Complete Care Model.
-
Every institution housing women shall provide obstetrical services, to include care for women with high-risk pregnancies. The California Institution for Women (CIW) is the designated institution for management of pregnancy and childbirth. All newly identified pregnant patients shall be referred for transfer to CIW.
-
-
Purpose
-
To ensure health care concerns are met for the pregnant patient population both pre- and post-partum and for the birth of children at local hospitals.
-
-
Responsibility
-
Statewide
-
CCHCS and California Department of Corrections and Rehabilitation (CDCR) departmental leadership at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial, and clinical systems are in place; and appropriate tools, training, technical assistance, and levels of resources are available so that care teams can successfully implement this procedure at all institutions housing female patients.
-
-
Regional
-
Regional Health Care Executives shall implement this procedure at the subset of institutions within an assigned region.
-
-
Institutional
-
The Chief Executive Officer (CEO), or designee, is responsible for the implementation, monitoring, and evaluation of this policy.
-
The CEO shall implement and manage a system to administer this procedure. The CEO shall delegate decision making authority to designated institutional health care executives for daily operations of the health care delivery system and ensure adequate resources are deployed to support the required elements of care including, but not limited to:
-
Provision of obstetrical services 24 hours per day.
-
Emergency treatment and management of preterm and/or precipitous delivery 24 hours per day.
-
Provision of hospital, surgical, and neonatal services.
-
Provision of social and educational services pertaining to pregnancy, childbirth, and health promotion.
-
-
The CEO and all members of the institution leadership team shall ensure all necessary resources are in place to support the successful implementation of this procedure at all levels; including, but not limited to:
-
Institution level
-
Patient panel level
-
Patient level
-
-
The CEO and all members of the institution leadership team shall ensure access to and utilization of equipment, supplies, health information systems, patient registries and summaries, and evidence-based guidelines, to ensure coordinated and integrated management of pregnancy and childbirth.
-
-
-
Local Operating Procedure
-
Each CDCR institution shall maintain a Local Operating Procedure to implement the statewide procedure.
-
-
Procedure
-
General Requirements
-
CCHCS staff shall conduct a health screening, including a pregnancy screening, for all female patients upon reception into CDCR per the Health Care Department Operations Manual (HCDOM), Section 3.1.8, Reception Center.
-
The Primary Care Provider (PCP) shall evaluate patients within seven calendar days of self-reporting a suspected pregnancy or when pregnancy is suggested by the patient’s physical appearance, and/or signs and symptoms of pregnancy are present.
-
CCHCS staff shall obtain diagnostic studies within three business days of evaluation by the PCP.
-
Upon confirmation of pregnancy, the Primary Care Team (PCT) shall provide and/or coordinate obstetrical and health care services to include a written plan of care.
-
A patient may request to receive medical services from the PCP of her choice who is credentialed and trained in obstetrics.
-
Any expenses incurred by the services of a non-CCHCS/CDCR PCP of the patient’s choice shall be the responsibility of the patient.
-
-
Patients with health care conditions that increase the risk of, or cause a high-risk pregnancy shall be identified, monitored, and referred to the appropriate level of care as indicated by health care needs of the patient.
-
PCPs at institutions housing female patients other than CIW shall refer newly identified pregnant patients to CIW by referral to the Classification and Parole Representative (C&PR) and the completion of a Medical Classification Chrono.
-
The PCT shall ensure communication with the receiving institution and other health care providers, regarding the patient’s status and treatment plans to ensure continuity and coordination of services.
-
Transfer shall occur in accordance with the procedure outlined in the HCDOM, Section 3.1.9, Health Care Transfer. Special consideration shall be given to ensure that the appropriate mode of transportation is selected and communicated.
-
-
Care of Pregnant Patients
-
Pregnant patients shall be provided the following:
-
An initial Obstetrician visit scheduled to occur within seven days of pregnancy diagnosis.
-
Diagnostic studies; ordered as medically necessary.
-
A determination of the gestational age of the pregnancy and the estimated due date.
-
The option to be tested for human immunodeficiency virus (HIV) and other communicable diseases that may be transmitted to the infant.
-
Education by the PCP or Obstetrician regarding the patient’s choice of pregnancy options such as:
-
Continuing pregnancy and childcare plans.
-
Prenatal health care.
-
Adoption services.
-
Termination services.
-
Emergency contraceptives.
-
Obtaining pregnancy information pamphlets or other pertinent educational material.
-
-
CCHCS staff shall document the provision of this education in the health record.
-
-
Pregnant patients shall be prescribed or issued:
-
Prenatal vitamins (which contain iron and folic acid).
-
Dietary supplementation to include:
-
Two extra cartons of milk,
-
Two extra servings of fresh fruit, and
-
Two extra servings of fresh vegetables daily.
-
Additional nutrients or supplements and/or referral to a Dietitian as necessary.
-
-
A CDCR 7410, Comprehensive Accommodation Chrono, for lower bunk and lower tier housing if housed in a multi-tier housing unit and any other necessary medical clearances or restrictions in their third trimester or earlier, if medically indicated.
-
A referral to the dentist on a priority basis, as early in the second trimester of gestation as possible, for a comprehensive dental examination and treatment of periodontal disease.
-
-
The PCP shall prepare a CDCR 7252, Request for Authorization of Temporary Removal for Medical Treatment, for all pregnant patients within 30 calendar days prior to the estimated delivery date. This form shall be taken to the Watch Commander’s office in advance to enable custody staff to prepare the patient for transportation to an outside facility in a timely manner.
-
Unless otherwise indicated by the Obstetrician, pregnant patients shall be scheduled for their obstetrical visits as follows:
-
Every four weeks in the first trimester and up to 24-28 weeks gestation.
-
Every two weeks thereafter up to 36 weeks gestation.
-
Weekly after 36 weeks gestation up to delivery.
-
-
Pregnant patients shall be provided additional health care services as medically indicated.
-
Every pregnant patient electing to continue her pregnancy shall be referred to a Medical Social Worker for case management to discuss placement of her child and options available for placement and care of the child after delivery.
-
The Medical Social Worker shall discuss the options available for feeding, placement, and care of the child after birth, including the benefits of lactation, and provide the pregnant patient with access to the written application for community-based programs serving pregnant, birthing, or lactating patients.
-
A Medical Social Worker shall assist the pregnant patient to contact relatives regarding newborn placement, even while still in process of reception.
-
The Medical Social Worker shall initiate and oversee the management of all newborn placements.
-
-
A pregnant patient electing to terminate her pregnancy shall be offered all due medical care and accommodations until she is no longer pregnant. Refusal of medical care shall be documented in the health record and signed by the patient.
-
If clinically appropriate, patients shall be provided opioid maintenance therapy during pregnancy.
-
-
Outside Consultation or Non-Emergent Labor and Delivery
-
Transportation shall occur in accordance with the procedure outlined in the HCDOM, Section 3.1.9, Health Care Transfer. Special consideration shall be given to ensure that the appropriate mode of transportation is selected and communicated.
-
Copies of all prenatal forms and the completed CDC 7243, Health Care Services Physician Request for Services, shall accompany the pregnant patient when sent for medical treatment or consultation to an outside facility, and when the patient is transported for labor and delivery.
-
When a patient returns, the Registered Nurse (RN) on duty in the Triage and Treatment Area (TTA), Outpatient Housing Unit (OHU), or Correctional Treatment Center (CTC) shall receive the patient’s paperwork from the outside facility. The receiving nurse shall notify the PCP and Obstetrician of the patient’s return, medical status, recommendations from the outside consultant, and ensure the documents are filed in the health record.
-
-
Emergency Onsite Delivery
-
In emergent circumstances where the patient cannot be transported offsite prior to delivery of a baby, the patient shall be given the maximum level of privacy possible during the labor and delivery process. If custody personnel must be present, they shall be stationed outside the room absent extraordinary circumstances.
-
Infants delivered onsite shall be provided all appropriate care until transported to an offsite facility.
-
-
Emergency Transport
-
A pregnant patient in labor shall be treated as an emergency and be transported immediately via ambulance. In the event of an emergency transport for the delivery of a baby, the Obstetrician and PCP shall be immediately notified and provide appropriate assistance and orders.
-
Application of restraint gear and physical restraints shall be in accordance with California Penal Code, Sections 3407 and 3423, and the Department Operations Manual, Sections 54045.1 and 54045.11. If handcuffs are applied, the patient’s arms shall be brought to the front of the body for application.
-
-
Postpartum Care
-
Postpartum patients who deliver a child via cesarean section shall be admitted to the OHU or CTC via the TTA upon return to the institution.
-
All postpartum patients who deliver a child vaginally shall be assessed by the TTA RN who shall review discharge orders and initiate postpartum care following communication and receipt of orders with the PCP or Physician-on-call and determine appropriate housing.
-
If postpartum mental health concerns present, the patient shall be referred to Mental Health Services for proper assessment and treatment.
-
CCHCS staff shall schedule follow-up appointments, articulate treatment plans, and determine the need for continued limited duty, for all postpartum patients including the following:
-
A postpartum examination within one week from childbirth and as needed for up to 12 weeks postpartum.
-
At least 12 weeks of recovery after any childbirth before being required to resume normal activity.
-
-
Patients shall be afforded family planning services if their release and/or parole date falls within 12 to 16 weeks after delivery.
-
-
Local Oversight – Designated Standing Improvement Committee
-
Institution leadership shall designate an existing standing committee that reports to the local Quality Management Committee for oversight of the systems and processes dedicated to the health care management of pregnant patients.
-
Tracking and monitoring activities shall be performed to identify trends and opportunities for improvement.
-
-
-
References
-
Code of Federal Regulations, Title 42, Chapter 1, Subchapter A, Part 8, Subpart B, Section 8.12(f)(3), Federal Opioid Treatment Standards
-
California Penal Code, Part 3, Title 2, Chapter 3, Sections 3406-3409
-
California Penal Code, Part 3, Title 2, Chapter 4, Section 3423
-
California Code of Regulations, Title 9, Division 4, Chapter 4, Subsection 5, Article 3, Section 10360, Additional Requirements for Pregnant Patients
-
California Code of Regulations, Title 15, Division 3, Chapter 1, Subchapter 2, Article 7, Section 3170-3182, Visiting
-
California Code of Regulations, Title 15, Division 3, Chapter 1, Subchapter 4, Article 7, Section 3337, Classification Hearing of Administrative Segregation Placements
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.1, Complete Care Model
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.8, Reception Center
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.9, Health Care Transfer
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.10, Specialized Health Care Housing
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.12, Outpatient Dietary Intervention
-
Health Care Department Operations Manual, Chapter 3, Article 2, Section 3.5.1, Pharmacy and Medication Services
-
California Department of Corrections and Rehabilitation, Department Operations Manual, Article 45, Sections 54045.1 and 54045.11
-
-
Revision History
-
Effective: 01/2006
Revised: 09/2021
-
3.1.17 Palliative Care and Treatment
-
Policy
-
California Correctional Health Care Services shall provide palliative care and treatment when the patient’s terminal condition results in pain or disability to such an extent that the Primary Care Provider (PCP) and the patient determine that only palliation shall be provided.
-
-
Purpose
-
To provide comfort, relief from pain, and support when the patient has received a terminal diagnosis.
-
-
Responsibility
-
The Chief Executive Officer (CEO), or designee, is responsible for the implementation, monitoring, and evaluation of this policy and procedure.
-
-
Procedure
-
The PCP shall evaluate the patient and prescribe a treatment plan as warranted for palliative or comfort care. The ordered treatment shall be documented in the patient’s health record.
-
A pain control plan (including narcotics) will be developed with the assistance of a specialist, as appropriate.
-
If the patient meets the criteria for hospice care, the PCP shall complete a referral to California Department of Correction’s and Rehabilitation’s hospice at the California Medical Facility.
-
Hospice Admission Criteria are as follows:
-
The hospice has obtained written acceptance for hospice care from the Chief Medical Executive (CME) or designee.
-
The hospice PCP and/or CME, or designee, certifies a prognosis six months or less if the disease follows its expected course.
-
The patient and/or designated legal representative agrees to palliative goals/philosophy of hospice services.
-
The custody level generally cannot be greater than a Close A and there cannot be any other precluding custody considerations (such as enemy situations).
-
The hospice has the ability to meet the needs of the patient according to the level and intensity of care required.
-
There are adequate, cooperative efforts by the patient to follow safety measures and the plan for medical and non-medical emergencies.
-
The hospice has adequate, suitable personnel and resources to provide the services required by the patient.
-
-
Patients receiving palliative care shall continue to receive medically necessary treatment for other medical conditions that may occur or coexist such as injuries, infections, and chronic conditions (e.g., hypertension, diabetes).
-
Refusal of medically necessary care shall be documented in the health record.
-
Health care staff shall inform patients that they may obtain a consultation with the Mental Health Services Department or Chaplain of their choice within the institution. Upon the patient’s request, an appointment shall be made with the requested discipline.
-
The CEO shall request a Compassionate Release when appropriate.
-
Health care staff shall follow the Department’s procedure for Advance Directives.
-
-
References
-
Health Care Department Operations Manual, Chapter 1, Article 2, Section 1.2.14, Medical Classification System
-
Health Care Department Operations Manual, Chapter 2, Article 4, Section 2.4.1, Advance Directive for Health Care
-
-
Revision History
-
Effective: 01/2006
Revised: 09/2015
-
3.1.18 Registered Nurse Pronouncement of Death
-
Policy
-
California Correctional Health Care Services (CCHCS) shall permit Registered Nurses (RNs) to make the determination and pronouncement of patient death under specified circumstances. This policy applies only to determining the irreversible cessation of circulatory and respiratory function.
-
-
Purpose
-
To ensure timely pronouncement of death by an RN when death is expected and the attending provider is not available onsite.
-
-
Responsibility
-
Statewide
-
California Department of Corrections and Rehabilitation (CDCR) and CCHCS departmental leadership at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial, and clinical systems are in place and appropriate tools, training, technical assistance, and levels of resources are available to ensure this policy is successfully implemented and maintained.
-
-
Regional
-
Regional Health Care Executives are responsible for implementation of this procedure at the subset of institutions within an assigned region.
-
-
Institutional
-
The Chief Executive Officer, Chief Medical Executive, and Chief Nursing Executive are responsible for the local implementation, monitoring, and evaluation of this policy.
-
-
-
Procedure
-
Conditions under which an RN may determine and pronounce death
-
All of the following conditions must be met:
-
A health care provider, (Primary Care Provider [PCP], or an Advanced Practice Provider [APP]) has determined and documented that the patient has a condition or illness whose natural course is expected to end in death and/or the patient’s life expectancy is six months or less.
-
The patient signed a CDCR 7465, Physician’s Orders for Life Sustaining Treatment indicating “Do Not Resuscitate.” The CDCR 7465 must be signed and dated by a physician, or an APP acting under the supervision of the physician, and the patient or legally recognized health care decision maker.
-
The patient’s health care provider has documented in a progress note in the health record that death is expected and has completed an order in the Electronic Health Record System (EHRS) stating that an RN may pronounce death.
-
-
Assessment and Determination of Death
-
When a patient appears to have died, the RN on duty shall be physically present at the bedside to assess the patient utilizing these steps:
-
Check the patient identification card or the Strategic Offender Management System database to verify the patient photo, CDCR number, and date of birth.
-
Assess response to tactile stimuli by gently tapping the patient in the shoulder and calling their name.
-
Assess pupils for response to light.
-
Assess for spontaneous respirations by observing the chest wall for rise and fall for a full minute to confirm absence of respirations.
-
Assess for carotid and radial or brachial pulse for a full minute and confirm absence of heart beat by auscultating heart sounds using a stethoscope for a full minute to determine cessation of cardiac activity.
-
After performing the above assessment, the RN shall pronounce the time of death.
-
-
Documentation of Death
-
The RN shall document the assessment in the health record on the Notification of Death/RN Pronouncement of Death PowerForm as follows:
-
No carotid and peripheral pulse.
-
Pupils are fixed and nonreactive to light.
-
No response to tactile stimuli.
-
No respirations for full minute.
-
No heart sound for full minute.
-
Date and time of death.
-
-
Notifications of Death
-
The RN shall provide the following notifications:
-
Nursing supervisor
-
Watch Commander
-
Physician on-call shall be notified via phone call or EHRS messaging if the PCP is not available in situations such as after business hours, weekends, and holidays.
-
-
Notification of the next of kin
-
After the RN has notified the watch commander of a patient’s death, the Warden or designee at the level of Correctional Lieutenant or Correctional Counselor II or above is responsible for the notification of the next of kin pursuant to the CDCR Department Operations Manual, Section 51070.10.
-
-
Training Requirements
-
RNs shall receive an initial training upon implementation of this policy and biennially as part of their competency validation.
-
-
-
References
-
California Health and Safety Code, Division 7, Part 1, Chapter 3.7, Article 1, Section 7180, The Uniform Determination of Death Act
-
California Business and Professions Code, Division 2, Chapter 6, Article 2, Section 2725
-
California Emergency Medical Services Authority, Physician Orders for Life-Sustaining Treatment
-
California Correctional Health Care Services, Health Care Department Operations Manual, Chapter 2, Article 4, Section 2.4.2, Physician Orders for Life-Sustaining Treatment (POLST)
-
California Department of Corrections and Rehabilitation, Department Operations Manual, Article 7, Section 51070.10
-
-
Revision History
-
Effective: 10/2020
-
Revised: 08/20/2025
-
3.1.19 Next of Kin Notification for Death, Serious Illness, or Serious Injury
-
Policy
-
California Department of Corrections and Rehabilitation (CDCR), California Correctional Health Care Services (CCHCS) shall comply with all applicable state laws and regulations for notifying next of kin (NOK) for death, serious illness, serious injury, including incidents of serious injury due to self-harm, suicide attempts, or accidents.
-
-
Purpose
-
To standardize a statewide process for the initial notification to NOK and for providing necessary medical and mental health information related to the patient’s death, serious illness, or serious injury including incidents of serious injury due to self-harm, suicide attempts, or accidents.
-
-
Responsibility
-
Statewide
-
CCHCS and CDCR departmental leadership at all levels of the organization, within the scope of their authority, shall ensure administrative, custodial, and clinical systems are in place and appropriate tools, training, technical assistance, and levels of resources are available to ensure this policy is successfully maintained.
-
-
Regional
-
Regional Health Care Executives are responsible to maintain this policy at the subset of institutions within an assigned region.
-
-
Institutional
-
The Warden, or designee, is responsible for the initial NOK notification for death, serious illness, or serious injury including incidents of serious injury due to self-harm, suicide attempts, or accidents.
-
The Chief Executive Officer (CEO) is responsible for the local monitoring and evaluation of this policy at their assigned institution.
-
-
-
Procedure
-
The Primary Care Provider (PCP) or the Provider on-Call (POC) shall inform the Chief Medical Executive (CME), or designee, as soon as practicable that a patient has a serious illness, serious injury, or has been declared deceased. For serious injury caused by a probable self-harm or suicide, the Triage and Treatment Area Registered Nurse shall inform the Chief of Mental Health (CMH), or designee, if indicated.
-
The CME, or designee, shall conduct a review of relevant patient health information to determine if the patient has a serious illness, or injury, or to determine possible etiology of death if the patient has been declared deceased. The CMH, or designee, shall conduct a review of relevant patient health information to determine if the patient committed serious self-harm or suicide attempts requiring hospital admission or resulting in death.
-
After determining that the patient met the criteria for a serious illness or injury, or has been declared deceased the CME/CMH, or designee, shall inform the Warden, or designee as soon as practicable, to initiate the NOK notification. For serious injury or death caused by a probable self-harm or suicide attempt, the CMH, or designee, shall have a follow-up call to the NOK if they are listed on the CDCR 7385, Authorization for Release of Protected Health Information within 72 hours from the initial call, to confirm that the serious injury or death is due to self-harm or suicide attempt.
-
The Warden, or designee, shall:
-
Contact the NOK as identified in the Electronic Records Management System (ERMS) within 24 hours after the patient’s condition is deemed to be serious by a health care provider or the patient has been declared deceased.
-
Document and track the initial NOK notification.
-
Provide the patient’s NOK, or authorized agent, the Institutional Patient Health Care Inquiry (PHCI) Hotline phone number to call for additional information related to the patient’s medical and or mental health condition.
-
-
All calls received by the Institutional PHCI Hotline shall follow procedures and operating standards pursuant to the CCHCS Health Care Department Operations Manual (HCDOM), Section 2.3.15, Headquarters Patient Health Care Inquiry Response.
-
If the patient lacks the capacity to make their own health care decisions, and a health care decision needs to be made, the primary source of information for a health care proxy is the advance directive or the CDCR 7465, Physician Order for Life Sustaining Treatment. The person listed on the advance directive or the CDCR 7465 shall receive relevant health care information regardless of whether they are listed on the CDCR 7385. If the patient does not have an advance directive, or CDCR 7465, the NOK listed in ERMS shall be notified.
-
Health care information shall only be provided to or shared with the person or agent listed on the CDCR 7385, unless as stated above in (d)(6).
-
In cases of serious illness or serious injury, health care information shall not be released to the NOK if the patient did not list the NOK on the CDCR 7385. However, if the patient is declared deceased, an estate executor, administrator, or any other person authorized by state law shall act as the representative of the decedent and shall have access to the decedent’s protected health information.
-
-
Training Requirements
-
The CEO shall ensure that all applicable health care staff complete the Next of Kin Notification for Death, Serious Illness, or Serious Injury training on the Learning Management System.
-
-
References
-
Title 45 Federal Code of Regulations, Section 164.502(g)(4), Uses and Disclosures of Protected Health Information: General Rules
-
California Penal Code, Part 3, Title 1, Chapter 2, Article 1, 2064.1
-
California Penal Code Part 3, Title 7, Chapter1, Section 5022
-
California Code of Regulations Title 15, Division 3, Chapter 2, Subchapter 3, Article 13, Section 3999.417
-
California Department Operations Manual, Chapter 5, Article 7, Section 51070.1 and 50170.10
-
California Correctional Health Care Services Health Care Department Operations Manual, Section 2.3.4, Release of Information
-
California Correctional Health Care Services Health Care Department Operations Manual, Section 2.3.15, Headquarters Patient Health Care Inquiry Response
-
-
Revision History
-
Effective: 06/2022
-
-
3.1.20 Clinical Photography/Digital Imaging
-
Policy
-
California Correctional Health Care Services (CCHCS) shall ensure availability of digital imaging equipment at California Department of Corrections and Rehabilitation (CDCR) institutions for health care staff to monitor the stages of healing or progression of disease, detect the presence of complications, and review the effectiveness of treatment. Digital imaging equipment shall not be used to document use of force incidents; however, consistent with this policy, digital imaging may be used for clinical management of any illness or injury, including those associated with use of force incidents.
-
-
Purpose
-
To provide guidelines for the use of digital imaging equipment for clinical documentation and to provide baseline and ongoing assessment for the surveillance and monitoring of medical conditions or disease status.
-
To serve as an adjunct to the written assessment.
-
To provide guidance for safeguarding the digital imaging equipment from tampering, loss, or destruction.
-
-
Responsibility
-
Statewide
-
CCHCS and CDCR departmental leadership at all levels of the organization shall ensure administrative, custodial, and clinical systems are in place and appropriate tools, training, technical assistance, and levels of resources are available to successfully apply this policy and procedure.
-
-
Regional
-
Regional Health Care Executives are responsible for application of this policy and procedure at the subset of institutions within an assigned region.
-
-
Institution
-
The Chief Executive Officer has overall responsibility for compliance with and ongoing oversight of this policy and procedure.
-
The Chief Medical Executive and Chief Nurse Executive are responsible for the overall daily operations.
-
The Health Information Management staff are responsible for ensuring all established rules and guidelines for the release of health information and record retention of digital images are followed.
-
Information Technology (IT) staff are responsible for ensuring equipment is maintained and in good working order.
-
-
-
Procedure
-
Camera Storage and Maintenance
-
The camera shall only be used by designated health care staff for conducting medical assessments, including skin or other anatomical area assessments and/or wound management.
-
The camera shall be affixed with a CCHCS asset tag for tracking purposes.
-
The camera shall be added to the clinic’s Master Tool Inventory.
-
The camera and its accessories (e.g., USB cable and wound measuring rulers), or a state issued cell phone, shall be stored in a locked drawer separate from any other items located in the locked medication room of one or more of the following areas:
-
Registered Nurse Line clinic of every yard
-
Triage and Treatment Area
-
Correctional Treatment Center
-
Outpatient Housing Unit
-
Licensed inpatient units
-
Telemedicine clinic areas that have locked telemedicine carts with cameras
-
-
The drawer in the locked medication room shall be clearly labeled with the drawer contents.
-
The camera shall be kept in the locked drawer until the time of use. After each use, the camera shall be cleaned, according to manufacturer instructions, placed in the carrying case, and returned to the designated drawer.
-
The camera and its accessories shall not be left exposed to the environment or left unsecured.
-
Nursing staff shall account for the camera and its accessories at the beginning of each shift and document the presence of the camera, by signing the Tool Inventory Report.
-
In the event of a broken or missing camera or accessory, nursing staff shall immediately notify the Supervising Registered Nurse (SRN) II and unit custody officer, and search the immediate area.
-
The SRN II shall notify the Watch Commander. The nursing staff shall submit a memorandum to the following staff:
-
Watch Commander (missing camera only)
-
SRN II
-
SRN III
-
Tool Control Officer
-
IT (for cameras that are broken or not in proper working condition)
-
Information Security Office (for missing cameras)
-
-
-
-
Cell Phone
-
If cell phones are used, only state-issued cell phones with CCHCS asset tags, belonging to the designated health care staff obtaining the digital image, are allowed for conducting medical assessments, including skin or other anatomical area assessments and/or wound management.
-
-
Clinical Photograph/Digital Image Quality and Accuracy
-
Designated health care staff shall obtain a digital image of anatomical/wound areas to be evaluated on initial assessment and if any changes are identified to assist in assessment and management. Digital images are an adjunct to assessment documentation and serve only to support the written wound documentation and does not replace it.
-
Unless clinically indicated, digital images shall not be obtained solely for the purpose of documenting a use of force incident. Refer to the Department Operations Manual, Chapter 5, Adult Custody and Security, Article 2, Use of Force.
-
Digital images taken as a part of the health record shall:
-
Be recorded only on digital cameras and/or cell phones owned by CCHCS and designated solely for clinical management. They shall not be taken on cell phones or cameras that are the personal property of staff.
-
Not be stored on external or removable memory devices (e.g., flash drives).
-
-
Prior to taking any digital images , the designated health care staff shall obtain the following patient consent:
-
Written Consent: Designated health care staff shall ensure a CDCR 7120, Informed Consent for Clinical Photography/Digital Imaging is completed and scanned into the health record for each patient.
-
Pursuant to the Health Care Department Operations Manual, Section 2.2.2, Use and Disclosure of Protected Health Information Based on Patient Authorization, in instances where release of HIV/AIDS status may be required to diagnose or treat a patient’s condition, written authorization from the patient is required on a CDCR 7385, Authorization for Release of Protected Health Information.
-
-
Verbal Consent: Designated health care staff shall request verbal consent prior to each encounter in which a digital image will be taken. If the patient has a signed CDCR 7120 within their health record but then verbally declines at the time of the encounter or appointment, the verbal consent would supersede the written consent. Refusals shall be documented on the CDCR 7225, Refusal of Examination and/or Treatment.
-
-
To ensure consistency within a series of digital images, the following steps shall be followed:
-
The date and time shall be pre-set on the digital camera so that the correct timestamp is recorded when the image is taken. State issued cell phones have the date and time automatically preset by the carrier.
-
The preferred background color is white (an under pad, pillowcase, towel, or sheet may be used) but solid; neutral colors are acceptable; blue and green backgrounds shall be avoided.
-
Ensure correct lighting.
-
Whenever possible, take the digital image at the same time of day to help with lighting consistency.
-
Use as much natural light as possible and keep artificial light use to a minimum to avoid shadows and discoloration.
-
If evenly distributed lighting exists in the room, turn the flash off. The use of a flash may obscure essential detail in the glare of moist surfaces or cause white out; however, the use of flash may help to eliminate shadows. When necessary, take the digital image with and without flash to test for best image quality.
-
-
Remove the existing dressing(s), if present, and cleanse the affected area(s) as ordered.
-
Place a fresh chux pad beneath the anatomic area to be photographed.
-
-
Position the patient.
-
If possible, the patient shall be photographed lying down, because some wounds/areas distort easily.
-
Where the patient’s body naturally curves, such as wounds/areas extending around a limb or heel, photograph such wounds in a “relaxed” position and then again with the assistance of other designated health care staff.
-
-
Prepare the wound measuring ruler by labeling it with the following:
-
Patient’s name;
-
Date of birth;
-
CDCR number;
-
Current date; and
-
Anatomic site being evaluated. Sites must be labeled consistently with the identification of the area within the health record.
-
-
Place the ruler in the field of view and position the digital device perpendicular (90-degree angle) to the wound/area at a distance of one to three feet.
-
Any subsequent photographic recordings shall be taken with the patient and digital device in the same position.
-
-
Focus the camera.
-
Consideration must be given at all times to protect the patient’s physical appearance within the digital image itself.
-
Care must be taken to respect the dignity, ethnicity, and religious beliefs of the patient.
-
The patient’s modesty must also be maintained by ensuring minimal patient skin exposure.
-
Avoid identifying characteristics (e.g., full face in the digital image).
-
Digital images of the face may be necessary if that is the location of wound/area needed for dermatology review or for documentation of injury to the face. Digital images shall only be taken to focus on the affected area.
-
-
Take the digital image.
-
Multiple digital images may be necessary to document detail, wound/area positioning, and involved body part.
-
Avoid including any staff hands in the image whenever possible.
-
Preview digital images taken to ensure they are at the proper angle, include all the pertinent information, and are clear and visible. Retake if necessary.
-
-
-
-
Transmission and Destruction
-
All digital images shall be uploaded to the health record multimedia manager by the designated health care staff who took the photograph as soon as possible.
-
Digital images shall not be altered in any way using any software, except for making the size of the entire photo larger or smaller for printing purposes.
-
Digital images taken of patients shall be stored on the digital camera and/or state-issued cell phone only until they are uploaded to the health record multimedia manager. Digital images intended for a consulting specialist shall be sent and received via Health Insurance Portability and Accountability Act (HIPAA)-compliant electronic transmission.
-
Once the digital images have been uploaded and verified, all digital images shall be immediately deleted from the digital device by the designated health care staff who took the photograph. The digital image must be destroyed to ensure the data cannot be reconstructed and the digital image is rendered unusable or inaccessible, including any data back-ups once transfer of the digital image is complete. No other copies shall be made or transferred to any other location.
-
Digital images shall be removed from the device before it is available for re-use. Regardless of the final intended destination, internal or external to CCHCS, the device shall not contain residual representation of any data that would allow re-construction.
-
If a device is found to contain digital images from previous documentation, staff shall ensure the digital images were appropriately documented in the patient’s health record prior to destruction. The SRN II or Physician Manager shall be notified and ensure proper training is provided to designated health care staff responsible for obtaining digital images.
-
Digital images may be transferred via the Health Information Exchange only when necessary to ensure consulting providers or specialists can evaluate progression of wound or disease in which the patient is being evaluated to provide appropriate treatment recommendations.
-
Digital images may be used as part of the peer review or disciplinary process in instances where a patient’s health care was delayed or deficient, or instances where the digital image was used, copied, transferred, or disclosed in any manner that violates HIPAA.
-
Digital images are subject to re-disclosure, if the patient has consented in writing for those images.
-
-
Training
-
Training and competency validation in clinical photography and safe storage of digital images shall be provided by the SRN II or Physician Manager to designated health care staff prior to use.
-
Competency in clinical photography shall be validated annually. Validation may include direct observation, review of digital images taken by designated health care staff, or other validation methods to ensure competence.
-
Designated health care staff shall read all digital device instructions prior to use.
-
-
-
References
-
Federal Agencies Digital Guidelines Initiative Glossary
http://www.digitizationguidelines.gov/term.php?term=digitalimage -
Law Insider
https://www.lawinsider.com/dictionary/medical-assessment -
California Office of Health Information Integrity, California’s Statewide Health Information Policy Manual, Chapter 3, Section 3.2.2, Device and Media Controls
-
Department Operations Manual, Chapter 5, Adult Custody and Security, Article 2, Use of Force
-
Health Care Department Operations Manual, Chapter 2, Article 2, Section 2.2.2, Use and Disclosure of Protected Health Information Based on Patient Authorization
-
-
Revision History
-
Effective: 12/2021
-
3.1.21 Care Team Enhanced Conference
-
Policy
-
The Care Team Enhanced Conference (CTEC) is an interdisciplinary forum to facilitate collaborative development of individualized patient care, treatment, and coordination plans. Care Teams shall use all available resources to optimize each patient’s physical, mental health, cognition, and functional status. The CTEC is an optional forum designed to support institutional, interdisciplinary Care Teams and enhance the effectiveness of their management of patients with complex conditions by mobilizing additional resources across the organization to overcome barriers. The CTEC promotes interdisciplinary communication, coordination, and collaboration to achieve optimal outcomes using a patient-centered whole person care approach. The CTEC process serves as a secondary intervention in establishing treatment plans for complex patients after institutional interdisciplinary teams perform an assessment and/or elevate the case to an institution designated Standing Improvement Committee (e.g., Institution Utilization Management Committee or Medical Program Committee), which reports to the local Quality Management Committee.
-
-
Responsibility
-
Statewide
-
California Correctional Health Care Services (CCHCS) Deputy Director, Medical Services, and the Deputy Medical Executive, Integrated Care and Complex Patient Populations Program, are responsible for the statewide oversight and ongoing support of CTEC.
-
Support of a CTEC is managed through a Headquarters (HQ) Complex Care Team, which includes clinical and support staff within the Integrated Care and Complex Patient Populations Program.
-
-
Regional
-
Regional leadership across all disciplines is responsible for ensuring the CTEC is used as needed to appropriately manage and care for patients at the subset of institutions within their assigned region.
-
-
Institution
-
The health care leadership is responsible for the following as it relates to their specific discipline:
-
Overall health care staff adherence to this procedure at the institution.
-
Health care staff participation in the CTEC as appropriate.
-
Implementation of the interdisciplinary care plans and documentation within the health record.
-
Elevate individual patient cases to a designated Standing Improvement Committee, as needed.
-
-
-
Procedure
-
Request for CTEC
-
A CTEC may be requested by any person advocating on behalf of the patient by emailing the HQ Complex Care Team at CCHCSComplexCare@cdcr.ca.gov.
-
Upon receipt of the emailed request, a member from the Complex Care Team shall send a CTEC Request Form to the Care Team for completion prior to scheduling a conference.
-
The CTEC Request Form shall include:
-
Patient identifying information and the clinical question(s) or specific issue(s) that are intended to guide the scheduled CTEC.
-
Preferred CTEC meeting day/time.
-
Any additional relevant accompanying information.
-
-
The CTEC Request Form and additional documentation (i.e., relevant progress notes, mental health notes, labs and/or studies) shall be submitted to the HQ Complex Care Team at: CCHCSComplexCare@cdcr.ca.gov.
-
-
CTEC Attendees
-
The HQ Complex Care team in collaboration with the Institution Care Team and Leadership determine the recommended CTEC attendees based on the patient’s condition and treatment needs, case complexity, and circumstances surrounding the clinical question(s). CTEC attendees may include representatives from various disciplines within the institution as well as regional, HQ and community health care facility staff.
-
CTEC attendees may include the patient’s Care Team and institution leadership, as follows:
-
Person who requested the CTEC
-
Primary Care Provider (PCP)
-
Primary Care Nurse
-
Primary Mental Health Clinician
-
Primary Psychiatrist
-
Chief Medical Executive or Chief Physician & Surgeon
-
Chief Nursing Executive or designated Nursing Supervisor
-
Institution Chief Mental Health Program or Senior Psychologist Supervisor
-
Institution Chief or Senior Psychiatrist Supervisor
-
Supervising Dentist
-
Pharmacist–In-Charge
-
Custody Representative(s)
-
Developmental Disability Program Psychologist (If patient is part of DDP)
-
Developmental Disability Program Officers
-
Other institution representatives as needed
-
-
Supplemental attendees are selected as needed to seek additional perspectives and the opinion and expertise of others relevant to the circumstances or challenges of the case. Other attendees may include, but are not limited to:
-
Hospital representative
-
Utilization Management Nurse Care Manager
-
Internal specialist (e.g., Addiction Medicine, Hepatitis C Virus, HIV, Wound Care, Physical Medicine and Rehabilitation, Pain)
-
Resource Nurse
-
Public Health representative
-
The Ethics Committee representative
-
California Department of Corrections and Rehabilitation and/or CCHCS Office of Legal Affairs representative
-
CCHCS Regional representatives of applicable disciplines/programs
-
CCHCS HQ representatives of applicable disciplines/programs
-
Board of Parole Hearings representative
-
Division of Adult Parole Operations representative
-
-
-
CTEC Meetings
-
Following the receipt of the CTEC Request Form, the HQ Complex Care Team member shall reach out to the attendees within five calendar days with date and time options for scheduling CTEC.
-
CTEC meetings shall be facilitated by a HQ Complex Care Team member.
-
CTEC meetings shall be scheduled using a video teleconferencing format.
-
During the CTEC meeting, the PCP shall present a brief summary related to pertinent history, diagnostics and treatments, as well as questions posed for discussion.
-
Discussions surrounding individualized care, treatment, and coordination plans shall take place between the CTEC attendees following the PCP presentation.
-
The patient’s plan of care shall be defined with action items documented by the patient’s PCP.
-
If clinically indicated, a follow-up meeting may be scheduled with CTEC attendees to discuss patient treatment progress and next steps.
-
-
Electronic Health Record Documentation
-
Within three calendar days after the CTEC, the PCP shall summarize recommendations and plan of care in a progress note within the patient’s health record. The progress note shall detail the specific action items discussed during the CTEC and persons responsible for carrying out those action items. Orders and referrals shall also be submitted within three calendar days on the treatment plan, if applicable.
-
The interdisciplinary care plans shall establish certain care goals and objectives with specific timeframes.
-
The interdisciplinary care plan shall be presented at a local subcommittee to ensure the documented action items are being completed.
-
-
-
References
-
Health Care Department Operations Manual, Chapter 3, Article 1, Section 3.1.3, Care Teams and Patient Panels
-
-
Revision History
-
Effective: 10/2021
-
3.1.22 Compassionate Release
-
Policy
-
California Department of Corrections and Rehabilitation (CDCR) and California Correctional Health Care Services (CCHCS) shall refer to the court a patient who meets the medical and statutory criteria for Compassionate Release and follow the processes set forth in Penal Code (PC) 1172.2.
-
-
Purpose
-
To outline the process for CDCR and CCHCS to refer a patient to the court for compassionate release consideration.
-
-
Responsibility
-
Statewide
-
CDCR and CCHCS departmental leadership at all levels of the organization, within the scope of their authority, are responsible for ensuring that administrative, custodial, and clinical systems are in place, along with appropriate tools and training to ensure that patient referrals for compassionate release are initiated and processed for all patients who meet eligibility criteria.
-
-
Regional
-
Regional Health Care Executives and the Division of Adult Institutions Regional Associate Directors are responsible to maintain this policy at the subset of institutions within an assigned region.
-
-
Institution
-
The Chief Executive Officer and Warden, or their designees, are responsible for the implementation, monitoring, and evaluation of this policy and procedure at their assigned institution.
-
-
-
Procedure
-
Primary Care Provider Referral
-
The Primary Care Provider (PCP) shall identify patients who meet the medical criteria for compassionate release set forth in PC 1172.2(b), which is either the patient:
-
Has a serious and advanced illness with end-of-life trajectory (examples include, but are not limited to, metastatic solid-tumor cancer, amyotrophic lateral sclerosis, end-stage organ disease, and advanced end-stage dementia); or
-
Is permanently medically incapacitated with a medical condition or functional impairment that renders them permanently unable to complete basic activities of daily living including, but not limited to, bathing, eating, dressing, toileting, transferring, and ambulation, or has progressive end-stage dementia and that incapacitation did not exist at the time of the original sentencing.
-
-
A patient, family member, or designee, may independently request consideration of a patient for compassionate release by providing a written request to the Chief Medical Executive (CME) at the patient’s institution. Upon receipt of a request, the CME, or designee, shall consider whether the patient meets the criteria set forth in (d)(1)(A) above and document the consideration in the patient’s health record.
-
If the PCP determines a patient meets the medical criteria for compassionate release, the PCP shall initiate the compassionate release process:
-
The PCP shall complete the CDCR 128-C, Medical-Psychiatric-Dental Chrono;
-
Nursing Services shall facilitate the patient in completing the CDCR 7385-CR, Authorization of Release of Protected Health Information – Compassionate Release; and
-
The PCP shall submit the CDCR 128-C and CDCR 7385-CR to the CME for consultation and notify the Headquarters (HQ) Complex Care Team of submission to the CME by emailing the CDCR 128-C to CCHCSComplexCare@cdcr.ca.gov.
-
-
Within two calendar days of the PCP’s request for consult, the CME, or designee, shall consult on the referral and either:
-
Deem the referral incomplete, document the reason(s) on the CDCR 128-C and notify the PCP, HQ Complex Care Team, and patient; or
-
Concur with the referral, submit the CDCR 128-C and CDCR 7385-CR to the Classification and Parole Representative (C&PR), and notify the PCP and the HQ Complex Care Team. This referral initiates the 45-calendar day window permitted by law within which a patient who meets the requirements of the law shall be referred to the court.
-
-
Within two business days of receipt of a referral, the C&PR shall determine whether the patient’s current commitment offense disqualifies the patient from compassionate release consideration under PC sections 1170.02 and 1172.2(o) and notify the Classification Services Unit (CSU) of the eligibility determination.
-
If the patient is eligible for compassionate release, the CSU shall submit the CDCR 128-C to the Director.
-
If the patient is ineligible for compassionate release, the C&PR, or designee, shall document on a CDCR 128-B, General Chrono, and notify the CME, patient, CSU, and the HQ Complex Care Team. A copy of the CDCR 128-B shall be saved in the central file.
-
-
-
Director of Health Care Services Review
-
Within 14 calendar days of receipt of a referral, the Director of Health Care Services (Director) shall review the referral.
-
If the Director concurs that the patient meets the medical criteria for compassionate release, the Director, or designee, shall notify the Warden, CSU, and C&PR to continue processing the referral.
-
If the Director does not concur that the patient meets the medical criteria for compassionate release, the Director, or designee, shall document the reason(s) for the decision and notify the PCP, C&PR, CME, CSU, HQ Complex Care Team, and patient via a denial letter.
-
-
Classification and Parole Representative Patient Notification and Preparation of Referral of Packet
-
Within 48 hours of receipt of the Director’s notification, the C&PR, or designee, shall do the following:
-
Meet with the patient and explain the compassionate release referral process.
-
Offer the patient the opportunity to complete a CDCR 3038, Notification and Authorization to Incarcerated Person Regarding Compassionate Release, whereby they may designate a family member or other outside agent to be notified of their medical condition and prognosis and to inform that person regarding the compassionate release process. If the patient does not appear to understand, the patient’s emergency contact shall be notified.
-
Offer the patient the opportunity to complete a CDCR 3039, Waiver of Defendant’s Physical or Remote Presence at Compassionate Release Hearing. If the patient does not appear to understand, the patient’s emergency contact shall be notified. The C&PR, or designee, shall indicate such on the CDCR 3039.
-
-
If the patient would be released on parole, the C&PR shall notify the Division of Adult Parole Operations (DAPO) of the patient’s referral and release plan. DAPO shall confirm the plan complies with parole residency restrictions.
-
Within 15 calendar days of receipt of Director’s notification, the C&PR shall prepare a referral packet and submit it to the CSU. The packet shall include, at a minimum, the following:
-
A Case Factor Summary in which the Correctional Counselor summarizes relevant information from documents in the patient’s central file including, without limitation, information regarding the current commitment offense, prior criminal history, institutional behavior, work and education assignments, participation in self-help activities, victim notifications, registration requirements, and known parole residency restrictions.
-
A CDCR 3038.
-
A CDCR 3039.
-
A CDCR 128-C.
-
A CDCR 7385-CR.
-
-
The C&PR shall obtain the Warden’s signature and submit the compassionate release referral packet to the CSU.
-
-
CSU Submission
-
Within 11 calendar days of receipt from C&PR, the CSU shall coordinate with the Director’s office to submit the referral letter and packet to the appropriate court, the District Attorney’s office and the Public Defender’s office. The CSU shall notify CDCR parties including the C&PR, the CME, the HQ Complex Care Team, the Transitional Case Management Program (TCMP) Program Management Unit (to request care coordination for the patient, including Medi-Cal preapproval), the Statewide Mental Health Program Pre-Release Coordinator and the Office of Victim and Survivor Rights and Services.
-
-
Sentencing Court Review and Notification of Court Decision
-
Within ten calendar days of receiving a referral for compassionate release, the sentencing court is expected to hold a hearing. CDCR shall facilitate a patient’s or the legal representative’s timely request to attend the hearing remotely. The PCP, CME, or designee, shall be available as necessary throughout the recall and resentencing proceedings.
-
Case Records staff shall follow up with the sentencing court within 15 calendar days of submitting the referral for compassionate release if a response is not received from the sentencing court.
-
Additional follow up with the sentencing court shall occur the day after any court hearings until the court’s decision is complete.
-
-
Upon reaching a decision, the court notifies CSU and the Case Records Manager at the institution. The C&PR, or designee, shall notify the patient of the decision. The court mails the original court order to the attention of the Case Records office at the institution. Case Records staff shall scan the order into the Electronic Records Management System. If the court has approved the release, CDCR and CCHCS shall initiate processing the release.
-
-
Patient Release
-
Pursuant to PC section 1172.2(l), if the sentencing court grants the recall and resentencing application, the patient shall be released by the CDCR within 48 hours of receipt of the court’s order, unless a longer time period is agreed to by the patient. If the patient has agreed to waive the 48-hour release requirement, the CDCR shall request the sentencing court include in its order that the patient shall be released within 30 calendar days to allow for the coordination of their housing and medical needs in the community to a location where access to care is available.
-
Upon release:
-
The Warden, or designee, shall ensure the patient has in their possession a state identification card or a photo prison identification card and all of their property.
-
The CEO, or designee, shall ensure the patient has in their possession a discharge medical summary, pertinent health records, appropriate medication, durable medical equipment, and has been provided TCMP services.
-
-
-
Tracking and Reporting
-
CCHCS shall maintain a compassionate release tracking log to ensure all referrals are reviewed and processed in a timely manner. This tracking log shall include the data required for reporting to the California Judicial Council set forth in the PC section 1172.2(p).
-
-
-
References
-
California Constitution, Article 1, Section 28(b)
-
California Penal Code, Part 1, Title 15, Chapter 2, Section 646.92
-
California Penal Code, Part 1, Title 16, Section 667(e)(2)(C)(iv)
-
California Penal Code, Part 2, Title 7, Chapter 4.5, Article 1, Section 1170
-
California Penal Code, Part 2, Title 7, Chapter 4.5, Article 1.5, Section 1172
-
California Penal Code, Part 2, Title 7, Chapter 4.5, Article 1, Section 1170.18 (c)
-
California Penal Code, Part 3, Title 1, Chapter 8, Article 1, Section 3007.05
-
California Penal Code, Part 3, Title 1, Chapter 8, Article 3, Section 3058.8
-
-
Revision History
-
Effective: 08/20/2025
